Abstract
Adrenomedullin (ADM) is closely related to calcitonin gene-related peptide, which has a known causative role in migraine. Animal studies have strongly suggested that ADM has a vasodilatory effect within the cerebral circulation. For these reasons, ADM is also likely to be involved in migraine. However, the hypothetical migraine-inducing property and effect on human cerebral circulation of ADM have not previously been investigated. Human ADM (0.08 µg kg−1 min−1) or placebo (saline 0.9%) was administered as a 20-min intravenous infusion to 12 patients suffering from migraine without aura in a crossover double-blind study. The occurrence of headache and associated symptoms were registered regularly 24 h post infusion. Cerebral blood flow (CBF) was measured by 133Xenon single-photon emission computed tomography, mean blood flow velocity in the middle cerebral artery (VMCA) by transcranial Doppler and the diameter of peripheral arteries by transdermal ultrasound (C-scan). ADM did not induce significantly more headache or migraine compared with placebo (
Introduction
There is strong evidence of a pivotal role of calcitonin gene-related peptide (CGRP) and nitric oxide (NO) in migraine pathogenesis (1–6). However, other substances may be involved, and a member of the CGRP family, adrenomedullin (ADM), is an obvious candidate for further investigation.
ADM was discovered 10 years ago among peptides extracted from pheochromocytomas (7). Subsequently, its pharmacological and physiological properties have been defined (8). The mRNA of the peptide, originating from a single locus on chromosome 11 (9), is found in the highest concentration in endothelial cells (10) and in particular within the cerebral circulation (11). From the precursor molecule two circulating products are formed, the inactive intermediate form (ADMgly) and the active mature form (ADM). Being a secreted product of the vascular endothelium along with NO and endothelin, ADM is largely distributed according to tissue vascularity.
ADM activates a combined calcitonin receptor-like receptor (CRLR) and the receptor activity modifying protein (RAMP2), possibly RAMP3, coupled receptor and increases the formation of intracellular cAMP (12). The best-described effect of ADM is vasodilation, but the exact function in human physiology and pathophysiology, including up-regulation during inflammation, remains to be determined. In healthy volunteers, administered ADM dose-dependently decreases systolic and diastolic blood pressure (BP) and increases heart rate (HR) (13, 14). Facial flushing, conjunctival injection and mild headache have furthermore been described. Headache was observed in six of eight subjects and, respectively, in one out of 11 subjects (15, 16) after ADM administration.
ADM receptors are situated on the endothelial and smooth muscle cells of cerebral vessels and ADM is produced within cerebral endothelial cells, in contrast to CGRP, which is released from perivascular sensory nerve endings. CGRP does not seem to play an important role in the maintenance of resting tone of cerebral vessels (17), whereas ADM might do so at high plasma levels (11).
The properties of ADM and its similarity to CGRP, as well as the possible activation of CGRP receptors by ADM, make it an obvious candidate to be involved in migraine mechanisms. We therefore conducted this double-blind study to evaluate whether ADM affects the cerebral and extracranial haemodynamics and whether it causes headache or migraine in migraine suffers.
Methods and patients
Design and patients
A placebo-controlled, double-blind, crossover design was applied in the study. The number of patients (
The migraine patients all fulfilled the criteria of the International Headache Society (IHS) (19) for migraine without aura (MoA) and had a maximum of six attacks per month. A second diagnose of tension-type headache was allowed with a maximum of four episodes per month. Entry criteria were: MoA (19) and age 18–65 years. Exclusion criteria were: present or previous cardiovascular, cerebrovascular, endocrine or neurological disorder, prophylactic treatment for migraine and concomitant medication that might influence the outcome measures.
The study population consisted of 10 women and two men with a mean age of 41 years (range 28–50 years). Written informed consent was obtained before randomization. The Ethical Committee of Copenhagen (KA01115s) and the Danish Medicines Agency (2612–1771) approved the study, which was conducted according to the Helsinki II declaration. A balanced randomization was performed using Medstat©. A double-blind, crossover design was used. Placebo (0.9% NaCl) or human ADM (0.08 µg kg−1 min−1) was administered intravenously in a balanced randomized order on the two different trials days. h-ADM(1–52) was purchased from Clinalfa AG (Bubendorf, Switzerland) and diluted in 0.9% isotonic saline immediately before administration.
