Abstract
In the UK, 4± of general practitioner consultations are for headache, yet the natural history of these presentations is unknown. The objective of this study was to describe the outcome of new headache presentations to the general practitioner. This was a prospective case-control study in adults over a period of 1 year using data from the General Practitioner Research Database, UK. Records of patients who presented with primary headache (migraine, tension-type headache, cluster headache) or undifferentiated headache (no further descriptor) were examined for the subsequent year for subarachnoid haemorrhage, primary brain tumour, benign space-occupying lesion, temporal arteritis, stroke and transient ischaemic attack. We identified 21 758 primary headaches and 63 921 undifferentiated headaches. The likelihood ratio was 29 (9.9, 92) for a subarachnoid haemorrhage after an undifferentiated headache and increased with age. The 1-year risk of a malignant brain tumour with new undifferentiated headache was 0.15±, rising to 0.28± above the age of 50 years. For primary headache the risk was 0.045±. The risk for a benign space-occupying lesion was 0.05± for an undifferentiated and 0.009± for a primary headache. The risk of temporal arteritis was the highest of the conditions studied, 0.66± in the undifferentiated and 0.18± in the primary headache group. Accepting the limitations of this approach, our data can inform management guidelines for new presentations of headache in primary care and confirm the need for follow-up, even if a primary headache diagnosis is made.
Introduction
In the UK, in a 3-month period 70% of the adult population experience headache (1). Headaches account for around 4% of general practitioner (GP) consultations (2), but the natural history of headache presented to primary care is unknown. Often, patients and doctors are concerned that their headache may be a symptom of a serious underlying cause, in particular a brain tumour (3). This worry can result in imaging studies with doubtful appropriateness, driven by non-clinical factors, including medico-legal concerns (4).
Headache is classified as primary when the headache and associated symptoms are an independent disorder, and secondary when the headache is part of another pathobiological process. The important primary headaches in primary care are migraine, tension-type headache and cluster headache, with migraine the most prevalent (5).
Although many secondary headaches are accompanied by other clinical features that facilitate a diagnosis, isolated headache can cause diagnostic concern. Important causes of secondary headache that we consider are subarachnoid haemorrhage (SAH), primary malignant tumour, benign space-occupying lesions, temporal arteritis, stroke and transient ischaemic attack (TIA). Secondary brain tumour was not included, as in most cases the primary tumour will have been identified (6).
Subarachnoid haemorrhage
SAH is frequently missed (7) and is the most common cause of neurological litigation (8). It can present with a ‘sentinel’ or warning headache occurring in 10–43% of cases (9). The most common presentation is a ‘thunderclap headache’ that reaches a maximum intensity within seconds and lasts for a few hours. Twelve per cent of such patients have a SAH, rising to 25% if clinical examination is abnormal (10–12). However, the importance of sentinel headache where the pain is less severe is contested. It has been suggested that the term is misleading and should be abandoned as it may represent a small bleed and should have been investigated (13).
Primary brain tumour and non-progressive space-occupying lesions
Headache is reported by 70% of patients with a primary brain tumour at some time in their illness (14). It is less common as a presenting complaint, with reports in retrospective studies ranging from 23 to 56% of patients with headache as their initial symptom (15–17). The proportion of patients with primary tumour who present with an isolated headache is between 2 and 16% (18, 19). We have reported the risk of primary tumour with a presentation of headache in primary care as 0.09% [95% confidence interval (CI) 0.08, 0.10] (20). Other non-progressive space-occupying lesions occur with similar frequency to primary tumours, but their contribution to headache is less certain (21).
Temporal arteritis
Headache is the presenting feature in 86% of patients with biopsy-proven temporal arteritis (22). It must always be considered in new headache presentation above the age of 50 years, where the annual incidence is approximately 1 per 1000 (23).
