Abstract
Several trials have asserted that some anticonvulsant drugs seem to be useful for the prophylaxis of migraine, but systematic reviews are sparse. We independently searched PubMed, EMBASE and the Cochrane Central Register of Controlled Trials until 2005, as well as Headache and Cephalalgia through April 2006, for prospective, controlled trials of anticonvulsant drugs. Data were calculated and pooled across studies and expressed as standardized mean differences, odds ratios and numbers-needed-to-treat. Anticonvulsants, considered as a class, reduce migraine frequency by about 1.3 attacks per 28 days compared with placebo, and more than double the number of patients for whom migraine frequency is reduced by ≥50% relative to placebo. Sodium valproate/divalproex sodium and topiramate were better than placebo, whereas acetazolamide, clonazepam, lamotrigine and vigabatrin were not; gabapentin, in particular, needs further evaluation. Trials designed with sufficient power to compare different drugs are also necessary.
Introduction
Drug therapy for migraine falls into two categories: acute and preventive. Acute therapy aims at the symptomatic treatment of the head pain and other symptoms associated with an acute attack of migraine. The goal of preventive therapy is to reduce the frequency and/or intensity of attacks, and thereby improve patient functioning and quality of life. There is a fairly substantial body of evidence from controlled trials supporting the efficacy of many of the agents used for preventing migraine, yet such therapies are used by only a small percentage of patients with migraine (3–5% in various studies (1–3)). The underutilization of migraine preventative drugs is considered to be a major cause in sustaining the unmet needs of migraine sufferers worldwide (4).
The use of anticonvulsants for the prophylactic treatment of migraine is theoretically warranted by several known modes of action which relate either to the general modulation of pain systems (5) or more specifically to systems involved in the pathophysiology of migraine (6). It is necessary to point out, however, that it is not currently possible to state with any certainty which particular mode or modes of action of anticonvulsant drugs as a class are relevant to the prophylaxis of migraine.
Substantiated guidelines on the treatment of migraine have now been developed and published by the American Academy of Neurology (7). These guidelines suggest that prophylactic therapy should be considered for patients with frequent migraine attacks (more than two per month) or disabling migraine refractory to acute treatment, those who experience intolerable side-effects of acute therapy, those with contraindications to acute medication and those at risk of overusing acute medication. The guidelines also suggest that patient preferences and the cost of acute and preventative medications should be taken into consideration in making treatment decisions.
This review considers the evidence for the efficacy and tolerability of anticonvulsants for preventing migraine in adults. The prophylactic treatment of migraine in children is the subject of a separate Cochrane review (8).
Methods
Types of eligible studies
Studies were required to be prospective, controlled trials of self-administered drug treatments taken regularly to prevent the occurrence of migraine attacks and/or to reduce the intensity of those attacks. Trials were included only if allocation to treatment groups was randomized or pseudo-randomized and if results were published in full; short conference and journal abstracts were not included. Trials were not required to be blinded. Concurrent cohort comparisons and other non-experimental designs were excluded.
Study participants were required to be adults > 18 years of age. The use of a specific set of diagnostic criteria [e.g. Ad Hoc Committee (9), International Headache Society (IHS) 1988 (10) or 2004 (11)] was not required, but migraine diagnoses needed to be based on at least some of the distinctive features of migraine, e.g. nausea/vomiting, severe head pain, throbbing character, unilateral location, phono/photophobia, or aura. Secondary headache disorders had to be excluded on clinical grounds or after adequate diagnostic tests.
Studies including subjects with additional non-migrainous interval headaches were required to report frequency data using the term ‘migraine’ rather than ‘headache’, or else give a clear description on how migraine and non-migrainous interval headaches were differentiated and denoted. Studies evaluating treatments for chronic migraine, transformed migraine or chronic daily headache were regarded as unsuitable for this review, as these terms are not uniformly used and may be confounded by medication overuse.