The choice of ADM dose was partly based on previously published studies using doses between 0.016 and 0.081 µg kg−1 min−1 and partly on a dose finding pilot study including three patients suffering from MoA. In a single trial day they received three intravenous (i.v.) cumulative doses of ADM [0.06, 0.08 and 0.1 µg kg−1 min−1 (i.v.)] as 20-min infusions separated by a 60-min wash-out period. The first patient developed severe tachycardia, increasing BP, peripheral vasoconstriction and cold sweating after the 0.1-µg dose. Therefore, the two remaining patients received only 0.06 and 0.08 µg kg−1 min−1 of ADM (i.v.), which was well tolerated.
Headache and adverse events
Every 10 min from baseline to
Cerebral blood flow measurements
Global and regional cerebral blood flow (CBF) was measured with 133Xenon inhalation and SPECT with a brain-dedicated camera (Ceraspect; DSI, Waltham, MA, USA). The apparatus consists of a stationary annular NaI crystal and a fast rotating collimator system. Each rotation took 10 s, thereby acquiring one frame in a 30-frame dynamic protocol of 133Xenon inhalation, three background, nine wash-in, 18 wash-out using the Kanno-Lassen algorithm (20). A photoelectric window of 70–100 keV was employed.
Thirty-two slices were reconstructed in a 64 × 64 matrix with each pixel measuring 0.33 × 0.33 cm using a Butterworth 1D filter (cut-off 1.5, order 6). The 32 slices were reduced to sets of eight transaxial slices generated by adding four slices together to a total slice thickness of 1.32 cm. Attenuation correction using the Chang algorithm (µ = 0.05 cm) and correction for nose artefact was performed. The output for each pixel was the ki-value and flow values were estimated from these using the partition coefficient (λ) of 0.85 ml/g (grey matter).
A Datex Normocap 200 (Dameca, Roedovre, Denmark) was used for end-tidal CO2 measurements during the CBF acquisitions. A Ceratronic XAS SM 320 (Randers, Denmark) was used for the 133Xenon administration. Each measurement lasted 5 min.
Calculations of flow in the perfusion territories of the major cerebral arteries were performed fitting standard vascular regions of interest on the five slices at 3.6, 5.0, 6.3, 7.6 and 9 cm above the orbito-meatal line. Flow in the territory of the middle cerebral artery (MCA) (rCBFMCA) was calculated as a mean of the left and right side.
Transcranial Doppler and C-scan
Transcranial Doppler (TCD) ultrasonography (2 MHz, Multidop × Doppler; DWL, Sipplingen, Germany) was used for the measurement of blood flow velocity. The recordings were done simultaneously and bilaterally as previously described, but with handheld probes (21). Along the MCA, a fixed point was found for the measurement. The fixed point was chosen as close as possible to the bifurcation of the anterior cerebral artery and MCA. The same fix-point was used for each individual and for each recording, at which the signal was optimized. Based upon the envelope curve (spectral TCD curve), a time-average mean (Vmean) over approximately four cardiac cycles (4 s) was calculated by the built-in software (TCD software for MDX 7.40x). The final measure used for each time point was an average of four cycles (VMCA). Simultaneously with the TCD recording, a mask covering the subjects' mouth and nose region was placed for the measurement of end-tidal pCO2 (PetCO2) (Datex Normocap 200; Dameca).
A high-resolution ultrasound scanner, C-scan (Dermascan C, 20 MHz, bandwidth 15 MHz; Hadsund, Denmark) was used to measure the diameter of the left temporal and left radial arteries (22). The diameter of the former was measured at the front branch of the superficial temporal artery (STA) and the latter at the wrist. To ensure that the repeated measurements with TCD and C-scan were performed in the same place, marks were drawn on the skin. After the last recording on the first trial day, the coordinates of the marks were recorded for re-use on the following trial day.
Pharmacokinetics
At the beginning of each trial day the haematocrit value was determined. Plasma samples for the analysis of ADM concentration (ADMTotal and ADMMature) were obtained four times during each trial day (
Trial procedures
The migraine patients began the study at 07.45 h with a short interview ensuring the following inclusion criteria: no migraine attack or tension-type headache for the preceding 48 h, no intake of triptans or analgesics, e.g. paracetamol, within the same period of time, no intake of ergotamine for the previous 72 h and abstinence from coffee, tea, caffeine-containing drinks and smoking tobacco for the preceding 8 h. The patients rested supine throughout the study period.
