Abstract
We investigated the consistency between the headache diagnosis based on medical history and three treated headache episodes diagnosed based on a diary. In a randomized double-blind study including individuals with either migraine or tension-type headache (TTH) we showed significant superiority of the fixed combination of acetylsalicylic acid + paracetamol + caffeine over the combination without caffeine, the single preparations, and placebo in the treatment of headache. A neurologist performed a classification of the usual headache episodes and each of the three treated ones in a blinded fashion based on a structured questionnaire. This was done for the 1734 patients included in the efficacy analysis who usually treated their episodic TTH or migraine attacks with non-prescription analgesics. The overall percentage of patients with migraine and TTH remained relatively stable. The treated headache episodes were between 75 and 77% migraine, 18–20% were TTH and 5–7% could not be classified. We observed some shift in headache type within patients from prior history and in treated attacks. In 60% of patients all three treated episodes were of the type initially diagnosed by the neurologist by history (56% migraine and 4% episodic TTH). Of those with an initial diagnosis of migraine, 24% had at least one attack meeting criteria for TTH. Of patients with an initial diagnosis of TTH, 54% had at least one attack meeting the diagnostic criteria for migraine. Our results demonstrate that an initial headache diagnosis does not accurately predict the headache type treated in a randomized trial. Symptom features of treated headaches should be captured to ensure that the attack is of the type targeted by the clinical trial. The International Headache Society Guidelines for controlled clinical trials should be updated accordingly.
Keywords
Introduction
A number of analgesic over-the-counter (OTC) agents—either as single preparations or as compound preparations—have shown their efficacy in the treatment of headaches in randomized, controlled clinical trials. These studies have to comply with the relevant legal requirements and International Conference on Harmonization Good Clinical Practice guidelines as well as the International Headache Society (IHS) Guidelines for controlled clinical trials (1, 2). On the other hand, the patients need to be representative of the patient population in which the investigated therapy is to be used under conditions of everyday life. This is also requested by various guidelines of the European [European Medicines Agency (EMEA)] and USA authorities [Food and Drug Administration (FDA)] (3–6).
The majority of headache patients, whether they suffer from migraine or tension-type headache (TTH), do not see a doctor for headache on a regular basis if at all. Many of these patients treat their headache successfully with OTC analgesics. Several studies have targeted an OTC headache population by recruiting through consumer advertisements, by random-digit dialling methods and by careful selection of patients in general practice who treat successfully with OTC analgesics (7–12).
Headache was diagnosed in all these studies based on the medical history according to the IHS diagnostic criteria [(ICHD-I (13); ICHD-II (14)]. For the most frequent diagnosis in these headache patients ICHD-II states: ‘Differentiating between 1.1 Migraine without aura and 2.1 Infrequent episodic tension-type headache may be difficult. Therefore at least 5 attacks are required’. The characteristics of at least five attacks can only be analysed post hoc from medical history if at all, which is not what ICHD-I/ICHD-II intends. This reveals a discrepancy between the demands for the rather rigid application of the diagnostic criteria and the demand for the inclusion of headache patients typical of the use of OTC analgesics.
A solution to this problem was seen in a new methodological approach for studies with OTC analgesics: being aware of the difficulties in separating migraine and TTH in cases of moderate headache, we decided to include both and to classify single headache episodes (15). This approach of the classification of the actually treated headache episodes—in addition to the classification of the headaches from the medical history—was used also for the much larger (1743 patients) randomized six-arm study to investigate combination analgesics with dual therapy and monotherapy (16).
Prior research has suggested that about one-third of patients diagnosed by a doctor with disabling TTH proved to have at least some attacks that meet criteria for migraine based on their headache diaries (17). In the OTC headache population is not clear if initial diagnosis will predict headache type as recorded in a diary. Herein, we investigate the relationship between the initial headache diagnosis and the type of headache treated in the context of the randomized trial.
Methods
The study was designed as a randomized, placebo-controlled, double-blind, multicentre parallel group trial. Patients were randomly allocated to one of six treatment groups: the fixed combination of acetylsalicylic acid + paracetamol + caffeine, the combination without caffeine, the single preparations, and placebo. For inclusion and exclusion criteria and details on efficacy, tolerability and safety end-points, see Diener et al. 2005 (16).
The study comprised the treatment of three headache episodes. Patients treated their first headache attack with their usual OTC analgesic. The following two headache episodes were treated with the investigational study medication. The patients were to answer standardized questions on their headache diaries. During the baseline examination the investigators completed the same structured questionnaire as the patients in order to record information on the patients' headache history. An independent neurologist performed a blinded classification of the usual and each of the three treated headache episodes according to the IHS Headache Classification 2.1, (b) as TTH or according to the IHS Headache Classification 1.1 and 1.2.1 (a), (b), (c) as migraine. It was not intended to diagnose aura symptoms. The headache type was recorded as unclassified in case an unambiguous categorization was not possible.