Studies were required to have at least one arm with an anticonvulsant (without concomitant use of other migraine prophylactic treatment). Acceptable comparator groups included placebo, no intervention, other drug treatments, same drug treatments with a clinically relevant different dose and behavioural or physical therapies. The analysis included only drugs used as anticonvulsants in the treatment of epilepsy or status epilepticus in either Europe or the USA. Only dosages and formulations that are commercially available and suitable for out-patient use were considered. The list of target drugs included the following, in alphabetical order: acetazolamide, carbamazepine, chlormethiazole, clobazam, clonazepam, clorazepate, diazepam, divalproex sodium, ethosuximide, felbamate, fosphenytoin, gabapentin, lamotrigine, levetiracetam, lidocaine, lignocaine, lorazepam, mephobarbital, methsuximide, midazolam, nitrazepam, oxcarbazepine, paraldehyde, pentobarbital, phenobarbital, phenytoin, primidone, sodium valproate, tiagabine, topiramate, valproic acid, vigabatrin and zonisamide.
Types of outcome measures
For the efficacy analysis, data on headache frequency and headache index as obtained directly from patients were collected. Among headache frequency measures, we preferred number of migraine attacks to number of days with migraine as the former is recommended by the IHS and used in most drug trials. Headache index was not considered to be an appropriate outcome measure, because of a lack of uniform definition and potential confounding of frequency and severity.
As we anticipated that outcomes would be measured over various units of time (e.g. number of attacks per 4 weeks), and for numerous different time points (e.g. 4-week headache frequency at 2 months), we attempted to standardize the unit of time over which headache frequency was measured at 28 days (4 weeks) wherever possible. Outcomes were recorded beginning 4 weeks after the start of treatment and continued through all later assessment periods that were not compromised by problems with drop-outs (an overall drop-out rate of 20% or an imbalance in drop-out rates between treatment groups). A decision about which time points to include in the final analysis was made once the data had been collected.
In addition, we tabulated adverse events for each included study. Where data on adverse events were missing or inadequate, we intended to obtain these data by correspondence with authors, but in several cases this was not possible because of the age of studies.
Identification of studies
Search strategies combining the subject search with the Cochrane highly sensitive search strategy for randomized controlled trials (12) were used and adapted to the database searched. Databases that were searched were: (i) The Cochrane Pain, Palliative & Supportive Care Trials Register; (ii) The Cochrane Central Register of Controlled Trials (CENTRAL) (Issue 3, 2005); (iii) PubMed 1966 to December 2005; and (iv) EMBASE 1974 to December 2005. A detailed description of the subject search strategies has been published elsewhere (13). In addition, the reference lists of review articles, including papers, and text books related to headache were studied. Two journals, Headache and Cephalalgia, were hand searched in their entirety, until April 2006. We attempted to identify all relevant published trials, irrespective of language.
Titles and abstracts of studies identified by the literature search were screened for eligibility by two independent reviewers. Papers that could not be excluded with certainty on the basis of information contained in the title and/or abstract were retrieved in full for screening. Disagreements were resolved through discussion. Papers passing this initial screening process were retrieved, and the full text was reviewed independently by both reviewers. Disagreements at the full-text stage were resolved through discussion.
Data extraction and analysis
Information on patients, methods, interventions, outcomes and adverse events were abstracted by two independent reviewers. Crossover trials were analysed as if they were parallel-group trials, combining data from all treatment periods. If a carry-over effect was found and data were reported by period, then the analysis was restricted to period-one data only. In no trial were complete within-patient data reported, so within-patient improvement scores were not calculated.
Efficacy outcomes reported in dichotomous form (success/failure) were recorded or converted as the number of subjects in each arm with a ≥ 50% reduction in headache frequency (50% responder rate), and used to calculate odds ratios (ORs) with 95% confidence intervals (CIs) and numbers-needed-to-treat (NNTs) with 95% CIs (14). For outcomes reported on a continuous scale (change in migraine frequency), we recorded pre- and post-treatment group means and variance, and attempted to calculate or estimate variances based on primary data, test statistics, and/or error bars in graphs. These data were analysed using the standardized mean difference (SMD, with 95% CIs) rather than the weighted mean difference (WMD), as standardizing continuous outcome data on headache frequency to a 28-day (4-week) period was not possible in every case.
Reviewers recorded the proportion of patients reporting adverse events for each treatment arm wherever possible. The identity and rates of specific adverse events were also recorded.
The internal validity of individual trials was assessed through consensus using the scale devised by Jadad and colleagues (15), assigning a score of 0–5 points to each trial, with higher scores indicating higher quality in the conduct or reporting of the trial. The consensus score was not used as a weighting in statistical analyses.