Stroke and TIA
Headache may accompany stroke, but is frequently unrecognized in clinical practice. Studies report headache in 60% of patients with intracerebral haemorrhage (24) and 17–34% of those with ischaemic stroke (25). Twenty-four per cent of patients with TIAs have related headache (26). Headache is also an independent predictor of both stroke and TIAs (27). In a large prospective study, men reporting chronic headache had a hazard ratio of 4.1 for stroke in the next year, although women did not have a significant extra risk (28). The incidence of stroke is increased in migraine (29), with a pooled relative risk reported in a recent systematic review of 2.12 (95% CI 1.8, 2.5) (30). A sentinel headache noted by the patient in days to weeks before stroke onset has been reported in 10% of strokes (31–33).
Although the vast majority of headaches are seen in primary care, the limited information on the outcomes of headache has been derived from retrospective secondary care studies. The aim of this study was to describe the outcome of new headaches in primary care. These were divided into new primary headache (migraine, tension-type headache and cluster headache) and headache where a diagnosis was not made, which we term ‘undifferentiated headache’. The study was approved by the Scientific and Ethical Advisory Group of the General Practice Research Database (GPRD).
Methods
This was a case–cohort study using data from the GPRD. Doctors contributing to the GPRD record full details of patient characteristics, including all consultations and diagnoses. Data are subject to thorough validation and stringent quality checks, and have been used in many epidemiological studies (http://www.gprd.com).
Cases
These were patients aged ≥ 18 years with a code for headache in their records, having had no other headache code in the previous year. A list of headache codes was compiled by the authors (available from them) and agreed with the GPRD. We included cases from the inception of the database in January 1987 to June 2005 with at least 1 year of full data in their records after the index headache consultation. These cases were identified by GPRD staff and the full anonymized electronic records were extracted. The index headache code was categorized into a primary headache (migraine, tension-type headache or cluster headache). Secondary headaches that had a further descriptor were discarded. All other codes were classified as undifferentiated headache.
Controls
Each patient was matched by age (to the same year of birth), sex and practice to a control with no record of headache during the year before the index date of their case, although they could have had a headache before or after that year. Controls also had to have a minimum of 1 year of full data after the index date.
Data collection and analysis
Libraries of codes relating to the outcomes of interest were assembled (available from authors). The records of all study patients with primary and undifferentiated headache were examined for the subsequent year to identify such codes. Undifferentiated headache records were also examined for a subsequent diagnosis of a primary headache.
Differences between cases and controls were analysed with conditional logistic regression, to account for the matching. An initial estimate of cases in the GPRD was 80 000; to allow for possible incomplete data, a power calculation was performed using 50 000 cases and controls. With estimates of an annual brain tumour incidence of 1:1200 in the headache population and 1:12 000 in the non-headache population, a study of this size would have had 99% power using a two-sided α of 5%.
Results
There were 85 679 patients with a primary or undifferentiated headache satisfying the inclusion and exclusion criteria. Of these, 21 758 (25.4%) had a primary headache code at their index consultation with migraine accounting for 15 891 (73.0%), tension-type headache 4987 (23.0%) and cluster headache 880 (4.0%). Undifferentiated headache, i.e. coded for headache but with no other descriptor, accounted for 63 921 (74.6%). Of the 85 679 controls, 17 087 (19.9%) had also reported headache previously, but not during the year before the index headache date of their case.
Of those with primary headache, 15 963 (73.4%) were female, a significantly higher proportion than in those with undifferentiated headache, where 40 721 (63.7%) were female (P < 0.001, χ2 test). Patients with primary headache were slightly younger than those with undifferentiated headache; median age 38 years (interquartile range 29–50) compared with 41 years (30–58; P < 0.001, Mann–Whitney test).
