Abstract
Background
The prophylactic treatment of migraine includes anticonvulsant drugs such as valproic acid and topiramate. However, these substances are often poorly tolerated by migraine patients. So far levetiracetam has hardly been studied as an episodic migraine prophylactic agent in adults.
Objective
To perform a prospective pilot study for the evaluation of the efficacy and tolerability of levetiracetam in the prophylactic treatment of episodic migraine.
Methods
Fifty patients with episodic migraine were enrolled in this prospective, open label study. After a baseline period of four weeks, patients received 1,000 mg (starting dose 500 mg) bid levetiracetam for 12 weeks. Migraine frequency and accompanying symptoms were recorded in a headache diary. The primary endpoint was the comparison of attack frequency during the baseline with attack frequency during the last four weeks of treatment (treatment period 3).
Results
In the Intent-To-Treat analysis, 46% of the patients had a migraine reduction of more than 50% in the third period as compared to the baseline period. The mean number of migraine attacks decreased from 5.2 +/− 2.1 (baseline) to 3.4 +/− 2.7 (period 3). The most frequently reported side effects were somnolence, nausea, and weight gain; all were mild and transient. In a post-hoc comparison, responders to levetiracetam had significantly less migraine attacks at baseline and had significantly more often migraine with aura.
Conclusion
The data suggest that levetiracetam has some potential in the prophylactic treatment of episodic migraine which seems, however, to be not superior to that of other anticonvulsant drugs. Levetiracetam was well tolerated and showed better efficacy in patients with migraine with aura and in less affected migraine patients. A larger placebo-controlled, double-blind study in adults seems justified on the basis of these data.
Introduction
Levetiracetam has proven to be an effective and well tolerated drug in the add-on treatment of focal epilepsies in adults and in the monotherapy of epilepsies in general (1,2). However, published prospective studies on the efficacy of levetiracetam in migraine prophylaxis of adults are very rare. Early case series on patients with migraine reported a responder rate to levetiracetam treatment of 58% (3), 55% (4), and – in migraine with aura – of 44% with attack freedom (5). Another small case series in elderly patients showed a decrease of monthly migraine days from 12.2 to 1.3 by levetiracetam (6). These case series used a daily dose of about 1,000 mg levetiracetam. In an open prospective study in the USA, levetiracetam in a daily dose between 1,000 and 3,000 mg was effective in reducing the number of migraine days in patients with transformed migraine (7). A small double-blind placebo-controlled trial from India confirmed a significant efficacy of levetiracetam with a responder rate of 64% for levetiracetam and 22% for placebo (8). In an Australian placebo-controlled trial on chronic daily headache, however, levetiracetam did not show a significant improvement of headache frequency (9). Of note, intravenous levetiracetam was also reported to be efficacious in a case report on status migrainosus (10).
Levetiracetam was also compared to valproic acid in migraine prophylaxis. In a randomized, double-blind trial, levetiracetam was significantly less effective than valproic acid with respect to migraine frequency, severity and Migraine Disability Assessment (MIDAS) score (11). In a similar study, valproic acid and levetiracetam were compared to placebo; this study showed similar efficacy of valproic acid and levetiracetam but a significant superiority of both substances against placebo (12).
In children and adolescents, an open retrospective study found levetiracetam in a dose between 125 and 750 mg bid to be effective in migraine prophylaxis (13). Another open-label study also showed a significant reduction of migraine frequency by levetiracetam in paediatric migraine in 18 out of 20 participants (14). This was confirmed by a placebo-controlled double-blind study in children and adolescents with a responder rate for placebo of 30% and for levetiracetam of 68% (15).
A recent review and meta-analysis concluded that levetiracetam can significantly reduce headache frequency and severity in adults and children and can, thus, be a therapeutic option for migraine prophylaxis, especially when considering the adverse effects or teratogenicity of other preventive treatments (16). This review was based on studies on chronic/transformed migraine, not on episodic migraine.
Conventional migraine prophylaxis with betablockers, amitriptyline, or flunarizine has the limitation that many patients show poor adherence due to dizziness, weight gain, sleep disturbances, and sometimes depression (17). Anticonvulsants with established use in migraine prophylaxis show similar side effects except topiramate which can lead to weight loss (17). Therefore, new migraine prophylactic drugs with a high efficacy and a high tolerability are warranted. Levetiracetam has the advantage that it shows only very few and mild side effects as compared to other anticonvulsants such as valproic acid and topiramate.
