Abstract
Calcitonin gene-related peptide (CGRP) and nitric oxide (NO) are two molecules shown to have a role in migraine pathophysiology. Our objective was to test the hypothesis that migraine subjects are particularly sensitive to these signal molecules. The cutaneous microvascular responses to endothelial and non-endothelial dependent dilators were tested using laser Doppler flowmetry in combination with iontophoresis. The blood flow responses to iontophoretic administration of the endothelium-dependent vasodilator acetylcholine (ACh), or to the endothelium-independent dilators sodium nitroprusside (SNP) and CGRP, and to local warming (44 °C) were compared in this controlled trial. The design was that of two arms: patients diagnosed with migraine without aura (n = 9) for >10 years were compared with nine healthy subjects matched for age and gender (seven female and two male, age range 30-60 years). Iontophoretic administration resulted in local vasodilation. ACh induced a relaxation of 1225 ± 245% (relative to baseline) in controls and 1468 ± 368% (P > 0.05) in migraine. The responses to SNP were 873 ± 193% in controls and 1080 ± 102% (P > 0.05) in migraine subjects. The responses to CGRP were 565 ± 89% in controls and 746 ± 675% (P > 0.05) in migraine patients. The responses to local heating which induced maximum dilation did not differ between the groups (1976 ± 314% for controls and 1432 ± 226% in migraine; P > 0.05. We conclude that there is no change in the microvascular responsiveness of the subcutaneous microvasculature in migraine.
Introduction
It has been hypothesized that there is a difference in sensitivity of the vasculature to key molecules involved in migraine pathophysiology, calcitonin gene-related peptide (CGRP) and nitric oxide (NO) (1, 2). This view is supported by the human in vivo observations that migraine subjects are more sensitive than non-migraine subjects to the systemic administration of the NO donor glyceryl trinitrate (GTN) (3) and CGRP (4) in inducing migraine-like headache attacks. Furthermore, increased vasodilation of the middle cerebral artery to organic nitrates has been reported in migraine patients compared with control subjects (3). Other data in support of an arterial supersensitivity to these molecules emanate from work on the NO releaser histamine (5) and the antagonism of migraine attacks by a NO synthase inhibitor (6). A connection between CGRP and NO has been implicated by both experimental (7) and clinical data (8).
Since migraine is associated with enhanced risk for cardiovascular disorders, it has been suggested that there is a general alteration in the arterial function in migraine (9). A study of the forearm blood flow response demonstrated no difference in the vasodilator response to intrabrachial infusion of CGRP or sodium nitroprusside (SNP) (10). On the other hand, enhanced peripheral sensitization of the skin in conjunction with migraine attacks (11) has been described, and also of central sensitization (12). Therefore, the aim of the present study was to examine another part of the circulation, the cutaneous microvascular bed with mainly arterioles and capillaries, and to determine if there is altered sensitivity of the peripheral microcirculation using iontophoretic administration of CGRP and the NO donor SNP (acting on the smooth muscle cells) and acetylcholine (ACh, which releases endothelial NO) by using laser Doppler flow measurements.
Methods
Design
The study was a controlled trial with two arms: nine patients diagnosed with migraine without aura for > 10 years were compared with nine healthy subjects and matched for age and gender. The two groups consisted of seven women and two men aged 30–60 years and randomly included. The experiments were carried out during a headache-free interval. Migraine patients were kept on their prescribed medication (candesartan, n = 2; citalopram, n = 2) but refrained from β-adrenergic blockers (metoprolol, n = 1) and caffeine on the day of measurement. There were no smokers. Migraine patients were recruited as ambulatory subjects at the Emergency Department conforming to the International Headache Society criteria; the healthy matched individuals were randomly found in the community registry. The study was performed at the Clinical Research Unit at Lund University Hospital and approved by the Ethics Committee.
Analysis
Inflammatory markers, C-reactive protein (CRP) and cytokine levels in plasma were also measured (at the Department of Clinical Chemistry, Lund University Hospital) and the subjects answered a lifestyle questionnaire (13, 14).
Blood flow measurements
Cutaneous blood flow was measured using the PeriFlux System 5000 (Perimed, Järfälla, Sweden). This method is non-invasive and gives minimal discomfort to the studied subject. Laser-generated light of wavelength 780 nm is directed to the skin using a fibreoptic probe. The light reflected from moving blood cells in the superficial cutaneous microvessels undergoes a shift in frequency (Doppler effect) that is proportional to the number and velocity of the moving blood cells. The laser Doppler output is semiquantitative and all data are presented as percent change compared with baseline perfusion value (perfusion unit = PU). The temperature of the skin is continuously recorded; it was stable throughout the recording period.
