Abstract
The role of glutamate in migraine treatment has not been much studied, even if this amino acid seems to be crucial in the pathogenesis of migraine. Our aim was to determine if there were differences in the plasma levels of glutamate between migraine patients and control subjects and if plasma levels of glutamate in migraine patients modified after 8 weeks of prophylactic treatment. We studied 24 patients with diagnosis of migraine without aura according to International Classification of Headache Disorders, 2nd edn criteria, and 24 age- and sex-matched healthy subjects, as controls. In migraineurs the level of glutamate was measured before and after 8 weeks of prophylactic treatment (topiramate 50 mg/ day, five patients; amitriptyline 20 mg/day, seven patients; flunarizine 5 mg/day, seven patients; propranolol 80 mg/day, five patients). Venous blood samples were taken in the morning, after overnight fasting, and at least 3 days after the last migraine day. Glutamate levels were measured by means of a fluorimetric detector high-pressure liquid chromatographic method. Plasma levels of glutamate were significantly higher in migraine patients—either before (61.79 ± 18.75 μmol/l) or after prophylactic treatment (17.64 ± 5.08 μmol/l)— than in controls (9.36 ± 2.1 μmol/l) (P < 0.05, ANOVA followed by Newman-Keuls' test). After prophylactic treatment, with headache frequency reduced, plasma glutamate levels were significantly lower in the same patient with respect to the prior baseline level (P < 0.0001, Student's t-test for paired data), without any differences depending on the kind of prophylactic drug. Effective prophylactic treatments reducing high glutamate plasma levels found in migraine patients could act on the underlying mechanism that contributes to cause migraine. Plasma glutamate level monitoring in migraine patients might serve as a biomarker of response to treatments and as an objective measure of disease status.
Introduction
Migraine affects 10% of the global adult population. This disorder can cause significant disability and suffering and it reduces patients' quality of life and workplace productivity (1). Even if not life threatening, episodic migraine progresses in some individuals to chronic migraine (2), which causes even more disability and suffering (3). Moreover, a relationship between migraine and cardiovascular and cerebrovascular diseases has been suggested, and young patients suffering from migraine with aura are at increased risk of stroke (4–6). For migraine management acute and prophylactic treatments are available (7). Prophylactic treatment is currently and predominately focused on reducing the frequency of migraine attacks (8). It is not known whether prophylactic treatment can also reduce the likelihood of developing chronic migraine and the increased risk of cardiovascular disease.
Migraine, like stroke, is characterized by a glutamatergic homeostasis disorder and, in particular, by an excessive glutamatergic signal (9, 10). Glutamate is the major excitatory neurotransmitter in the central nervous system (CNS) (11) and, through activation of N-methyl-D-aspartate (NMDA) receptors, seems to be implicated in the triggering, propagation and duration of cortical spreading depression (CSD), the presumed substrate of migraine with aura (12). Furthermore, NMDA-mediated glutamatergic transmission is probably implicated in the activation of the trigeminovascular system (13) and can therefore cause the clinical symptoms of migraine attack and central sensitization (9, 14).
Higher glutamate levels than in controls have been found in plasma, platelets, saliva and cerebrospinal fluid (CSF) of migraine patients (15–23). In particular, higher plasma glutamate levels have been observed in patients suffering from migraine with aura during the attack (16). Also in intercritical periods, plasma glutamate concentrations have been reported to be higher in subjects suffering from migraine without aura than in control subjects (18, 23) and in patients suffering from tension-type headache (19). Platelet levels of glutamate have been found increased in patients suffering from migraine with aura (17). Glutamate levels significantly higher than in controls have been measured in the CSF of chronic migraine patients (21, 22). These data have been considered as suggestive of the cortical hyperexcitability typical of migraine and as clearly proving the role of glutamate in the pathogenesis of migraine (9, 11, 24). In contrast, the role of possible modifications of glutamate levels in migraine treatment and, even more, in the course of prophylactic treatment, has been studied to a lesser extent. It may be surmised that prophylactic treatment, by reducing the frequency of attacks, should also modify the levels of glutamate. Our study therefore had two objectives, i.e. to assess (i) if there were differences in migraine patients' and control subjects' plasma glutamate levels; and (ii) if glutamate levels changed in the same migraine patient after 8 weeks' treatment with different first-choice prophylactic drugs.