The questionnaire comprised 15 criteria for the diagnosis of headache, each ticked yes or no by the investigator in the case report form or by the patient in their diary: pulsating, penetrating, gnawing pain; dull compressive pain; bilateral; unilateral and/or changing pain; bilateral, but concentrated on one side; nausea; retching; vomiting; sensitivity to noise; sensitivity to light; sensitivity to smell; chill; visual disturbances; flickering vision; pain exacerbated on exercise.
Statistical analysis
Graphical displays were given to describe the frequency distribution of the criteria for the diagnosis of headache as recorded in the questionnaires. The consistency of the usual headache type and the headache types in the treatment phase was explored by means of shift tables. The analysis was based on the patients included in the full analysis set.
Results
Patient characteristics
The full analysis set comprised 1743 patients recruited in 133 centres (16). Of these, 76% were women and 24% men with a median age of 38 years (range 16–72 years). Without treatment, the usual pain intensity was severe or very severe in 62% and moderate in 37% of patients. The severity of pain was associated with disability of performing usual daily activities.
Differences between treatment groups
The superior efficacy of the triple combination acetylsalicylic acid + paracetamol + caffeine could be shown for all efficacy end-points, such as the time to 50% pain relief (primary end-point), time until reduction of pain intensity to 10 mm, pain intensity difference, weighted sum of pain intensity difference (% SPID weighted), extent of impairment of daily activities and patient's global efficacy assessment (16).
Diagnostic criteria and overall frequency of headache types
Figure 1 shows the percentage of patients suffering from a particular symptom described in the diagnostic criterion separately for either headache type based on the questionnaires provided to the investigators to assess usual headache from medical history and to the patients to assess the treated headache episodes. The profile of diagnostic criteria clearly differed between migraine attacks and TTH episodes. Migraine attacks occurred frequently for the items: pulsating, penetrating, gnawing pain; unilateral and/or changing pain; nausea; sensitivity to noise; sensitivity to light, and pain exacerbated on exercise. For TTH episodes high frequencies were found for the items: dull compressive pain; bilateral, and also for pain exacerbated on exercise. Overall, there was high consistency between the usual and treated headache episodes within the group classified as migraineurs and within the group classified as TTH patients. Consistently, the percentage of patients with migraine (between 75% and 77%) and episodic TTH (18–20%) was similar in all treated headache episodes (Table 1). The slightly higher percentage of patients classified as usually suffering from migraine (84%) based on the headache history compared with the diagnosis in the treatment phase could be traced back to the profile of diagnostic criteria.
Frequencies (%) of usual and treated headache types diagnosed at screening and before each treatment phase (full analysis dataset)

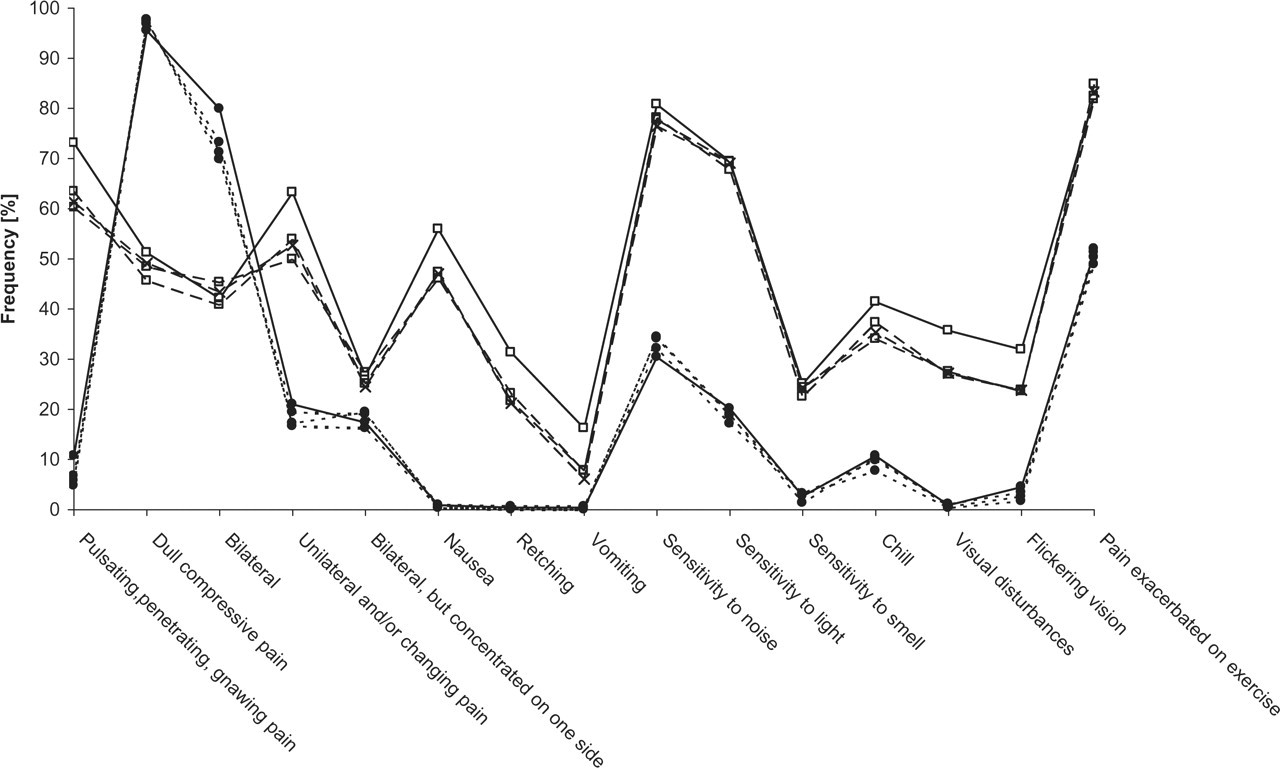
Profile of diagnostic criteria by headache type and treatment phase based on the questionnaire completed by the investigators and patients. Open square, migraine headache; dot, episodic tension-type headache; solid line, usual headache type (medical history); dotted line, headache classification in treatment phase.
Shift in headache type within patients