Estimates of efficacy (both SMDs and ORs) were tested for homogeneity. If study estimates were homogeneous, they were combined using a fixed-effect model. When significant heterogeneity was present, an attempt was made to examine the differences based on the clinical characteristics of the included studies. Only when studies with statistically heterogeneous results appeared to be clinically similar were the study estimates combined using a random-effects model.
The main analysis was based on these statistics for the comparison of anticonvulsants as a class vs. placebo. Subgroup analyses were undertaken by drug against placebo and against active comparator, and by dose where possible. Data on the proportion of patients reporting adverse events were used to calculate numbers-needed-to-harm (NNH).
Results
Description of studies
The search strategy yielded 1089 studies as possible candidates in PubMed, 290 studies in EMBASE, and 6952 studies in CENTRAL, whereas no studies were retrieved from the Cochrane Pain, Palliative & Supportive Care Trials Register. Of these, 58 published papers were selected for full-text review. Thirty-five were excluded, for the following reasons: no appropriate control or comparison group in 11; seven were letters or abstracts; case studies only in four; review papers in three; data on chronic migraine or chronic daily headache only in three; no single anticonvulsant arm in two; inappropriate randomization in one; publication unobtainable in one; inadequate reporting in one; one was a basic science paper; and one concatenated data from two included trials.
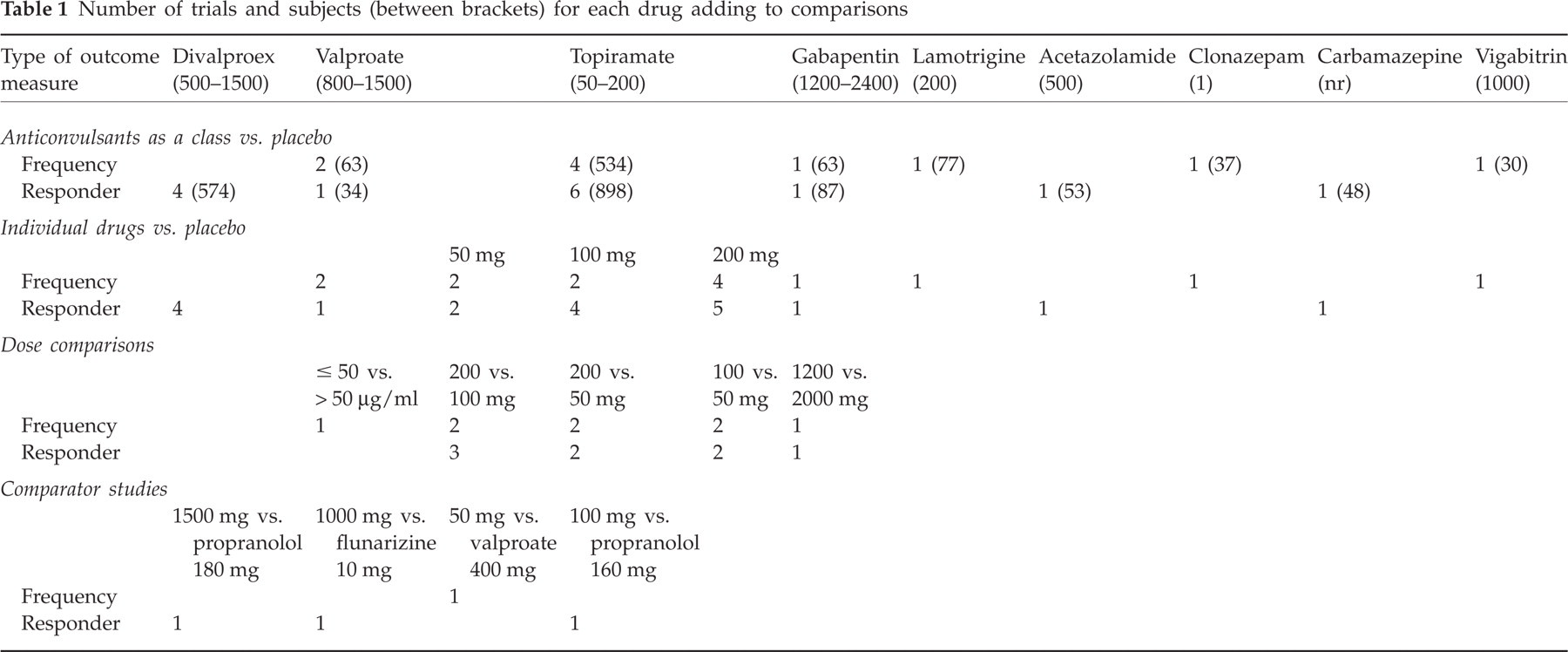
Twenty-three papers were therefore included in the review. The number of trials contributing to each of the analyses, the total number of subjects for each drug and the daily doses employed are depicted in Table 1. As three trials compared different doses of topiramate (i.e. 50 mg (16, 17), 100 mg (16–18) and 200 mg (16–18)) with placebo, the combined analyses contain data only for the 100-mg daily dose, which is considered the clinically optimal dose. The duration of the treatment phase of the included trials varied from 4 to 24 weeks, with a mean of 12.3 weeks.