Patients and methods
Subjects and procedures
We studied 24 subjects (Table 1), all White, suffering from episodic migraine without aura, according to the International Classification of Headache Disorders, 2nd edn (ICHD-II, 2004) criteria (25), and 24 age- and sex-matched healthy subjects as control group. Migraine patients were recruited among those consecutively attending during May and June 2007 the out-patient service of the Headache Centre of the University Hospital of Modena. We included both male and female migraine patients, between 18 and 65 years old, to whom the specialist of the Centre had prescribed a prophylactic treatment (topiramate 50 mg/day, five patients; amitriptyline 20 mg/day, seven patients; flunarizine 5 mg/day, seven patients; propranolol 80 mg/day, five patients). In this sample, the average (±
Migraine patients' and control subjects' characteristics

Patients' characteristics were recorded by medical examination, and migraine frequency was determined by the patients' diaries kept during the months preceding the visit to the Headache Centre.
Migraine patients were studied before and after 8 weeks of prophylactic treatment. Venous blood samples for the assay of glutamate concentrations were taken in the morning, after overnight fasting and after an interval of at least 3 days from the last migraine day.
Blood samples (2 ml) were collected in vacutainer tubes containing K2-ethylenediamine tetraaceticacid. Plasma separation was performed at 1000
Assay of glutamate
Glutamate concentrations were measured on plasma, by means of a slightly modified fluorimetric detection high-pressure liquid chromatographic method (26). Stock solutions of glutamic and 2-aminoadipic acid were prepared, 1 m
Calibration and quality control samples were freshly prepared in 20% methanol, diluting the glutamate working solution to yield 400-μl volume samples, to be processed as previously described. For calibration, a six-point standard curve was used, with glutamate concentration being respectively 100, 50, 25, 10, 5 and 3 μ
High-performance liquid chromatography was performed on a Beckman System Gold apparatus consisting of a Programmable Solvent Module 126, an Autosampler 508 and a fluorimetric detector Jasco FP-2020plus (Beckman, Fullerton, CA, USA). Separation was carried out on a reversed-phase Synergi Hydro-RP 4 μ
Data analysis
The data collected were inserted into a fit database. Plasma glutamate concentrations and the frequency of migraine days before and after 8 weeks of prophylactic treatment were compared by Student's t-test for paired data. Analysis of variance (
Results
Of the 24 patients initially recruited, one female patient, who had been prescribed propranolol 80 mg/day, did not come back for the second blood sample, after 8 weeks. This patient's glutamate level in the first blood sample was 71.3 μmol/l. Another patient, having a starting glutamate level of 56.5 μmol/l and four migraine days per month, had discontinued flunarizine treatment after only 5 days because of excessive sleepiness. After 8 weeks, this patient's frequency of headache had slightly increased (5 days per month), and the glutamate level was also higher (67.9 μmol/l) than that measured in the first sample.