Iontophoresis
Constant current iontophoresis was used to enhance the perfusion of charged molecules into the skin of the dorsal side of the lower arm. The current alone did not affect the blood flow (results not shown). Endothelium-dependent vasodilation was provoked by iontophoresis of the cation ACh (2% dissolved in MilliQ water; Sigma, St Louis, MO, USA) using anodal current. Endothelium-independent vasodilation was provoked by iontophoresis of the negatively charged NO donor SNP (1% in MilliQ water; Sigma) using cathodal current. Human CGRP (1–37, 0.02% dissolved in MilliQ water; Sigma) is also positively charged.
All subjects rested comfortably in a supine position with the right arm on a pillow stabilizing the position, for 10 min before commencing; ambient room temperature 22–23°C. The laser Doppler probe and the drug delivery systems were attached. A chamber filled with ACh was applied to the forearm. Skin temperature was noted and basal blood flow was registered for 2 min. Addition of ACh was then begun by iontophoresis (0.2 mA at the anode for 1 min). The stimulation was repeated five times with 1 min between. SNP was applied to a new chamber (0.1 mA at the cathode). The current was switched on for 1 min for a total of four periods with 1 min between. Human CGRP was applied to the third chamber (0.2 mA at the anode) and treated the same way as for SNP. Finally, the heat response was measured. A probe was heated to 44°C and the increase in blood flow was registered under a period of 10 min, which provided the maximum vasodilation in these experiments (13, 14).
Statistics
A two-tailed paired t-test was used to compare the results between the migraine subjects and the matched control subjects. All values are given as means ±
Results
The demographics are given in Table 1. There was no difference between the groups for gender, age, body mass index or blood pressure. In addition, to test if any difference existed for inflammatory markers, we analysed CRP, tumour necrosis factor-alpha, interleukin (IL)-6 and IL-2r. Their levels were similar to those seen in other control groups (14) and did not differ between the two groups studied (Table 1). The life style questionnaire was completed in a headache-free period and did not reveal any difference between the two groups (all comparisons, P > 0.05).
Demographics in the two groups studied: migraine (n = 9) and matched controls (n = 9)
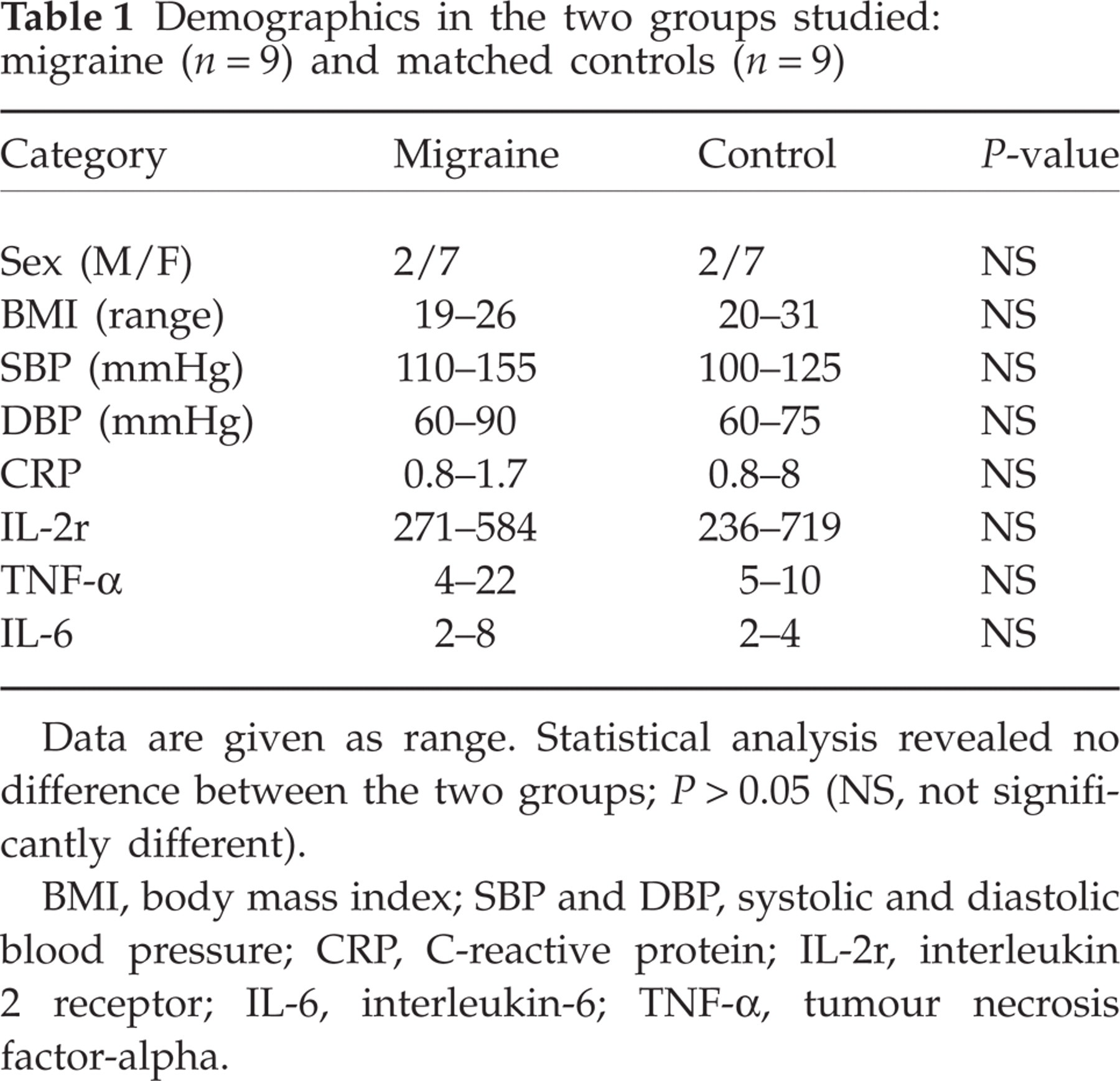
Data are given as range. Statistical analysis revealed no difference between the two groups; P > 0.05 (NS, not significantly different).
BMI, body mass index; SBP and DBP, systolic and diastolic blood pressure; CRP, C-reactive protein; IL-2r, interleukin 2 receptor; IL-6, interleukin-6; TNF-α, tumour necrosis factor-alpha.