Two i.v. catheters [Optiva*2, (18G); Johnson & Johnson, Ethicon S.p.A., Italy] were inserted into the cubital veins, one for the administration of placebo or h-ADM, the other for blood sampling. The volunteers rested at least 30 min before baseline values of CBF, VMCA, temporal and radial diameter, BP, HR and electrocardiogram (ECG) were recorded. The start of infusion of h-ADM or placebo was designated time zero (
The SPECT measurements were repeated twice at
rCBFx in the area of a given artery (x) is related to mean blood flow velocity (Vmean(x)) and cross-sectional area, π × r2, of the artery.
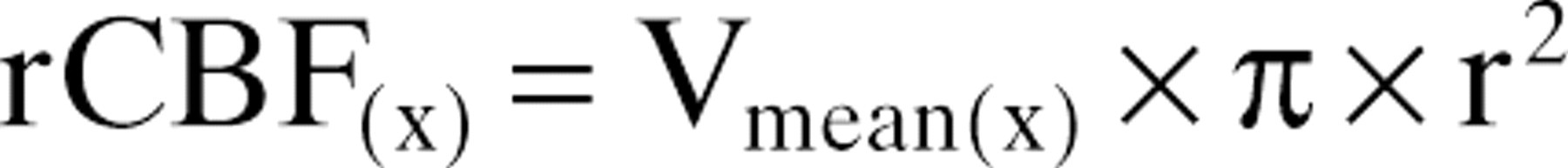
If the rCBF changes the following equation is valid:

Δ Diameter is the relative percent change in diameter; Vmean1(x) is the mean blood velocity before infusion of drugs and Vmean2(X) the velocity at a relevant time point after the infusion. The same nomination is applied for rCBF (25, 26).
Statistics
Baseline was calculated as a mean of the values at
For changes over time on each trial day global CBF, rCBFMCA, VMCA, diameter of the temporal and radial artery, BP, HR and PetCO2 were analysed by univariate analysis of variance for the factors time and subject. If a significant change was found, a
Immediate headache was defined as any headache during the first 60 min after the start of the h-ADM administration. Any headache occurring thereafter was called delayed headache. Peak values and AUCHeadache (area under the headache curve) were compared between the two trial days using Wilcoxon's signed rank test. The occurrence of headache and AEs on the two trial days was compared with McNemar's test.
Results
Baseline characteristics
No significant differences were found between the two experimental days in values of haemodynamic variables, haematocrit or start and end room temperature.
Headache and associated symptoms
Of the 12 included patients, six experienced immediate headache on days with administration of ADM and one on placebo days (

Individual curves of headache scores for all patients on days of h-ADM infusion (a) and placebo (b). The median headache score is in bold.
ADM induced flushing in all patients and placebo in one (
Pharmacokinetics
Baseline plasma concentration of ADMtotal (11.67 ± 2.43 fmol/ml and 11.09 ± 2.7 fmol/ml) and ADMmature (2.78 ± 0.68 fmol/ml and 2.77 ± 0.58 fmol/ml) were similar on the two trial days. The Cmax of ADMtotal (76.4 ± 16.6 fmol/ml) and ADMmature (24.4 ± 6.0 fmol/ml) were measured at time point

Plasma concentration of ADMMature on the two trial days; ▪, h-ADM; ▴, placebo.
Cerebral and peripheral haemodynamics
There were no significant differences in baseline values between ADM and placebo administration days (Table 1). After ADM there was no change in global CBF (
Baseline values on the two trial days with comparison using a paired

BP, blood pressure.

rCBF (ml 100 g brain tissue−1 min−1) vs. time (minutes) for, respectively: ▪, h-ADM administered; ▴, placebo. Vertical line = initiation of infusion.
PetCO2-corrected VMCA increased significantly over time after ADM (
Temporal artery diameter increased significantly compared with placebo (

Diameter change in superficial temporal (mm) artery vs. time (minutes) for, respectively: ▪, h-ADM AND; ▴, placebo.
There were no significant changes after ADM compared with placebo in systolic BP (
Discussion
The present study has demonstrated that intravenously administrated ADM at the maximal tolerated dose did not induce migraine attacks in migraine patients, indicating that ADM does not play a major role in migraine pathogenesis. ADM dilated the superficial temporal artery and affected the systemic circulation, but had no effect on CBF or diameter of MCA.
