Abstract
Neuroimaging studies in cluster headache (CH) patients have increased understanding of attack-associated events and provided clues to the pathophysiology of the condition. They have also suggested stimulation of the ipsilateral posterior inferior hypothalamus as a treatment for chronic intractable CH. After 8 years of experience, stimulation has proved successful in controlling the pain attacks in almost 60% of chronic CH patients implanted at various centres. Although hypothalamic implant is not without risks, it has generally been performed safely. Implantation affords an opportunity to perform microrecordings of individual posterior hypothalamic neurons. These studies are at an early stage, but suggest the possibility of identifying precisely the target site by its electrophysiological characteristics. Autonomic studies of patients undergoing posterior hypothalamic stimulation provide further evidence that long-term stimulation is safe, revealing that it can cause altered modulation of the mechanisms of orthostatic adaptation without affecting the baroreflex, cardiorespiratory interactions or efferent sympathetic and vagal functions. Chronically stimulated patients have an increased threshold for cold pain at the site of the first trigeminal branch ipsilateral to the stimulated side; when the stimulator is switched off, changes in sensory and pain thresholds do not occur immediately, suggesting that long-term stimulation is required to induce sensory and nociceptive changes. Posterior inferior hypothalamic stimulation is now established as a treatment for many chronic CH patients. The technique is shedding further light on the pathophysiology of the disease, and is also providing clues to functioning of the hypothalamus itself.
INTRODUCTION
Trigeminal autonomic cephalalgias (TACs) are a group of primary headaches characterized by disabling, short-lasting pain attacks associated with autonomic phenomena mainly affecting the eye and nose (1, 2). They comprise cluster headache (CH), paroxysmal hemicrania and short-lasting unilateral neuralgiform headache attacks with conjunctival injection and tearing (SUNCT) (1).
In CH, the most common TAC, the pain occurs as severe paroxysmal strictly unilateral attacks, with maximum pain typically in the retro-orbital area (1); the autonomic symptoms are ipsilateral to and contemporaneous with the pain, and mainly consist of ptosis, miosis, lacrimation, conjunctival injection, rhinorrhoea and nasal congestion (1). These autonomic manifestations are generally a sign of parasympathetic hyperactivity and sympathetic impairment (2, 3).
CH is relatively uncommon, with a prevalence of < 1%; it mainly affects men, with a male:female ratio of between 2.5 and 7.1 to 1 (4, 5). The cluster headache attacks last 15–180 min and often occur daily (1). In the episodic form, attacks occur during a cluster period typically lasting 6–12 weeks, followed by remission (3). Remissions can last for up to 12 months or even longer. In the chronic form, attacks continue without significant remission. About 10–20% of patients with chronic CH suffer daily attacks that are severely debilitating, despite optimum medical therapy (3).
RATIONALE FOR HYPOTHALAMIC STIMULATION IN CH AND OTHER TACs
A number of clinical observations lend support to a central nervous system origin of cluster CH: the attacks often occur at fixed times of the day or night (circadian recurrence); the cluster periods typically recur during spring or autumn (circannual recurrence) (3); lithium, which accumulates in the brain, is an effective CH prophylactic (6); and alterations in the circadian release of hormones are amply documented in the condition (7).
Over the last decade, neuroimaging studies during ongoing CH and other TAC attacks have greatly increased understanding of attack-associated events, and provided clues to the mechanisms underlying their pathophysiology.
Positron emission tomography (PET) studies during both spontaneous (8) and nitroglycerine-induced CH attacks (9) show activation of the posterior inferior hypothalamus on the pain side. A voxel-based morphometry study has revealed a structural anomaly (increased neuronal density) in this area in remission-phase CH (10), and proton magnetic resonance spectroscopy has revealed lowered N-acetylaspartate and creatine levels, compared with controls, both during and outside CH pain periods (11, 12). The structural anomaly was particularly significant, as it was the first identification of a putative lesion site responsible for a primary headache. Subsequently, functional magnetic resonance imaging (MRI) and PET studies have shown that the posterior inferior hypothalamic grey matter was also activated during SUNCT (13) and paroxysmal hemicrania (14). The activation seems specific to TACs (15). Taken together, these findings suggest that the CH generator is located in the posterior inferior hypothalamus.