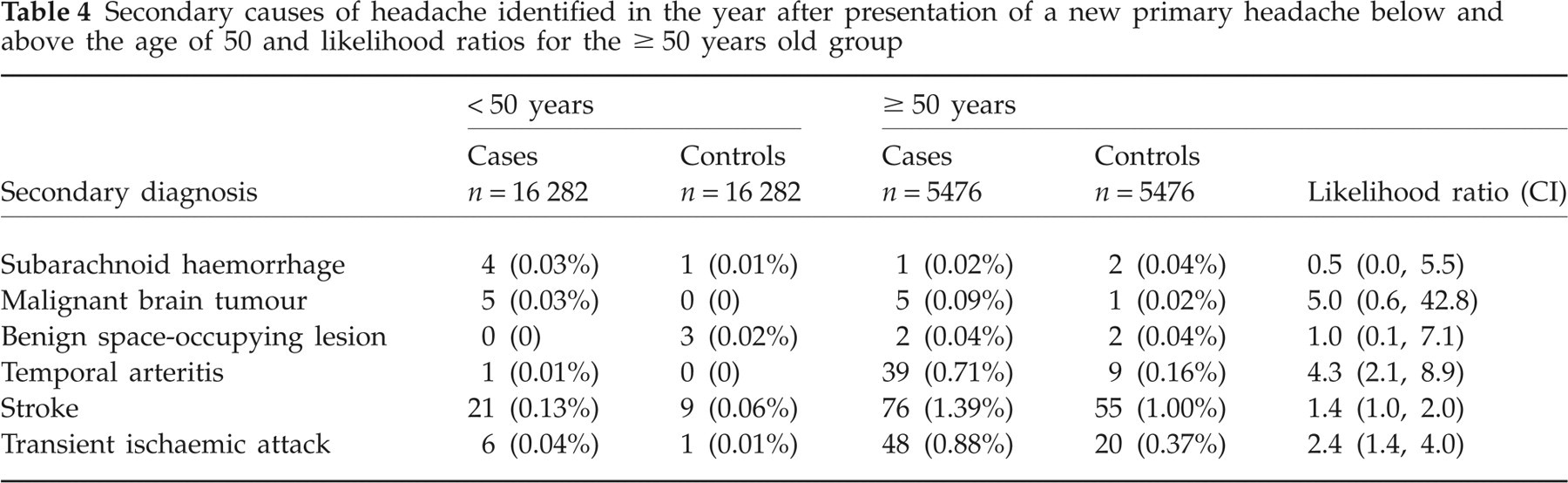
Figure 1 shows the main findings. Table 1 shows the outcomes of undifferentiated headaches, in terms of subsequent diagnosis and the time to diagnosis. Of this undifferentiated group, 5.3% received a subsequent diagnosis of a primary headache. Of the 108 patients with SAH preceded by undifferentiated headache, 52 (48%) had their diagnosis coded within the first 7 days following presentation. Table 2 shows the same outcomes for patients diagnosed with a primary headache. Tables 3 and 4 show undifferentiated and primary headache groups divided into ≥ 50 years and < 50 years old.

Risk in subsequent year of secondary pathology following a new headache presentation in primary care. (Risk for control group without headache.)
Secondary causes of headache identified in the year after presentation of a new undifferentiated headache to primary care and time to diagnosis
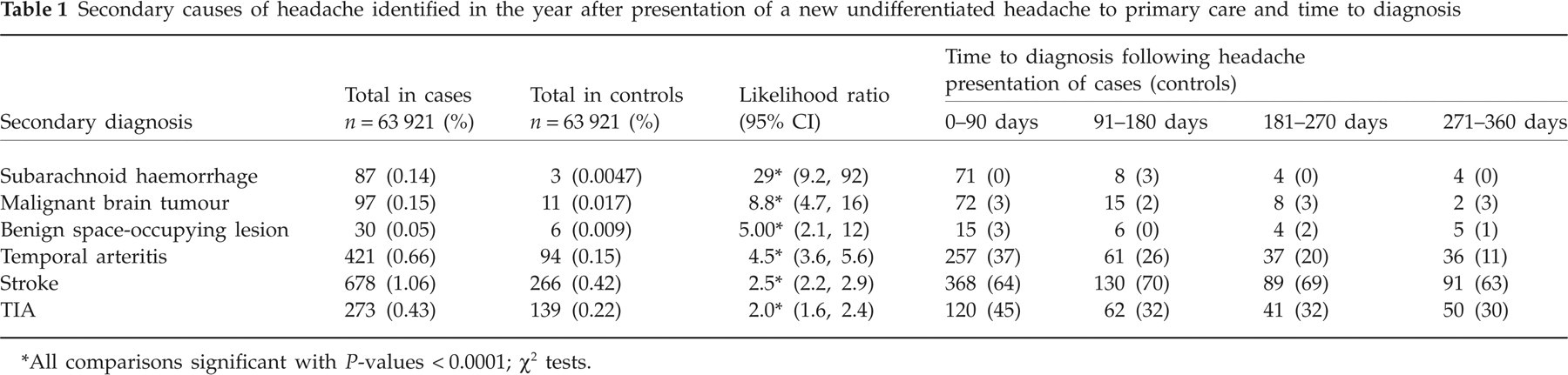
All comparisons significant with P-values < 0.0001; χ2 tests.