Haemodynamic effects of ADM
Meeran and co-workers administered ADM to healthy volunteers and found that a low dose of ADM (3.2 pmol kg−1 min−1) did not affect BP or HR, whereas a higher dose (13.4 pmol kg−1 min−1) resulted in a significant decrease in diastolic BP (69 ± 2 to 53 ± 2 mmHg) and a significant increase in HR (57 ± 3 to 95 ± 4 bpm) (13). With a dose of 0.05 µg kg−1 min−1 Nagaya et al. found smaller effects, but a significantly increased cardiac index and decreased pulmonary arterial pressure in patients with chronic heart failure (15). Long-term administration of AM in lower doses (2.9 and 5.8 pmol kg−1 min−1) showed a minor effect on systemic haemodynamics (14, 16). Administering 0.08 µg kg−1 min−1 for 20 min, we found that the systolic, diastolic and mean arterial BP were unaffected by ADM compared with placebo. HR was significantly increased.
The effect of ADM on human CBF or MCA diameter has not been assessed previously. Data on endogenous production within the brain are conflicting. It seems likely that cerebral endothelial cells produce a high amount of ADM (10, 27, 28). ADM has been shown to pass the blood–brain barrier (BBB) and may be involved in the regulation of BBB function (29, 30). Since ADM receptors are present on both endothelial and vascular smooth muscle cells and possess vasodilatory properties, one would expect ADM to be a strong cerebral vasodilator. However, we found no significant effect on CBF or MCA diameter. This finding contrasts with animal studies, which have all shown vasodilation and increase in CBF. Species differences may partly explain the findings. Thus, the expression of functional ADM receptors might differ between humans and rodents, and the physiological role of ADM is more likely to be regulation of BBB permeability than vasodilation (30–32).
The infusion of ADM resulted in a significant dilation of the superficial branch of the temporal artery compared with placebo. This dilation seemed to be restricted to the cephalic circulation since no effect was seen on the radial (control) artery. The dilation was associated with a concomitant flushing of the face and chest in all but one patient, and a sensation of heat was an often-reported side-effect of the ADM infusion.
Extracranial arterial dilation causes no headache
ADM did not induce a migraine headache in our study population despite its close relation to CGRP and its vasodilatory properties. A possible cause of the failure of ADM to induce migraine headache could be that the dose of ADM was too low. However, plasma ADMtotal (ADMgly + ADMmature) increased 6.5 times and ADMmature 8.8 times. In patients with an altered cerebral circulation after subarachnoid haemorrhage only a four-times increase in ADM plasma concentrations was measured (33). Furthermore, the infusion of a higher dose in the pilot study induced a substantial effect on BP and HR and was deemed unsuitable. We therefore feel confident that the administered dose of ADM was sufficient to induce migraine if the peptide was indeed a mediator of migraine.
Our human model of experimental headache has demonstrated its ability to describe the headache/migraine-inducing potential of several substances, e.g. NO, CGRP and histamine (2, 34, 35). In healthy individuals this headache is monophasic, occurring during and shortly after the infusion. The experimentally induced headache in migraine patients is characterized by a biphasic course with an immediate headache, often similar to the headache experienced by non-migraineurs, and a delayed headache occurring between 1 and 12 h later. The delayed headache fulfilled criteria for MoA in three out of nine patients after CGRP administration (2), in five out of 12 after histamine (36) and in approximately 80% after GTN. In previous studies both intracranial and extracranial dilation were seen, and in spontaneous migraine attacks both the MCA and STA were dilated (37, 38). It remains uncertain which of these territories is most important for migraine induction (39). ADM is the first substance shown to dilate STA without affecting MCA or CBF. Our results indicate that dilation of extracranial vessels is not enough of itself to induce migraine.
In conclusion, the infusion of ADM failed to induce headache or migraine in migraine patients, compared to placebo. It is therefore unlikely that ADM plays a pivotal role in migraine pathogenesis. The vasodilatory properties of ADM were confirmed for the STA and extracranial arterioles (flushing), but neither the MCA diameter nor CBF changed.
Footnotes
Acknowledgements
The technical equipment used was partly sponsored by The Villum Kann Rasmussen Foundation and The Toyota Foundation. The Lundbeck Foundation funds the research of the Danish Headache Centre. Special thanks to Lene Elkjær and Kirsten Bruunsgaard for excellent technical support.