Baseline blood flow was not different between the two groups. All procedures resulted in local vasodilation, and the responses are given below in percentage of change relative to baseline. ACh induced a mean relaxation of 1225 ± 245% in controls and 1468 ± 368% in migraine subjects (P = 0.59). The responses to SNP were 873 ± 193% for controls and 1080 ± 102% for migraine (P = 0.36). The responses to CGRP were 565 ± 89% for controls and 746 ± 675% for the migraine group (P = 0.13) (Fig. 1). The responses to local heating were 1976 ± 314% for controls and 1432 ± 226% for migraine patients (P = 0.18).

Responses (perfusion unit) to iontophoretic administration of acetylcholine (ACh), sodium nitroprusside (SNP), calcitonin gene-related peptide (CGRP) and local heating to 44°C. Values are given as means +
Discussion
We observed that in this controlled study of well-matched healthy subjects with a group of migraine subjects without aura, there was no difference in baseline flow or in microcirculatory responses to iontophoretic administration of ACh (releaser of NO from endothelial cells), SNP (NO donor), CGRP (acts directly on CGRP receptors in the vascular smooth muscle cells) or to low local heating of the skin (to 44°C). The work indicates that migraine does not involve a general difference in microvascular sensitivity; this would be implied if there was a genetic difference in vascular receptor expression. One objection to our study is that the study was carried out in subjects who were outside migraine attacks, but for obvious reasons this could not be done during attacks of migraine because autonomic activation on its own has effects on the peripheral circulation.
Another objection is whether the method is sensitive enough to record subtle changes in skin microcirculatory responses in any patient group. We have earlier observed that in subjects suffering from chronic heart failure (13, 15) and in hypertensives (14) there are alterations both in endothelial responsiveness to ACh and to smooth muscle cell responses to SNP and CGRP. Thus, the method used is sensitive and non-invasive, hence easy for the patients to comply with and useful in a clinical setting.
It has been suggested that migraine is associated with cardiovascular disorders such as Raynaud's syndrome (16), coronary disease (17, 18) and ischaemic stroke (19, 20). In support, enhanced peripheral arterial stiffness has been observed in migraine sufferers (8), but there was no enhanced sensitivity to CGRP or NO in studies of the human forearm resistance vessels (10). These data suggest that in some migraine subjects there exists a general reduced flow response to different stimuli (9). Thus, taken together with our observations on the cutaneous microcirculation, there are no data to support the view of a general systemic vascular hypersensitivity to CGRP or NO in migraine subjects.
Footnotes
Acknowledgements
This study was supported by the Swedish Research Council (grant no. 5958).