Number of trials and subjects (between brackets) for each drug adding to comparisons
Methodological quality of included studies
Although the median quality score of 4 (range 1–5) was relatively high, there were a few studies with serious methodological weaknesses such as lack of blinding (19) or inadequate blinding (20).
Significant statistical heterogeneity was evident across trials for both efficacy outcomes. Although there was some methodological variation (e.g. diagnostic criteria, baseline migraine frequency, wash-out periods for previous medication, rules for rescue medication, statistical power of the comparison), the included trials were fundamentally similar with regard to basic design, patients and measures. It was therefore decided that it would be maximally informative to report analyses both for all agents combined, and drug by drug.
Comparisons against placebo (Figs 1 and 2)
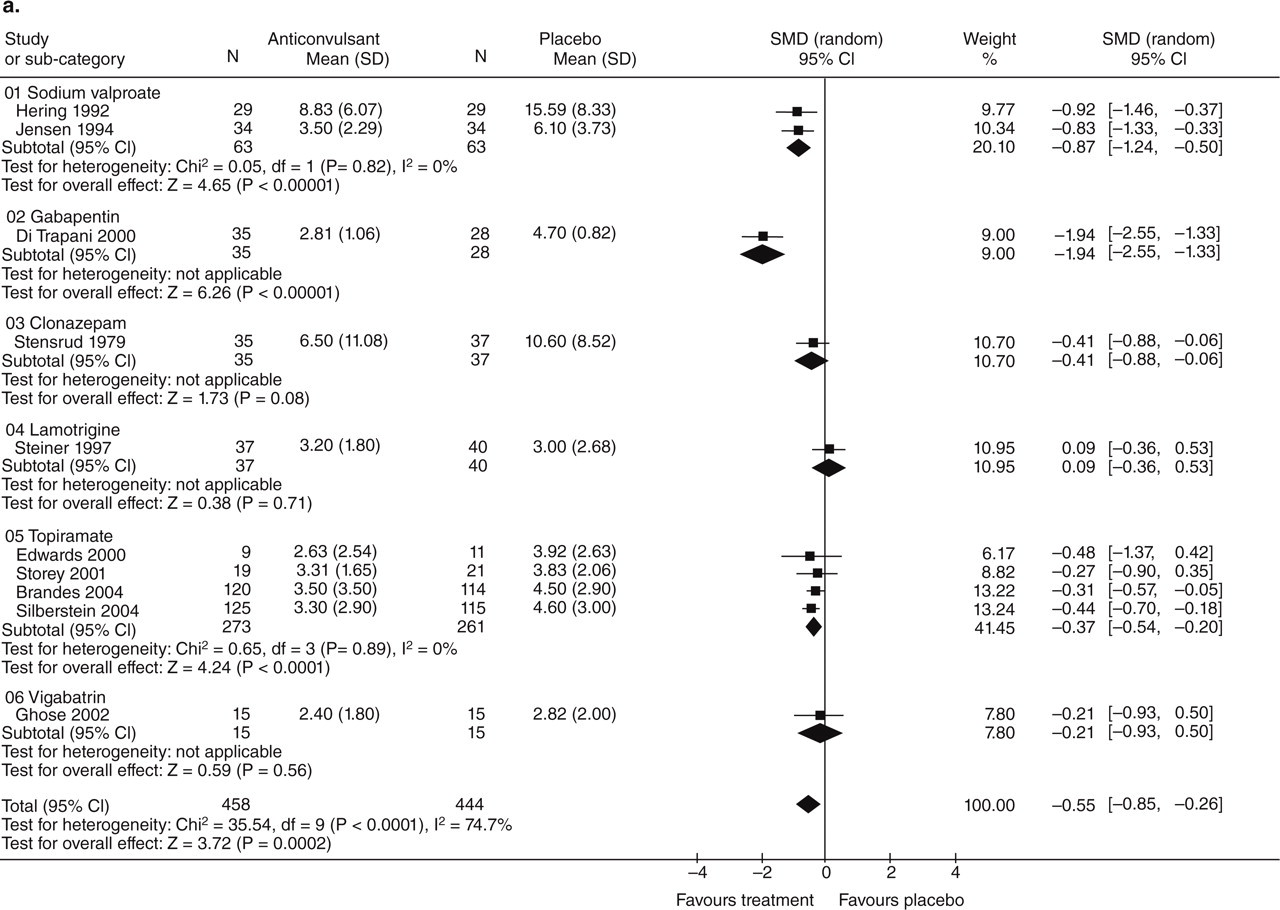
Efficacy of anticonvulsants vs. placebo. (a) Migraine frequency. (b) 50% responders.

Efficacy of various topiramate doses vs. placebo. (a) Migraine frequency. (b) 50% responders.
Anticonvulsants, considered as a class, reduce migraine frequency (per 28-day period) and increase the number of patients for whom migraine frequency is reduced by ≥ 50%, relative to placebo. In clinical terms, the observed effect corresponds to a reduction in migraine frequency of 1.3 attacks per 28 days (WMD −1.31; −1.99, −0.63) and a 2.3 times greater likelihood to experience a ≥ 50% reduction in frequency (relative risk 2.25; 1.79, 2.84).
Two trials of sodium valproate (21, 22) showed a significant reduction in migraine frequency in the active group, and one trial (22) showed a 50% responder rate significantly superior to placebo. Four trials of divalproex sodium (20, 23–25) found a significantly superior 50% responder rate. It is notable that the largest of these four studies (25) found no significant difference between active treatment and placebo.