This prospective open-label study was designed to evaluate the prophylactic efficacy of levetiracetam monotherapy in episodic migraine prophylaxis in an adult sample which has not been studied yet. Further, we aimed to identify subgroups of migraine patients with a particular benefit from this substance.
Methods
This study was designed as a prospective, open, monocentre, non-randomized trial with a run-in period of 4 weeks (baseline) and a treatment period of 12 weeks on episodic migraine according to the diagnostic criteria of the International Headache Society (18). Patients were treated with levetiracetam 1,000 mg bid as monotherapy (500 mg bid in the first week). Migraine frequency was evaluated by a paper-based headache diary. The number of migraine attacks, the number of migraine days, occurrence of accompanying symptoms (nausea, vomiting, photophobia, phonophobia), and the amount of drugs for acute attack treatment were registered for every four week period (baseline and three periods of four weeks each).
The inclusion and exclusion criteria are listed in Table 1. Patients were recruited from the supraregional headache outpatient clinic at the Department of Neurology, University of Münster. The primary efficacy parameters were defined as follows:
Inclusion and exclusion criteria for the patients to be enrolled.

(1) number of migraine attacks per period compared to the run-in period (attacks separated by at least 24 h free of symptoms);
(2) percentage of patients experiencing at least 50% reduction in migraine attack frequency in the last treatment period as compared to the run-in period (defined as ‘responders’).
In addition, secondary outcome parameters were defined such as the number of migraine days per treatment period; number of acute migraine medication drugs per treatment period; frequency of the occurrence of migraine-specific accompanying symptoms (nausea, vomiting, photophobia, phonophobia); and frequency and type of adverse events possibly or probably related to the study drug as judged by the investigator. The study design followed the recommendations and definitions of the International Headache Society for trials on migraine prophylaxis (19).
The study was approved by the local ethics committee. All patients gave written informed consent prior to the baseline period.
We regarded a power of 80% to detect a 20% difference in the primary efficacy parameters per 4 week period compared to run-in period as sufficient for this pilot study. The placebo rate in controlled trials on migraine prophylaxis is about 20% to 30% for the primary efficacy parameters. Based on the assumptions of a type I error (alpha) of 50%, an expected responder rate of 50% (30% placebo responder assumed), and standard deviation of change with less than 50% (absolute), the Wilcoxon one-sample test resulted in a sample size of 50 patients. The sample size calculation was performed using nQuery Advisor 4.0 (Statistical solutions) for a one-sample t-test. In case of normally distributed data, the statistical power of the Wilcoxon test is only slightly lower.
The statistical analysis was performed on the intent-to-treat (ITT) population. Any patient who received treatment was included in this population. Missing follow-up data were entered in the database as the last observation carried forward. The primary efficacy parameters were also analysed for the per-protocol (PP) subset of the ITT population, which is characterised by the following criteria:
(1) Completion of the whole study period; (2) Availability of measurements of primary efficacy parameters; (3) Absence of any important protocol violation.
The data are presented as arithmetic mean with standard deviation or as percentage. For the comparison of baseline versus treatment data, the Wilcoxon-test was used. For comparison of the data responders versus non-responders, the Mann-Whitney-U-test and the X2-test (Fisher’s exact test if applicable) were used. The significance level was set at p = 0.05.
Results
In total, 58 patients were screened for the study. Out of these, 50 entered the treatment period, the remaining eight patients did not have enough migraine attacks in the baseline period to qualify for treatment entry (n = 6) or they withdrew consent (n = 2). The demographic data of the ITT population are presented in Table 2. Eighty eight per cent of the patients had experience with other migraine prophylactic drugs before, 12% had not. The mean number of previous migraine prophylactic drugs used was 2.5 +/− 1.9 (range 0 to 8). 64% of these prophylactic medication treatments were judged as inefficacious by the patients (mean number given as efficacious 0.5 +/− 0.9, range 0 to 4).
Demographic data of the patients at the onset of treatment presented for the ITT population (range given in brackets).