Stereotactic stimulation of specific brain areas is used to relieve drug-resistant movement disorders and epilepsy. We reasoned that stimulation of the ipsilateral posterior inferior hypothalamus in chronic intractable CH patients might prevent neuronal activation there and relieve pain. The first hypothalamic stimulation was performed in 2000 on a drug-refractory chronic CH patient; it proved successful (16, 17). Here we present long-term results of hypothalamic stimulation for chronic CH in patients treated at our own centre and at other centres.
PATIENTS WITH HYPOTHALAMIC STIMULATION
Information on 50 CH patients who had received hypothalamic implant was available either in the literature or via personal communication (see Table 1). Sixteen of these (14 men; mean age at implant 43 years; mean duration of chronic CH 3 years, range 1–10 years) were operated on at our centre (18). All our patients suffered multiple daily attacks (five to eight attacks/day) and were proposed for hypothalamic implant after at least 2 years of follow-up (18), during which all drugs, alone and in combination, were tried and found to produce no benefit. Prolonged use (years) of steroids in our patients produced a number of severe conditions including chronic intestinal bleeding, bone demineralization leading to fractures and aseptic necrosis of the femoral head, fluid retention leading to heart failure, hypertension, bulimia and marked weight increase, severe myopathy with loss of ability to walk, glaucoma, agitation, insomnia, and frank psychosis. Patients with alternating side CH attacks received bilateral implants on separate occasions (17, 18). All our patients had normal neurological examination, normal cerebral MRI (including the craniocervical transition), normal arterial and venous MRI angiography, and were psychologically stable (18). The two women were not pregnant. The operation and stimulation procedures were approved by the Ethics Committee of our institute. All patients gave written consent after being informed about the risks and benefits and told that the stimulator could be turned off without their knowledge after a pain-free period of at least 3 months (18).
Summary of results of hypothalamic stimulation in drug-resistant chronic cluster headache patients from various centres

Only 38 of the 50 patients were considered in the improvement evaluation.
Improvement: pain free or almost pain free.
In press; with authors' and editor's permission.
Personal communication, with authors' permission.
Nationwide multicentre double-blind study (not included in improvement evaluation).
In the other 34 patients (Table 1), attack frequencies were similar to those reported in our studies, i.e. multiple daily attacks with no pain-free periods in the preceding years (19–25). However, it is possible that the patient subject of one study (25) had episodic CH and is therefore not included in evaluation that follows.
STEREOTACTIC METHODOLOGY
At our centre, stereotactic implantation is performed with a Leksell frame (Eleckta, Stockholm, Sweden) under local anaesthesia (16, 18, 26, 27). Preoperative antibiotics are administrated in all cases. Preoperative MRI (brain axial volumetric fast spin-echo inversion recovery and T2 images) are used to obtain high-definition images for the precise positional determination of the anterior and posterior commissures and midbrain structures below the commissural plane (mammillary bodies and red nucleus). The MR images are fused with 2 mm thick computed tomography (CT) slices obtained under stereotactic conditions using an automated technique based on a mutual-information algorithm (Frame-link 4.0; Sofamor Danek Steathstation, Medtronic, Minneapolis, MN, USA). From these, the workstation provides stereotactic coordinates of the posterior hypothalamus ipsilateral to the pain side: 3 mm behind the midcommissural point, 5 mm below this point, and 2 mm lateral from the midline (Fig. 1). Target planning based exclusively on the mid-commissural point caused electrode misplacement in one patient (27) attributable to individual variation of the angle between the brainstem and the commissural plane. To correct this, we introduced a third anatomical landmark to define the implantation coordinates. This is the apex of the interpeduncular cistern, at the level of the maximum diameter of the mammillary bodies. This point has been called ‘interpeduncular point’ and lies on the axial brain slice 6–9 mm below the commissural plane (the average value is 8 mm in our series) (Fig. 2). The result is that, when three landmarks are used, the anteroposterior coordinate (Y coordinate) of the target is corrected to a position about 1–2 mm posterior to the interpeduncular point, instead of 3 mm posterior to the midcommissural point as previously. A program and atlas have been developed to determine coordinates and are freely available (http://www.angelofranzini.com/BRAIN.htm, password 5588).

Top: virtual ventriculograms, anterior-posterior view. Lower left: virtual ventriculograms, lateral view from the right side. The stereotactic trajectory and the two available active contacts for unipolar or bipolar stimulation are shown as red circles along the trajectory itself. Yellow: thalamic nuclei; pale blue: subthalamic complex; green: globus pallidus internus; pink: postero-medial hypothalamus. Lower right: magnetic resonance imaging showing the tips of two implanted electrodes positioned in the postero-medial hypothalamus (http://www.angelofranzini.com/BRAIN.htm, password 5588).