In the combined analysis, topiramate (16, 17, 26, 27) showed a significantly greater reduction in migraine frequency against placebo. Separate analysis of all the data on the three topiramate doses found that the 200-mg dose led to significantly lower headache frequency than did placebo, as did 100 mg, whereas 50 mg did not. Furthermore, in the combined analysis of the 50% responder rate, topiramate was significantly superior over placebo, although there is noticeable variability in the ORs across these studies (16–18, 26–28). Separate analysis showed that all three doses were significantly better for this outcome measure.
In the smaller trials, gabapentin was statistically superior over placebo in reducing migraine frequency (29). Another trial (30) demonstrated a significantly higher 50% responder rate of active treatment. One trial of carbamazepine demonstrated a significantly higher 50% responder rate of active treatment (31). Neither lamotrigine (32), clonazepam (33), vigabatrin (34) nor acetazolamide (35) were significantly different from placebo, although it should be mentioned that the latter trial was discontinued prematurely because of the high number of adverse event-related withdrawals.
Dose comparisons of single active drugs (Fig. 3)

Dose comparison analyses. (a) Gabapentin 1200 mg vs. 2000 mg. (b) Sodium valproate 21–50 μg/ml vs. > 50 μg/ml. (c) Topiramate, frequency. (d) Topiramate, responder rate.
Five trials contributed to this analysis, providing data for three different drugs.
Comparison of different doses of sodium valproate by measuring serum valproate concentrations showed that lower (21–50 µg/ml) serum levels gave rise to slightly but significantly lower headache frequency than higher ones (> 50 µg/ml) (36). An explanation for this counterintuitive observation was not provided. For topiramate, data from two studies (16, 17) were used to directly compare 50 mg, 100 mg and 200 mg, with additional data from another trial (18) contributing to the comparison between 200 mg and 100 mg. The 200-mg dose was significantly superior to 50 mg both in terms of reducing migraine frequency and increasing the number of 50% responders. Likewise, 100 mg was superior to 50 mg. The 200-mg dose was not significantly superior to 100 mg for either outcome measure. It is important to note, however, that none of these studies was designed to have the statistical power to make comparisons between doses.