Whereas the overall percentage of patients with migraine and TTH remained relatively stable, some shift of headache type within patients was observed. In total, 60% of patients had identical usual and treated headache type (56% migraine and 4% episodic TTH, Table 2). Headache diagnoses differed in 13% of patients in one episode from usual headache type, in 9% of patients in two and in 5% of patients in all three treated headache episodes. Headache type could not be classified in 13% of the patients either from assessment at baseline or during the treated headache episodes. Restricting to the three treated headache episodes, 58% of patients were always classified as migraineurs and 7% as TTH patients (Table 3). There were 23% of patients with inconsistent headache type and 12% who could not be classified in the three treated headache episodes. Among individuals with migraine as the initial diagnosis 24% had at least one attack of TTH. Among those with TTH as the initial diagnosis, 54% had at least one attack of migraine.
Frequencies (%) of patients by number of identical usual and treated headache types sorted by usual headache type (patients with complete diagnoses of usual and treated headache types)

Frequencies (%) of patients by number of identical headache types during the three treatment phases (patients with complete diagnoses of all treated headache types)

Discussion
This largest headache study with OTC analgesics with its 15-item diary allowed the classification of the headache type according to both medical history and the independent diagnostic assessment of three consecutive headache episodes treated in the study using the ICHD-I diagnostic criteria valid at the time when the study was conducted. This study was conducted using the ICHD-I criteria for migraine and for TTH as it was conducted prior to the publication of ICHD-II. The ICHD-II criteria for migraine without aura remain unchanged and those for TTH differ largely because of the introduction of frequency categories. Had we used ICHD-II criteria instead, we believe the results would be virtually identical. Based on medical history, the majority of patients (84%) met the ICHD-I criteria for migraine without aura (1.1) or migraine with aura (1.2). In addition, 13% had episodic TTH (2.1); 3% could not be classified. The treated headache episodes were between 75 and 77% migraine, 18–20% were episodic TTH and 5–7% could not be classified. The 15-item questionnaire enabled us to differentiate clearly between migraine and TTH (15). The determined migraine profile agrees very well with the diagnostic criteria for migraine without aura. For TTH, the frequent occurrence of the items dull compressive pain, bilateral pain and the correspondingly low values for the other items agree very well with the diagnostic criteria of episodic TTH; somewhat less so the comparatively high value for the item pain exacerbated on exercise.
The important question for the present analysis was whether the classification deducible from medical history would remain for the treated headache episodes, in particular considering the fact that in almost all headache studies classification was done based on the medical history data. Only for 60% of the treated patients does the classification of the medical history data correspond with that of the three treated headache episodes.
Inconsistent headache types were recorded in 27% of the patients. Headache type could not be classified in 13% of patients either from assessment at baseline or during the treated headache episodes. It can consequently be assumed that in various headache studies with OTC analgesics the treated headache episodes do not always correspond to the headache diagnoses the patients were assigned based on medical history.