After 8 weeks of prophylactic treatment (Fig. 1), 22 patients had both mean plasma glutamate levels (17.64 ± 5.08 μmol/l) and mean number of migraine days per month (2.64 ± 1.14) significantly lower than before treatment (glutamate levels 61.79 ± 18.75 μmol/l; number of migraine days per month 5.64 ± 1.22) (P < 0.001, Student's t-test for paired data). However, migraine patients' plasma glutamate levels, both before and after treatment, were significantly higher than those of controls (9.36 ± 2.1 μmol/l) (P < 0.05,

Plasma concentrations (mean ±
Plasma glutamate levels and frequency of migraine days (Table 2) significantly decreased after 8 weeks' treatment with each of the four prescribed prophylactic drugs (Student's t-test for paired data), without differences among the drugs used (
Plasma concentrations (mean ±
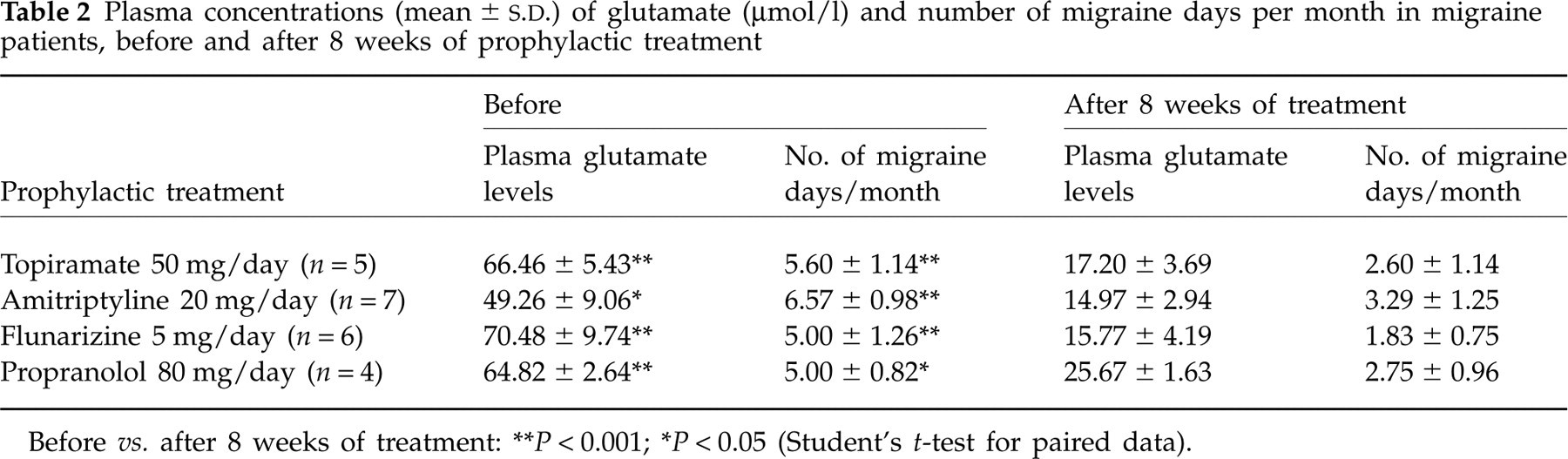
Before vs. after 8 weeks of treatment:
∗∗ P < 0.001;
∗ P < 0.05 (Student's t-test for paired data).
Discussion
The results of our study support the idea that migraine is characterized by a dysfunction of glutamatergic brain metabolism (28, 29). The patients suffering from migraine without aura that we studied (Fig. 1) had different biochemistry from controls, even during headache-free periods, when their conditions were apparently normal. Indeed, they had significantly higher glutamate levels than control subjects. Moreover, 8 weeks' prophylactic treatment (Table 2) with four different drugs (topiramate, amitriptyline, flunarizine and propranolol) significantly reduced in the same patient both the frequency of migraine days and glutamate levels in comparison with prior values, without any differences among the drugs used. However, plasma glutamate levels continued to be significantly higher (before treatment, 61.79 ± 18.75 μmol/l; after treatment, 17.64 ± 5.08 μmol/l) than those of control subjects (9.36 ± 2.1 μmol/l) (P < 0.05,
The mechanism which makes most antimigraine prophylactic drugs effective is not completely understood. Our results support the hypothesis that the prophylactic treatments which proved to be effective influence glutamatergic transmission (30). This could increase the stimulation threshold and/or slow the progression of CSD (31). Indeed, prolonged treatment with different prophylactic drugs (topiramate, valproate, propranolol, amitriptyline or methysergide) reduces the number of potassium-evoked CSDs and elevates the electrical stimulation threshold for the induction of CSD in rats (31).