In one trial comparing daily doses of 1200 mg and 2000 mg gabapentin, there were no significant differences between the groups on either outcome measure (37).
Comparisons against active drug (Fig. 4)

Active comparator analyses. (a) Divalproex vs. propranolol. (b) Sodium valproate vs. flunarizine. (c) Propranolol vs. topiramate. (d) Topiramate vs. valproate.
Sodium valproate and flunarizine were not significantly different in the number of 50% responders (19), nor were divalproex sodium and propranolol (20). A comparison of propranolol with 100 mg of topiramate showed no significant difference in the number of 50% responders (18). Although a comparison trial of topiramate 50 mg with sodium valproate 400 mg reported no significant differences between the two drugs (38), our analysis of post-treatment mean headache frequencies demonstrated a slight but significant advantage for topiramate over valproate. It should be noted that the doses used in this study are not those used in routine clinical management of migraine.
Safety
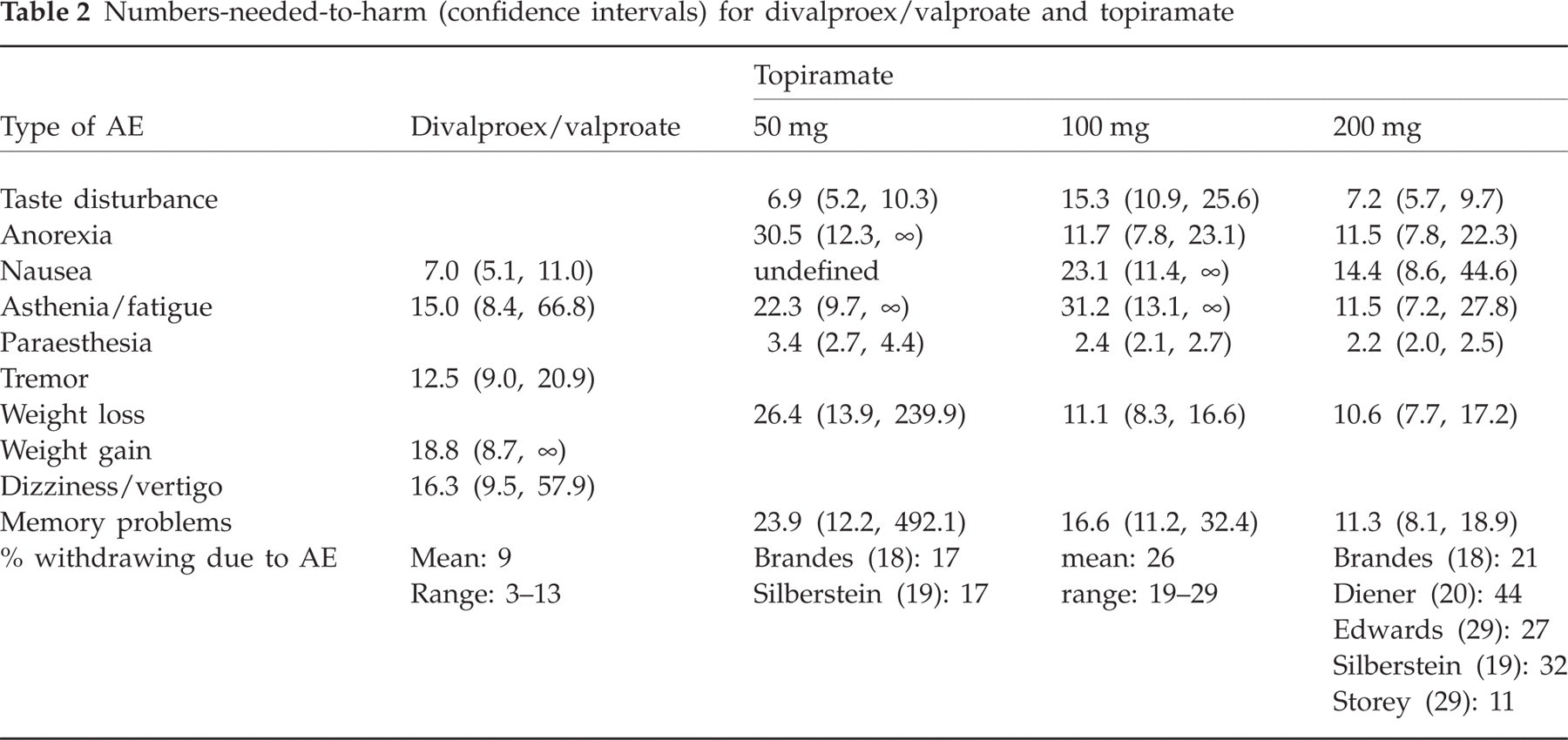
The range of adverse events considered, and the method of their reporting, varied considerably and did not allow a quantitative safety analysis for the majority of anticonvulsants studied. Only for valproate, divalproex and topiramate could a safety analysis based on the frequency of adverse events reported be performed (Table 2). Because of the fundamental similarity of sodium valproate and divalproex sodium, safety data from the six trials of these drugs against placebo were analysed together.