Secondary causes of headache identified in the year after presentation of a primary headache and time to diagnosis

Secondary causes of headache identified in the year after presentation of a new undifferentiated headache below and above the age of 50 years and likelihood ratios for the ≥ 50 years old group

Secondary causes of headache identified in the year after presentation of a new primary headache below and above the age of 50 and likelihood ratios for the ≥ 50 years old group
Discussion
This study has estimated the incidence of the main types of secondary headaches in the year after a new headache presentation to primary care. For undifferentiated headache, all the major diagnoses were more common in cases than controls, and SAH had the highest likelihood ratio, with 5.3% of this group receiving a subsequent diagnosis of primary headache. In patients with an initial diagnosis of primary headache, all outcomes were more common than in controls except for benign space-occupying lesions, whereas all outcomes were less common than in the undifferentiated headache group.
From a practical perspective, if the GP can make a diagnosis of a primary headache disorder and, in particular, migraine, the likelihood of subsequent pathology is very small. Clear evidence in the medical record of the basis for a primary headache diagnosis should reduce medico-legal concerns and missing serious pathology.
An important strength is that data were collected prospectively in primary care—the setting where the majority of headache patients are seen. Previous studies have been undertaken in secondary care settings and are mainly retrospective. The size of the study enabled us to identify small, but significant, increases in the diagnoses of interest and we were also able to study control patients to allow adjustment for the background incidence of major diagnoses. We made a pragmatic decision to allow controls to have had a headache, albeit not in the year before the period of study. In general, controls would be free from the disease being studied, though they should be eligible to become cases later (34). With such a high frequency of headache complaints in the whole GPRD population, the alternative of requiring all controls to be free of a history of headache could have rendered the control population artificially healthy. Clinically, having had no headache for a year seems a sensible place from which to consider investigative guidelines.
Our study has a number of limitations. First, practitioners may misdiagnose the presenting headache. In particular, 19% of our population received a diagnosis of migraine compared with studies that suggest that 84% of headache presentations in primary care are migraine (35). We were only able to use the headache label as recorded by the GP and not formal diagnostic criteria. However, the percentage of patients labelled by the GP with a primary headache who subsequently received a secondary headache diagnosis was smaller than those with an undifferentiated label, suggesting that the GP's diagnosis is still identifying a different population. Second, as we could examine only codes and not free text, we were unable to identify whether undifferentiated headache was isolated or associated with other clinical signs or symptoms. Third, when considering the time to diagnosis, there may be a potential ‘coding delay artefact’. For example, a SAH may present and be coded as ‘headache’. Although the diagnosis may have been clear at presentation, it would not have been accurately coded until confirmed by hospital studies and, as such, would not qualify as a sentinel headache.