Our data must be interpreted cautiously, considering the limited size of the sample. This study was carried out in the clinical setting of a specialized Centre, and the prophylactic treatment was prescribed depending on the patients' clinical characteristics and the severity of their migraine. If we had carried out a longer study we could perhaps have obtained a further reduction in headache frequency, and perhaps also in plasma glutamate levels, by increasing the dose of topiramate to 100 mg/day. The patients treated with amitriptyline (Table 2) had lower baseline glutamate levels. We are not able to explain this difference according to medical history and migraine characteristics, which were similar in the patients treated with other medications. We cannot exclude that these seven out-patients could have taken other medications or suffered from other disorders (without having respectively declared or known them), which could have affected their plasma glutamate levels. Since there was no placebo group, we cannot be sure that the decrease in glutamate levels was caused by the prophylactic treatment. However, after 8 weeks, the only patient who discontinued treatment with flunarizine early because of an adverse event (sleepiness) had increased both migraine frequency and glutamate plasma level with respect to prior values. In contrast, in the other six patients (Table 2), who had regularly taken flunarizine 5 mg/day for 8 weeks with migraine improvement, plasma glutamate levels were significantly lower after prophylactic treatment than before. Flunarizine, a calcium channel blocker, could inhibit the release of glutamate just blocking voltage-dependent calcium channels (32).
The high plasma glutamate levels that we detected in migraine patients could depend on altered functioning of platelets, such as excessive release of the amino acid or impaired up-take. Indeed, platelet glutamate uptake has been reported to be reduced in migraine without aura patients with respect to control subjects (23). Platelets have high-affinity glutamate transporters which re-uptake glutamate from the blood (33), express vesicular transporters that store glutamate in specific secretory vesicles and release it after aggregating (34). Evidence of increased platelet activation, even during the headache-free period, has been reported in migraine without aura (35). Platelet activation and release of the constituents of the granules are thought to play an important role in the pathophysiology of migraine, stroke and other excitotoxic disorders (36).
The changes in migraine patients' glutamate levels after prophylactic treatment have been observed in the peripheral compartment. Under normal conditions, the passage of polar solutes, such as glutamate, through the blood–brain barrier (BBB) is limited (37). Nevertheless, with the use of microdialysis, significant elevation in brain extracellular fluid glutamate concentration has been found after large systemic doses of monosodium glutamate (38). Conversely, it has also been supposed that raised levels of excitatory amino acids in brain tissue (which have been shown to occur in dysfunctional states such as hypoxia and epilepsy) could reach the systemic circulation (39). Moreover, it has been reported that stress situations greatly increase the permeability of the BBB, so that plasma levels of drugs and amino acids could affect the CNS (40). CSD could alter the permeability of the BBB by the activation of proteolytic enzymes known as matrix metalloproteinases (MMPs) (41). In particular, a significant increase in MMP-9 has been detected in plasma during migraine attacks (42), and also in patients suffering from migraine without aura during headache-free periods (43).
In conclusion, migraine patients' high plasma glutamate levels could reflect cortical neuronal hyperexcitability—also present in the interictal period—that is now widely recognized as pivotal in predisposing to acute migraine attacks (44). Other studies in chronic migraine patients comparing plasma glutamate levels, either before or after successful or unsuccessful treatment, could give further information about the role of glutamate in the process of migraine chronification.
Our results have shown that effective prophylactic treatments reduce plasma glutamate levels and suggest that prophylactic treatments could act on one of the underlying mechanisms that contribute to cause migraine, and might therefore modify the progression of this disorder. Finally, the monitoring of plasma glutamate levels outside migraine days might be used as a marker of response to treatments and an unbiased measure of disease status.