Numbers-needed-to-harm (confidence intervals) for divalproex/valproate and topiramate
Discussion
Meta-analysis of the 23 studies included in this review suggests that anticonvulsants are efficacious for the prophylaxis of migraine. Mean migraine frequency is significantly reduced (by approximately 1–2 attacks per month) compared with placebo, and patients are more than twice as likely to have a ≥ 50% reduction in the number of migraine attacks with anticonvulsants than with placebo.
The overall effect can be mainly attributed to the studies with topiramate and valproate/divalproex. Topiramate has been investigated in six trials against placebo and with the largest number of patients. The three larger trials (16–18) all show statistically significant superiority of 100-mg and 200-mg daily doses over placebo, and this observation is supported by the favourable 50% responder rate of another 100-mg trial (28). The 50-mg dose was efficacious on the 50% responder outcome, but not on monthly migraine frequency end-point. Two small preliminary trials of the 200-mg dose (26, 27) were equivocal, in that the analysis for migraine frequency did not reveal a significant difference from placebo in either study, although one trial reported topiramate 200 mg to be significantly more efficacious considering 50% responder rates (26). Both trials were almost certainly underpowered. Of the six included studies on valproate/divalproex, five showed a statistically significant superiority of active treatment over placebo. We were unable to analyse data on reduction in mean migraine frequency from the most recent and largest trial of divalproex sodium (25), but the investigators' analysis demonstrated a statistically significant difference between active treatment and placebo for this outcome. The clinical relevance of this effect was, however, less compelling, as both the investigators' analysis and our own found no significant difference between treatments in the number of patients reporting a ≥ 50% reduction in migraine frequency. Gabapentin was investigated vs. placebo in two trials, both of which demonstrated reasonable to good efficacy (29, 30). In a single trial, lamotrigine was no more effective than placebo in reducing migraine frequency, but this result is likely to have been influenced by a large difference in baseline frequency between active treatment and placebo groups (32). Also in single trials, acetazolamide (35) and vigabatrin (34) were found to be no more effective than placebo. Two trials from the 1970s investigated older drugs, carbamazepine (31) and clonazepam (33), of which only carbamazepine showed evidence of superiority over placebo. The age of these trials means that recent diagnostic standards (10, 11) could not be employed.
The studies including more than one dose of a single drug were generally not designed to enable direct dose comparisons, and the results of the dose comparisons reported here should therefore be viewed with caution. The comparisons suggest that the 200-mg dose of topiramate is no more effective than 100 mg, and also that gabapentin 2000 mg is no more effective than 1200 mg. The finding that lower serum valproate levels produced lower migraine frequency than higher serum levels is somewhat counterintuitive, has not been replicated and should be regarded as preliminary (36).
The four trials using active comparators found no significant difference in efficacy between sodium valproate and flunarizine (19), between divalproex sodium and propranolol (20), nor between topiramate and propranolol (18), but a slight but significant advantage of topiramate over valproate (38). It should be noted that all four trials are potentially problematic for reasons including lack of blinding, insufficient statistical power and possibly incomplete statistical analysis. Further well-designed trials, both of anticonvulsants against other drug categories and comparing different anticonvulsants, are desirable.