Subarachnoid haemorrhage
The likelihood ratio of 29 (CI 9.9, 92) for this condition after an undifferentiated headache is high, and the risk is three times higher above the age of 50 years. The majority presented in the first 3 months from presentation. However, 48% of cases were coded within the first week, which may be artefactual due to coding delays as outlined above. Nevertheless, more cases than controls had this diagnosis for every time period up to a year, albeit with small numbers. This suggests that sentinel headaches are a feature of SAH, although rare.
Primary tumour
We found that the risk of a malignant brain tumour with new undifferentiated headache was 0.15%, rising from 0.08 to 0.28% below and above the age of 50 years. This compares with our previous case–control study of brain tumours in primary care that estimated a risk of 0.09% of a primary brain tumour in the 6 months after a report of headache to general practice (5). That study examined all headaches, whereas this one was restricted to new headaches, which may explain the difference. Tumours were diagnosed within 3 months in 72 (74%) patients and in 87 (90%) patients by 6 months. Again, a diagnosis of primary headache was associated with a lower risk of a tumour.
Space-occupying lesions
Although benign space-occupying lesions occur with similar frequency to primary tumours, the risk with a headache presentation was lower than for malignant brain tumours, 0.05% for undifferentiated with the risk doubled over 50 years of age and 0.009% for a primary headache. These lesions present over a longer time span than tumours, in that only 50% had been diagnosed at 3 months compared with 74% of tumours.
Temporal arteritis
The risk of temporal arteritis was the highest of the conditions studied, 0.66% in the undifferentiated and 0.18% in the primary headache group. The likelihood ratio in both groups was similar, 4.5 and 4.4, so being given a label of a primary headache was little protection. Over a third of the cases had temporal arteritis diagnosed ≥ 3 months from presentation, a delay that may have important clinical consequences.
Stroke and transient ischaemic attack
We were unable to distinguish between haemorrhagic and thrombotic stroke. Our results suggest that stroke and TIA patients are more likely to have presented with previous headache than controls. In these conditions there should be no coding delay artefact, as the GP is unlikely to record the headache at time of diagnosis, yet omit to record the stroke or TIA. For strokes 368 (54%) and TIAs 120 (44%) occurred within 3 months of the onset of the headache. For undifferentiated headache, the risk was doubled in the subsequent year. We postulate three reasons for this observation. First, a number of these undifferentiated headaches may have been migraine, where there is a recognized association. Second, these patients may be having headache from drugs taken for underlying vascular risk factors. Third, headache may be accompanying silent stroke. A recent population MRI survey found that 7.2% of people > 45 years old had asymptomatic brain infarcts (36). There was a similar increase in risk with a primary headache diagnosis, which may reflect the recognized association between migraine and stroke. The likelihood ratios we describe are in line with previous studies. The mechanism of this relationship is unclear. In this study, we may be describing undiagnosed migraine, where a relationship with stroke has been identified, although the causality remains speculative.
In conclusion, we have quantified the risk of a number of important headache disorders in primary care. The risk of temporal arteritis was the highest of the conditions studied. Primary care practitioners can be reassured about the low risk of primary brain tumour in presentations of undifferentiated headache, although there is a significant increase at > 50 years old. We have confirmed that although rare, sentinel headaches are a feature of SAH. The relationship between prior headache and stroke is an interesting finding and needs further clarification.
Although there are a number of limitations to our study, in particular the lack of accompanying clinical data that can help to confirm a diagnosis, the data we present are the most comprehensive available for the practitioner. They can not only inform management guidelines for new presentations of headache and, in particular, reassure practitioners that the chances of a brain tumour presenting with headache are low, but also alert the practitioner to the need for follow-up, even if a primary headache diagnosis is made.
Competing interests
D.K. chairs the British Association for the Study of Headache. This body receives educational support from individual pharmaceutical companies. No company had any input into this study.
Acknowledgements
The authors thank members of the council of the British Association for the Study of Headache for their comments on this study. D.K. and W.H. receive research practice funding from the Department of Health (St Thomas, Exeter, and Barnfield Hill, Exeter). Access to the GPRD was with an MRC licence.