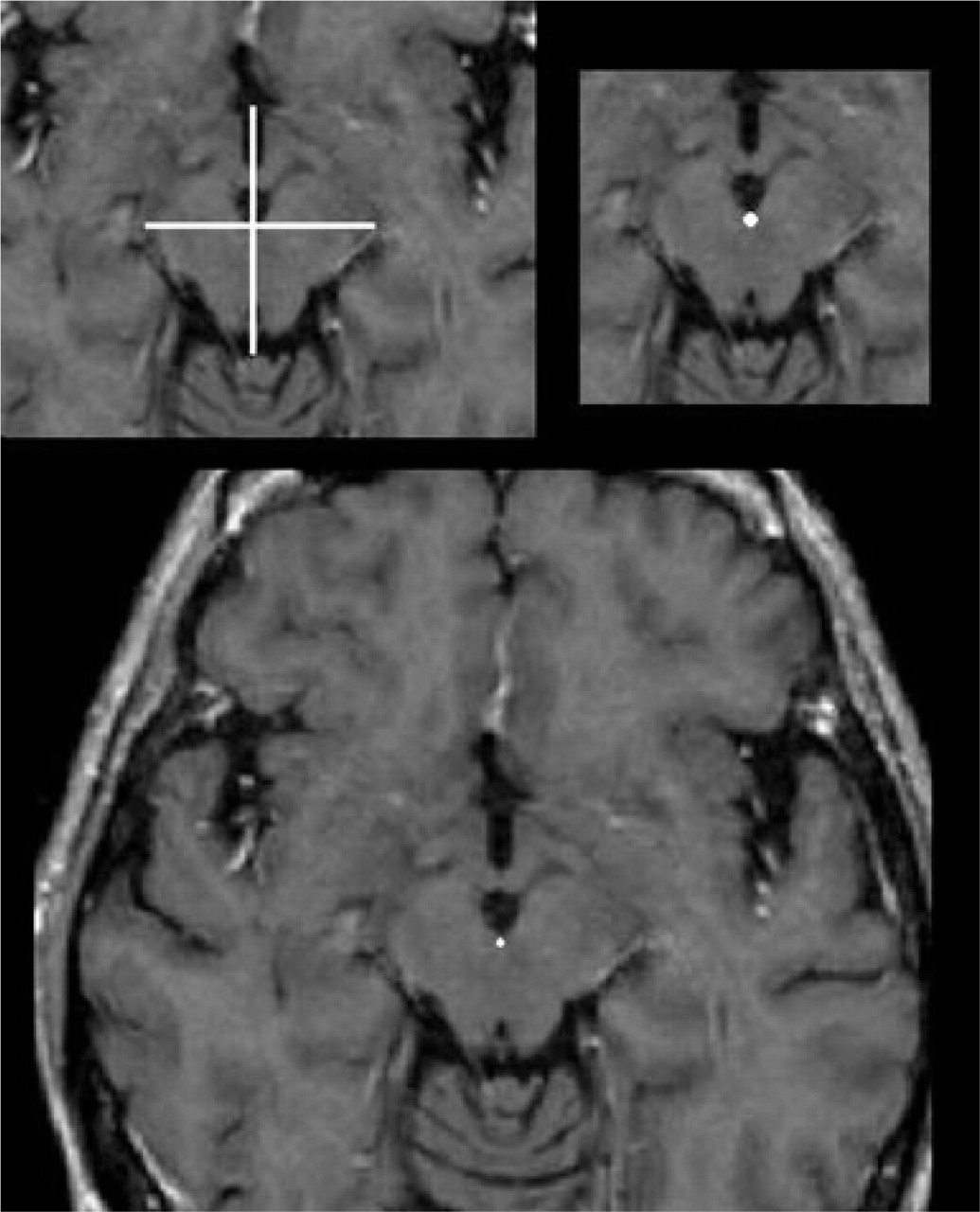
Magnetic resonance images illustrate the location of the new third stereotactic reference point, the ‘interpeduncular’ point (marked by the white cross in the top right picture). The interpeduncular point is located at the apex of the interpeduncular cistern at the level of the mammillary bodies (top left picture) in the brain axial slice 8 mm below the commissural plane (lower picture).