If a migraine sufferer experiences an attack of phenotypic TTH, that is entirely consistent with clinical experience and expectation; this occurred in 24% of the migraine sufferers in the study. Patients with TTH should not experience migraine headaches except as a rare event. If they have five lifetime attacks of migraine they should be so classified. In the present sample, an astonishing 54% of patients with TTH had an attack of migraine. A similar phenomenon was observed in a smaller sample of episodic TTH sufferers selected for high levels of headache-related disability (17). In aggregate, these findings suggest that a diagnosis of TTH may not reflect the range of headaches a patient experiences and that migraine sufferers often have phenotypic TTH.
The Guidelines for controlled trials of drugs in migraine (1) demand that ‘the diagnostic criteria should conform to those of the IHS’ and comment: ‘Diagnostic criteria of the IHS should be adhered to strictly …. For clinical drug trials, however, requirements are more rigid than in clinical practice’. Accordingly, the Guidelines for trials of drug treatments in tension-type headache (2) also contain these demands, while the comments explain: ‘Although the nosological borders of TH are still vague, the present IHS criteria are the best available and should be strictly adhered to’, although ‘diagnosis may be uncertain, especially in TH of recent onset, or at the first consultation, because of the lack of specificity of diagnostic criteria or the presence of uncommon features’ (2). Headache studies with OTC analgesics will hardly fully meet these demands. Part of the problem is due to the well-founded demand that patients included in headache studies should be representative of the patient population for whom the therapy is intended. This request is also made by the relevant EMEA and FDA guidelines (3–6). For example, the EMEA Note for Guidance for migraine (6) explicitly explains: ‘Migraine sufferers attending specialist clinics may not be representative of the larger number of patients seen by primary care physicians. Neither group is likely to match those in the general population who usually do not seek medical advice. Clinical trials need to recruit widely in order to reflect the population who will use the agent when marketed’.
Consequently, it should be recommended for headache studies with OTC analgesics, first, to include in the study patients representative of OTC use and, second, to classify not only medical history, but also the treated headache episode by means of suitable instruments based on the ICHD-II. The IHS Guidelines for controlled clinical trials should be updated accordingly.
Competing interests
H-C.D. has received honoraria for participation in clinical trials, contribution to advisory boards or oral presentations from: Addex Pharma, Allergan, Almirall, AstraZeneca, Bayer Vital, Berlin Chemie, CoLucid, Boehringer Ingelheim, Bristol-Myers Squibb, GlaxoSmithKline, Grünenthal, Janssen-Cilag, Lilly, La Roche, 3M Medica, MSD, Novartis, Johnson & Johnson, Pierre Fabre, Pfizer, Schaper and Brümmer, SanofiAventis, Weber & Weber. Financial support for research projects was provided by Allergan, Almirall, AstraZeneca, Bayer, GSK, Janssen-Cilag, Pfizer. Headache research at the Department of Neurology in Essen is supported by the German Research Council (DFG), the German Ministry of Education and Research (BMBF) and the European Union. H-C.D. has no ownership interest and does not own stocks of any pharmaceutical company. L.P. has no conflict of interest. V.P. has received honoraria for participation in clinical trials, contribution to advisory boards or oral presentations from: Addex Pharma, Allergan, Almirall, AstraZeneca, Bayer Vital, Berlin Chemie, Boehringer Ingelheim, Bristol-Myers Squibb, GlaxoSmithKline, Janssen-Cilag, Lilly, 3M Medica, MSD, Novartis, Pfizer, Schaper and Brümmer, SanofiAventis, Weber & Weber. Financial support for research projects was provided by Allergan, Almirall, AstraZeneca, Bayer, GSK, Janssen-Cilag, Pfizer. V.P. has no ownership interest and does not own stocks of any pharmaceutical company. H.P. and B.A. are employees of Boehringer Ingelheim Pharma GmbH & Co. KG. R.B.L.'s disclosures are advisory board, speaker's bureau, research or educational grants, currently or in the past, related to the following pharmaceutical companies: Advanced Bionics, Allergan, AstraZeneca, Boehringer-Ingelheim, Bristol Myers Squibb, Cierra, Endo, GlaxoSmithKline, Minster, Merck, Neuralieve, Novartis, OrthoMcNeil, Pfizer, Pozen, ProEthics, St. Judes.
Footnotes
Acknowledgements
Supported by Boehringer Ingelheim Pharma GmbH & Co. KG, Germany. The study was designed by H-C.D. and V.P. The statistical analysis was performed within Boehringer Ingelheim. The authors had unlimited access to all data.