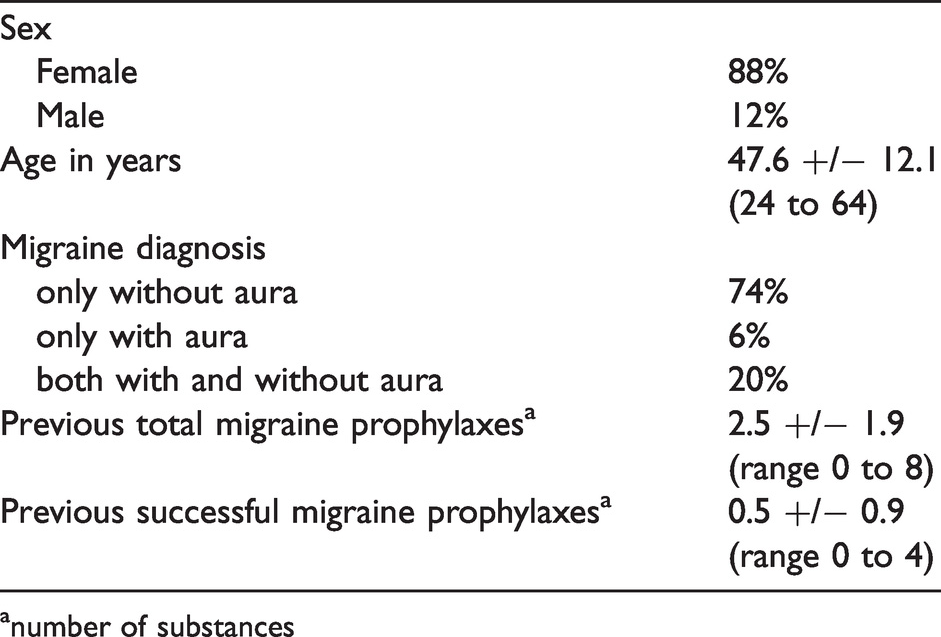
anumber of substances
In total, six patients dropped out during the treatment period. One of these patients was lost to follow-up. The other five patients dropped out due to side effects, three of them because of cognitive disturbances and two of them because of nausea and dizziness.
With respect to the primary efficacy parameters, 46.0% of the ITT population were responders in the last treatment period (52.3% of the PP population). The mean number of migraine attacks and the other secondary efficacy parameters for the ITT population are presented in Table 3. The mean number of migraine attacks for the PP population were 5.2 +/− 2.1 (baseline), 4.2 +/− 2.4 (period 1), 3.5 +/− 1.8 (period 2), and 3.4 +/− 2.7 (period 3) and, thus, nearly identical to the data of the ITT population. The decrease of migraine attacks, of days with migraine, and of acute drugs for migraine attack treatment as compared between baseline and the third treatment period was significant (p < 0.001, p = 0.001, p < 0.001; respectively).
Efficacy parameter given as arithmetic mean and standard deviation or in percentage of the ITT population (range given in brackets).

The side effects which were possibly or probably related to the study drug as judged by the investigator are listed in Table 4. The most frequently reported side effects were somnolence, nausea, and weight gain. No serious adverse event occurred.
Side effects possibly or probably related to levetiracetam as judged by the investigator given as absolute numbers of patients.

After analysing the total study population, we performed a post-hoc comparison between the patients with and without response (i.e., decrease of migraine attack frequency in the third treatment period as compared to baseline by 50% or more). The results of this comparison are presented in Table 5. Responders showed significantly fewer migraine attacks, migraine days, and intake of acute migraine drugs at baseline. Furthermore, they had significantly fewer trials of migraine prophylaxis in their history, these trials were significantly more frequently successful than in the non-responders. Finally, responders to levetiracetam often had significantly more migraine with aura than non-responders.
Demographic and migraine data of the study population after baseline assessment presented separately for the responders and the non-responders. Statistical comparison by Mann-Whitney-U-test and X2-test (Fisher’s exact test, if applicable).

Discussion
This study was powered to show a superiority of levetiracetam in migraine prophylaxis by 20% as compared to a placebo response by 30%. This aim was not reached for the ITT population. In the PP population, however, the response rate was above 50%. In general, the ITT and the PP population did not differ in a relevant way for all other outcome parameters.
This result does not indicate that levetiracetam is of major efficacy in migraine prophylaxis or superior to other established migraine prophylactic drugs. The decrease of migraine attack frequency, of days with migraine attacks, and of the intake of acute migraine drugs was significant during the study period as compared to the baseline. However, this decrease, and also the percentage of responders, lies within the range known from typical migraine prophylaxis trials (20) and does not support the hypothesis that levetiracetam is of better efficacy than the approved migraine prophylactic drugs. It has to be considered that the study population was recruited from a tertiary university headache centre with mostly very severely affected migraine patients. This is reflected by the high number of previous migraine prophylactic drugs taken by the patients with a low number of successful ones. Therefore, further evaluation of levetiracetam as a migraine prophylactic drug in severely affected migraine patients cannot be recommended on the basis of these study results. However, it might be that levetiracetam is effective in an average migraine population recruited from primary or secondary care and with less severe migraine. This hypothesis is supported by the subgroup analysis of responders in our study and has to be studied in further trials which should be placebo-controlled.