A rigid cannula is inserted through a paramedian coronal drill hole (3 mm) to within 10 mm of the target. The cannula is use for microrecording and for placement of the definitive electrode (Quad 3389; Medtronic) later connected to a subclavicular pulse generator (Kinetra Medtronic) via a subcutaneous lead (16, 18, 26, 27).
RESULTS: PAIN IMPROVEMENT
In our patients the mean follow-up was 4 years (Table 1) (18). After the first 2 years, major improvements in pain or pain abolition were obtained for 15 (83.3%) of the 18 implants (13 patients). After a mean of 4 years a persistent pain-free state was still present in 10 patients (62%). Four patients (25%) required prophylaxis to control the attacks (18). Over the last 2 years deep brain stimulation (DBS) has become ineffective in three patients notwithstanding many changes in stimulation settings. Interestingly, however, the illness changed from chronic to episodic in these three patients, with the appearance of months of complete remission punctuated by periods of typical attacks. Similar improvement has been reported by other authors (Table 1): an overall mean of 59% of patients have obtained improvement (reported as ‘pain free’ or ‘marked improvement’).
RESULTS: SIDE-EFFECTS AND EVENTS
A small, transient, non-symptomatic haemorrhage into the third ventricle was evident on postoperative CT in one of our patients (18). In no patients did EEG, blood pressure, temperature, breathing, affective state or state of consciousness undergo untoward modification during the operation (18, 20–25). In one study, one of the six patients died soon after the operation due to implantation-induced intracerebral haemorrhage, and procedure was stopped during the electrode implantation in another patient because of a panic attack (19). The latter patient may have had an anxiety disorder or, alternatively, the electrode could have missed the target; panic attacks have been reported during periaqueductal stimulation (28), but not during hypothalamic implantation/stimulation (16–18, 20–25).
Visual disturbance, mainly diplopia, is the main limiting side-effect induced by hypothalamic stimulation (16–24). Diplopia occurs in most patients when the amplitude is increased too rapidly and subsides within a few minutes, although a few days may be required for complete disappearance (16–24). No change in electrolyte balance, body temperature, blood pressure, sleep–waking cycle, appetite or thirst have been observed during prolonged stimulation (16–24). Levels of cortisol, prolactin, thyroid hormones, thyroid-stimulating hormone and testosterone have not been observed to change during stimulation. EEG also remains normal (16–24).
Weight loss in the first three to six postoperative months (mean 3.0 kg,
One patient had ceased menstruating 4 months before implantation as a result of excessive drug use; a month after implantation, her cycles returned to normal (18). Depressive or dysphoric symptoms, or changes in character or behaviour, have not been observed. All patients in one study have returned to work after implantation (18). Similar results have been reported in other studies (19–24). The results of a double-blind multicentre study have provided further evidence that hypothalamic stimulation is safe in CH (Lanteri-Minet, personal communication).
M ICRORECORDINGS OF INDIVIDUAL POSTERIOR HYPOTHALAMIC NEURONS IN CH: TOWARDS IDENTIFICATION OF A SPECIFIC NEURONAL DISCHARGE PATTERN
Use of DBS in CH has provided an opportunity to investigate neuronal activity in the target region (posterior hypothalamus) using microrecording techniques. Microrecording allows acquisition of signals from individual neurons (29) and may provide data to help understand the basic biological defect in CH and other TACs.
We shall discuss microrecordings obtained from the stereotactic target site corresponding to the hypothalamic area of increased blood flow shown by PET during CH attacks (9). This area does not correspond to a specific anatomical entity, and there is no consensus as to whether it is part of the posterior hypothalamus or the anterior periventricular grey matter (30–32). Two sets of stereotactic coordinates have been used to identify this target: those published by Leone et al. in 2001 (x = 2 mm lateral to the midline, y = 6 mm behind the midcommissural point, z = 8 below the commissural plane) (16); and the revised coordinates published by Franzini et al. in 2003 (x = 2 mm lateral, y = 3 mm posterior, and 5 mm inferior to the midcommissural point (26). Four studies (19, 21, 22, 26) have reported single unit microrecordings from this region in CH patients. All were obtained from CH patients not experiencing an attack (basal conditions), and no data are available from patients experiencing a CH headache.