Anticonvulsants do not appear to give rise to an unexpectedly high rate of adverse events when used for migraine prophylaxis, although clearly (i) nausea is a problem when trials of sodium valproate and divalproex sodium are considered together, and (ii) a large percentage of patients taking topiramate report paraesthesia. It should also be noted that drop-outs due to adverse events are somewhat higher in trials of topiramate than would generally be expected on the basis of trials of other anticonvulsant drugs, particularly sodium valproate or divalproex sodium.
These relatively straightforward results should be viewed with caution. As usual in the context of clinical trials research, there is considerable heterogeneity in both headline results and general levels of analytic and statistical sophistication. It is appropriate therefore to review a number of caveats and methodological issues.
As investigations of the efficacy of various agents become more commonplace, it seems increasingly important that scientists and clinicians are at least aware of the trial guidelines suggested by the IHS (39). Even if these guidelines cannot—for operational or scientific reasons—be adhered to in their entirety, they provide a useful consultative framework at the early stages of trial design.
Several difficulties arose in deriving adequate information from the results of the 23 included studies. First, means and standard deviations were not always fully reported for each phase of trials. In tandem with this problem, reported measures of variability—either appearing in the text, tabulated, or as error bars in graphs—were not always adequately described or labelled. Second, methods of statistical analysis were generally under-specified, leading in some cases to lack of clarity as to which comparisons were significant and which were not. Third, there was considerable variability in how intent-to-treat analyses were performed. In a few cases, this gave rise to uncertainty about the numbers of patients continuing to each phase of the trial.
Bearing in mind the limitations invoked by the methodological and reporting issues mentioned above, this review nevertheless has helped to provide a rational framework for the application of anticonvulsants to the preventative management of migraine headache in real-life clinical practice. It can be concluded from this review that sodium valproate and divalproex sodium are of proven efficacy in migraine prevention and are suitable for routine clinical use, but probably not more effective than propranolol or flunarizine. One important caveat should be noted: these drugs are known to be teratogenic (40), and appropriate caution must be used when prescribing to women of child-bearing age. Similarly, topiramate has been proven to be efficacious in routine clinical management of migraine and possibly marginally more effective than valproate, but no more effective than propranolol. On a case-to-case basis, rational prescriber preferences may be appreciated due to differences in side-effect profile. Very few data pertaining to the likely teratogenicity of topiramate in humans are available; appropriate caution in prescribing is again warranted. Although adverse events are reported by a large proportion of migraine patients treated with sodium valproate/divalproex sodium or topiramate, these are usually mild and of a non-serious nature. It is the clinical impression that adverse events occur more frequently and earlier in the course of treatment with topiramate, although this observation has not been extensively studied. Thus it can be concluded that both drugs are reasonably well tolerated.
The evidence derived from trials of gabapentin suggests a beneficial effect in migraine prophylaxis, but this drug needs further evaluation. Although three clinical trials of reasonable size have been reported, the interpretation of two is hampered by some aspects of their method or data analysis, whereas the third does not provide unequivocal evidence for efficacy, as it is primarily a dose comparison study. In the meantime, it may be advocated with some reservation that gabapentin may be used for those cases that are difficult to manage with other currently available strategies, since it has a reasonable tolerability and safety profile.
For all other anticonvulsants there is, at present, either insufficient or no evidence concerning either efficacy or tolerability to warrant their use in clinical practice. Although anticonvulsants as a class, and the newer anticonvulsants in particular, appear to be amply used in the management of headache, as judged by the vast number of publications of small retrospective series and studies in chronic daily headache or transformed migraine, these results cannot be translated as compelling evidence, and thus we would like to argue against the liberal use of anticonvulsants other than those mentioned above.
Footnotes
Acknowledgements
During the course of updating the Cochrane review, Professor E. P. Chronicle, PhD, sadly passed away on 9 February 2007. W.M.M. wishes to acknowledge Professor Chronicle's major contribution and tremendous effort in compiling all statistical analyses and much of the text of the review. Without his relentless dedication the review would have never seen the light of day.