Levetiracetam was in general well tolerated and did not lead to relevant side effects or adverse events. All side effects were mild and transient, serious adverse events were not noted. Somnolence and dizziness were the most frequently reported side effects which is in concordance with other trials on levetiracetam in epilepsy treatment (2). Emotional lability was also reported in our study and is a typical but rare adverse event under levetiracetam (21). The number of drop-outs due to side effects was very low as compared to other migraine prophylaxis trials and confirms the good tolerability of this drug.
When analysing the results in detail, however, some interesting observations can be made which might lead to further investigations of levetiracetam in migraine treatment. First of all, the improvement of accompanying symptoms was remarkable, in particular vomiting improved best out of all these symptoms. It might be that levetiracetam is of specific efficacy in the prophylactic treatment of vomiting (and perhaps nausea) which in some patients can be even more impairing than the headache itself (22).
Further interesting observations were obtained when comparing responders and non-responders (defined as a decrease of migraine frequency by 50% or more in the last treatment period as compared to baseline). The responders to levetiracetam had, in average, a lower number of migraine attacks and migraine days at baseline. This finding adds to the hypothesis that one reason for the negative primary outcome of this study was that the patients were recruited from a tertiary university headache centre and not from primary care. Many migraine patients in tertiary care might be in part resistant to prophylactic treatment. Furthermore, levetiracetam was more effective in migraine patients with aura than in those without aura. This observation was independent from the lower attack frequency in migraine with aura. A confounding factor was the better response to other prophylactic drugs in the responders. It might be that a subgroup of migraine patients is in general a better responder to prophylactic drugs. A previous case series (5) had also focussed on levetiracetam treatment of migraine with aura and found a marked efficacy in this particular patient group. This interesting observation has also been made for lamotrigine, which failed to show efficacy as a migraine prophylactic drug in general (23) but which was successful in the prophylaxis of migraine with aura (24,25). It might be that the mechanism of some anticonvulsants in migraine prophylaxis is more related to migraine aura than to migraine headache.
This study has significant limitations. First, of course, this is an unblinded study without a placebo control, therefore the results can only be preliminary and can only be the basis for a larger double-blind controlled study. The lack of placebo control is in particular relevant since the responder rate was not very high; therefore the responder rate found in this study probably reflects the real responders. However, we believe that the results are robust enough to support further research into levetiracetam as a migraine prophylactic drug in certain subgroups (e. g. migraine with aura). Second, the dose of levetiracetam chosen in this study is arbitrary. A total dose of 2,000 mg levetiracetam per day is a little above the average used in epilepsy treatment. It might be that lower doses are of similar efficacy in migraine treatment. This could be evaluated also in a larger double-blind controlled trial with placebo and two different doses of levetiracetam. Third, the sample size is relatively small (however large enough to support the predefined assumption of the study) and recruited only in one tertiary centre. This can include a bias since every tertiary centre depends on the admission by other centres or clinics.
In conclusion, this prospective open-label pilot study showed that levetiracetam 1,000 mg bid was effective in reducing migraine attack frequency by at least 50% in 46% of all patients studied in a tertiary headache centre. The mean reduction of migraine attack frequency was 29%. This rate is lower than expected but still in the range of responder rates known from placebo-controlled migraine prophylaxis studies. Analysing the responders, it is suggested that those migraine patients who are not severely affected and those migraine patients with a migraine aura benefit most from prophylactic treatment with levetiracetam. We focussed on episodic migraine. It might be that the efficacy of levetiracetam is different in larger studies on chronic migraine.
Clinical implications
Levetiracetam 1,000 mg bid might reduce attack frequency, headache days and days with acute drug intake in episodic migraine. Levetiracetam 1,000 mg bid was well tolerated in migraine treatment. Patients with less severe migraine might respond better to levetiracetam than patients with a high migraine frequency. The findings have to be confirmed in a larger placebo-controlled, double-blind study.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SE reports serving as a consultant for the following corporations: Lilly, Lundbeck, Novartis, Perfood, Teva. MM reports serving as a consultant for Novartis and Teva. AF reports serving as a consultant for Lilly. OS and IWH report no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by an unrestricted grant of the company UCB Chemie GmbH Germany. The study was initiated and designed by the primary investigator. There are no other conflicts of interest reported by the investigators.