Franzini et al. (26) have reported using a Medtronic Lead Point system to make microrecordings of the target area. Quantitative data were not presented, but only a figure illustrating single unit electrical activity at the target site. Schoenen et al. have reported microelectrode recordings in four patients with chronic CH (19), making use of the stereotactic coordinates of Leone et al. (16). A Medtronic 9013-SD-08411 microelectrode and a Medtronic Lead Point were used (19) to obtain recordings starting 10 mm above the target and continuing at 1-mm intervals down to the target. No additional details of the procedure and no quantitative data are given. The authors noted that specific neuronal activity similar to that found in DBS of the subthalamic nucleus or globus pallidus was not identified; they also mentioned that in two patients bursts of action potentials synchronous with heart beats were recorded (19).
A subsequent study has reported quantitative microrecording data in two patients with CH and one with SUNCT (33). Target coordinates were those suggested by Franzini et al. (26). Patients were studied fully awake using the Medtronic Lead Point system. Recordings began as the microelectrode reached the hypothalamic target. This procedure allowed the collection of signals from a single cell in each patient long enough to permit postoperative off-line analysis with the Spike2 analysis package (CED, Cambridge, UK). The average firing rate was 24 spikes/s. Most recordings showed isolated action potentials, with most interstimulus intervals in the 10–15 ms range. Autocorrelograms of two cells showed no periodicity in discharge rate, but in the SUNCT patient showed a 1-Hz oscillatory pattern possibly due to a pulse artefact (33).
Starr et al. (21) implanted a hypothalamic electrode in four CH patients using the coordinates of Franzini et al. (26). Single unit discharges were recorded with glass-coated platinum/iridium microelectrodes (Medtronik–FHC, Inc.) that were advanced into the brain in a single penetration. Signals were collected using the Guideline System 3000 (FHC, Bowdoinham, MN, USA). Stable recordings amenable to formal analysis were infrequent due to significant pulsation artefact. Nevertheless, seven units recorded for at least 10 s within 5 mm of the anatomical target were amenable to analysis. The mean (±
Finally, Bartsch et al. (22) have reported on neuronal activity in four CH patients who received posterior hypothalamic implant to relieve drug-resistant CH. Franzini et al's coordinates were used (26). Microrecordings were performed at ±1 mm around the target. In three patients sensory stimulation of the trigeminal and spinal dermatomes was performed. In two patients, motor stimulation (passive movement of elbow), autonomic testing (counting backwards under stress, cold pack and bladder filling) and affective stimulation (pictures with emotional content) were also performed. Microrecordings showed a tonic non-rhythmic firing pattern of average discharge rate 17 Hz (range 13–35). None of the applied stimuli produced an obvious neuronal response.
Microrecording data are obtained using high-impedance microelectrodes, making it possible to pick discharges from a single neuron. The acquisition of valid data requires expertise, specific equipment and careful attention to recording conditions; drugs and level of vigilance can affect the pattern obtained. Activity should be recorded in a large number of cells for least 10 s each to detect any oscillatory activity. The available microrecording studies in CH hardly meet these criteria, but allow some preliminary conclusions. First, neuronal activity is expected when targeting the posterior hypothalamus in CH (Fig. 3). Lack of neuronal activity and abnormal impedance suggest that the exploring microelectrode may be in the interpeduncular cistern (21). Second, pulsation artefact is frequent (Fig. 3). Third, a discharge rate around 20 Hz and lack of a specific rhythmic pattern are the most consistent characteristics of neuronal discharge in this area. These considerations apply to neuronal activity recorded in the area defined by the coordinates of Leone et al. (16) and Franzini et al. (26) in patients with CH and probably other TACs (33). Neuronal activity of this region may differ in other conditions and perhaps during a CH attack. Further data are required.

Microrecording samples at estimated target in posterior hypothalamus in patient with cluster headache. Bottom trace (b) shows pulsation artefact not present in top trace (a) (33).
A UTONOMIC CONTROL OF THE CARDIOVASCULAR SYSTEM DURING CONTINUOUS STIMULATION OF THE POSTERIOR HYPOTHALAMUS
The human hypothalamus consists of 4 cm3 of neural tissue, making up 0.3% of the volume of the entire brain (34). Nevertheless, it contains integrator systems that, via the autonomic and endocrine effector systems, regulate fluid and electrolyte balance, food ingestion and energy balance, various reproductive functions, thermoregulation, immune responses and many emotional responses, including pain. Many of the hypothalamic nuclei appear to have changed little in anatomy and function compared with their counterparts in lower animals (34, 35). The hypothalamus influences the parasympathetic and sympathetic autonomic systems. In general, parasympathetic effects predominate when the anterior hypothalamus is stimulated; sympathetic effects depend more on posterior hypothalamic stimulation (34). However, this does not imply discrete parasympathetic and sympathetic ‘centres’. Stimulation to disparate parts of the hypothalamus can cause profound changes in heart rate, cardiac output, vasomotor tone, peripheral resistance, differential blood flow in organs and limbs, frequency and depth of respiration, alimentary tract motility and secretion, erection and ejaculation (34). Lesions of the ventromedial nucleus cause increased vagal and decreased sympathetic tone; and the paraventricular nucleus is an important integrative centre (36). Remarkably, cortical limbic and visceral information is relayed to the arousal system and to the sleep and circadian systems of the hypothalamus, providing a basis for the behavioural adaptation and survival functions of the hypothalamus (34). Many of these activities and functions are modulated by discrete hypothalamic areas.
The advent of hypothalamic stimulation to relieve chronic CH has meant that, for the first time, the human hypothalamus has received long-term stimulation (16), providing a new window on to this extraordinary part of the brain.
Our group extensively investigated aspects of autonomic nervous system function in eight patients with drug-resistant chronic CH, 19 ± 22 days before surgery and 87 ± 52 days after surgery to implant an electrode for unipolar stimulation of the posterior hypothalamus (37). The patients were studied in a temperature-controlled clinical investigation room. Systolic and diastolic blood pressure, cardiac output, total peripheral resistance, heart rate and oronasal and abdominal breathing were monitored continuously at supine rest, during head-up tilt test (HUTT), Valsalva manoeuvre, deep breathing, cold face test and isometric handgrip. The results of cardiovascular reflex testing and autoregressive power spectral analysis of heart rate variability during supine rest and HUTT obtained before and after surgery were compared. It was found that continuous long-term unipolar stimulation of the posterior hypothalamus in CH patients was associated with an enhanced sympatho-excitatory effect on the cardiovascular system during HUTT, whereas the Valsalva manoeuvre, deep breathing, cold face test, isometric handgrip and baroreflex sensitivity were not affected (Fig. 4). These findings suggest that chronic stimulation of the posterior hypothalamus can cause altered modulation of the mechanisms of orthostatic adaptation without affecting the baroreflex, cardiorespiratory interactions or efferent sympathetic and vagal functions (37). Thus, in the supine resting position, the cardiovascular system is not influenced by stimulation of the posterior hypothalamus, providing important evidence that long-term stimulation in safe.
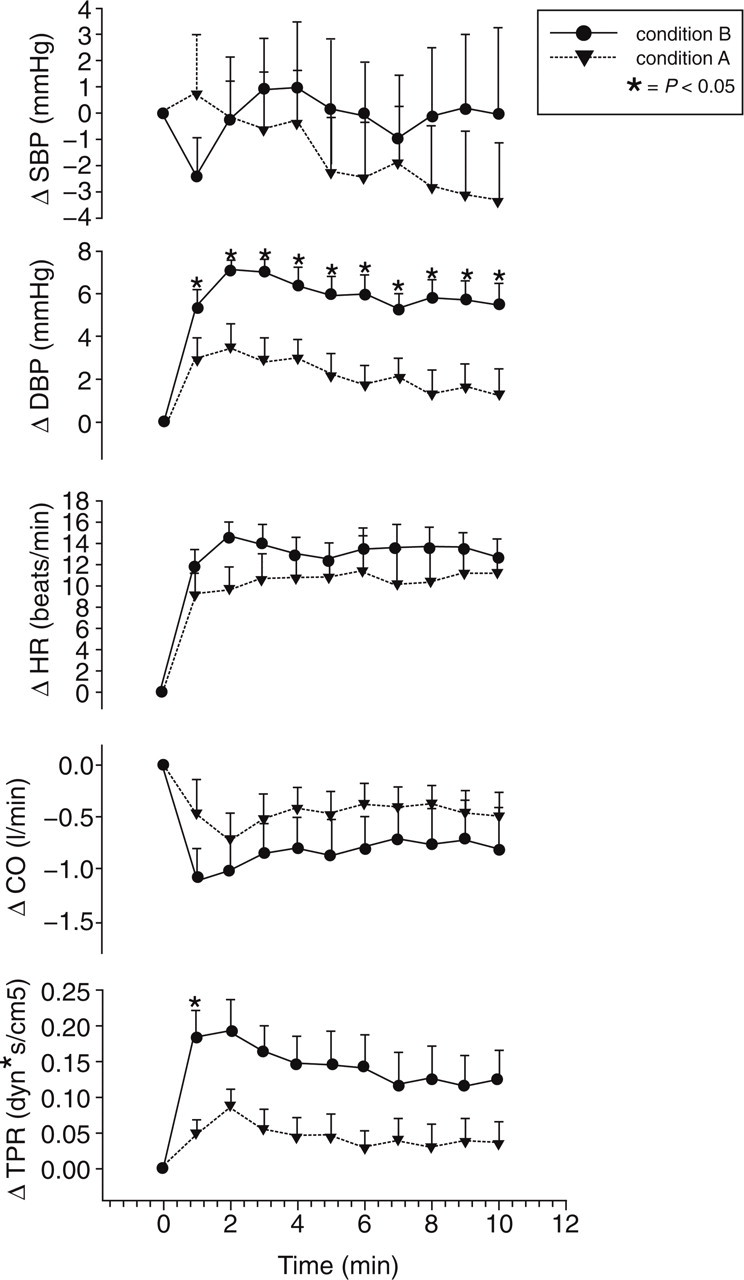
Cardiovascular responses to head-up tilt test (HUTT) in eight chronic cluster headache patients preoperatively (condition A) and during chronic stimulation of the posterior hypothalamus (condition B). Systolic blood pressure (SBP) changes were similar in both conditions. Diastolic blood pressure (DBP) was significantly higher in condition B compared with condition A, from the first minute and throughout the HUTT. Cardiac output (CO) somewhat lower in B (not significant). A prompt increase in total peripheral resistance (TPR) (P < 0.05 at first minute) is also evident. Heart rate (HR) was greater in condition B than in condition A, but the difference was not significant. Δ is the change with respect to the basal value set at 0. ∗P < 0.05 compared with condition A. Error bars =
Nevertheless, the increased sympathetic drive during HUTT sheds light on the role of the posterior hypothalamus in cardiovascular regulation in humans.
Posterior hypothalamic stimulation in animals produces marked changes in autonomic functions (38). In humans, continuous long-term posterior hypothalamic stimulation induces much fewer marked changes than in animals; this could be due to differences in stimulation pattern: low amplitude, slowly increasing amplitude, long-lasting stimulation in humans (18–25).
EFFECTS OF CHRONIC POSTERIOR HYPOTHALAMIC STIMULATION IN CH PATIENTS ON SLEEP AND THE CIRCADIAN RHYTHM OF BODY CORE TEMPERATURE
The hypothalamus, especially its posterior part, is involved in the control of the sleep–wake cycle and the regulation of arousal (39). The structure and quality of sleep and body core temperature (Bc T°) were investigated in three drug-resistant chronic CH patients before and during long-lasting stimulation of the posterior hypothalamus (40). The patients underwent 48-h consecutive polysomnography (PSG) using the Vitaport system (for details see 40) with Bc T° monitored by rectal probe. In these patients nocturnal CH attacks disappeared during hypothalamic stimulation, although occasional daytime attacks occurred in two patients 4 months after implantation. PSG during stimulation revealed more continuous sleep with increased total sleep time, sleep efficiency and quantity of slow-wave sleep stages, whereas indices of fragmented sleep [arousals, periodic limb movements during sleep (PLMS) and stage shift indices] were reduced. Thus, posterior hypothalamic stimulation was associated with improved sleep architecture and sleep quality compared with the preoperative condition (40). However, the study was too restricted in scope to allow inference of a direct effect of stimulation on sleep, and it is probable that disappearance of the nocturnal CH attacks was the main cause of the improvement. Nevertheless, the reduction in the number of PLMS during stimulation suggested a direct effect of stimulation on these abnormal nocturnal movements.
Since PLMS were reduced in light as well as in non-rapid eye movement sleep, the reduction could not be attributed simply to decreased length of time spent in light sleep. Remarkably, posterior hypothalamic stimulation was not associated with daytime sleepiness or sleep-related breathing disorders, and Bc T° rhythm was also normal—further indications of the safety of hypothalamic stimulation (40).
NEUROPHYSIOLOGICAL STUDY OF THE NOCICEPTIVE SYSTEM
Although the efficacy of hypothalamic stimulation in chronic CH is established, the mechanism by which it blocks headache attacks remains mysterious. One possibility is that long-term hypothalamic stimulation modulates sensory perception and, in particular, increases the pain threshold. Studies prior to the development of DBS for CH indicate that the pain threshold is altered in CH patients. Cephalic warm detection threshold was significantly increased on both sides in 22 CH patients compared with healthy subjects; however, pain thresholds were not examined (41). Increased warm and cold detection thresholds on the pain side have been reported in another study on chronic (n = 8) and episodic (n = 17) CH patients (42). In another study, bilaterally increased warm detection thresholds on the cheek, and bilaterally increased warm detection and heat pain thresholds on the hand, were found (43). Schoenen et al. (19) were the first to report on neurophysiological changes induced by hypothalamic stimulation. They studied only four patients, finding increased pressure and electrical pain thresholds extracephalically after a month, whereas cephalic pain thresholds were unchanged or were only slightly diminished at that time. They also found that, after a month, the response area of the nociceptive blink reflex was significantly increased ipsilateral to the stimulation electrode (19).
We studied 11 patients with chronic CH receiving unilateral stimulation of the posterior hypothalamus and compared them with 29 healthy volunteers and 15 chronic CH patients with ongoing attacks but no medication (44). Detection and pain thresholds for cold and warm stimuli were determined. We found that stimulated patients had increased threshold for cold pain ipsilateral to the stimulated side at the first trigeminal branch (44). This is consistent with the PET finding of May et al. (45) that the ipsilateral trigeminal nucleus is activated by hypothalamic stimulation. We found no significant changes in sensory or pain thresholds soon (60 min) after the stimulator was switched off (44), an indication that long-term hypothalamic stimulation is required to induce changes in the sensory and nociceptive system, and in line with the observations that weeks or months of stimulation are required to achieve significant pain control, and that a considerable time elapses before attacks reappear after the stimulator has been switched off (18, 46).
The anatomical basis for the trigeminal system–hypothalamic interaction has been described (47, 48). Non-nociceptive and nociceptive inputs are conveyed from facial and intracranial structures to the hypothalamus by neurons of the trigeminohypothalamic tract (47, 49). Nociceptive inputs (both dural and facial) to the trigeminal nucleus caudalis can be modulated by differential regulation of orexin A and B receptors in the posterior hypothalamus (50, 51). At least some of the clinical and neurophysiological effects of hypothalamic stimulation could be conveyed by this system, including the increased threshold for cold pain at the site of the ipsilateral first trigeminal branch in hypothalamic stimulated patients (44). This pathway could be the common final pathway through which other brain structures involved in pain modulation, and influenced by hypothalamic stimulation (45), modulate the trigeminal system (52).
CONCLUDING REMARKS
The efficacy of hypothalamic stimulation as a treatment for chronic drug-resistant CH is now well established (18–24). Although hypothalamic implant is not without risks (19), it has generally been performed safely (18, 20–25). Nevertheless, given the invasiveness of the procedure, we strongly recommend that it should be offered only to patients with chronic totally drug-resistant CH who suffer attacks several times a day. A diagnosis of chronic CH according to the criteria of the International Headache Society is not sufficient to indicate surgical procedures of this type, since the diagnosis can encompass low-frequency forms (three to four attacks per month) that may respond well to prophylactic medication. Even if the attacks are poorly controlled by medication, however, patients with low-frequency chronic CH should not, in our opinion, be proposed for hypothalamic implant. At our centre, hypothalamic implant is proposed only to patients with intractable, daily attacks several times a day, and then only if peripheral (greater occipital nerve) stimulation proves ineffective (53–55).
The safety of long-term hypothalamic stimulation has also been established, both by clinical results at several centres (18, 20–25) and by studies on autonomic changes (37, 40). Nevertheless, it is essential that stimulated patients be closely followed, since frequent adjustment of stimulation parameters is required to maintain optimal benefit (18–25).
Recent studies at various centres have made progress in elucidating the neurophysiological characteristics of the hypothalamic area chosen for stimulation, and in confirming its validity as a legitimate target (19, 21, 22, 33). More precise localization may lead to more reliable efficacy and reduced risks in patients undergoing hypothalamic implant. All studies concur in affirming that hypothalamic stimulation requires time to become effective; similarly, acute hypothalamic stimulation in ongoing attacks remains ineffective (56).
Neuroimaging studies have shown that hypothalamic stimulation activates several cerebral structures of the pain matrix and deactivates others (45). In particular, acute hypothalamic stimulation activates the ipsilateral trigeminal complex (45), which, however, does not cause the patient to perceive disturbances within the distribution of the nerve. This finding suggests that activation of the hypothalamus and of the trigeminal system are both necessary, but not sufficient, to generate CH attacks, and that other cerebral structures play a primary role in the pathophysiology of the disease (46).
