Abstract
Nummular headache (NH) is a primary disorder characterized by chronic pain that is exclusively felt in a small area of the head surface. We describe five patients with circumscribed head pain and sensory dysfunction consistent with NH, who in addition developed colocalized trophic changes. All of them had a round or oval patch of skin depression (1-2 cm in diameter) inside of the painful area (2-4.5 cm in diameter). Three of them also showed hair loss, reddish colour, and local increased temperature. Skin biopsies were performed in three patients, and were not specific for any particular dermatological disease. Local trophic changes may be a clinical feature of NH. Together with pain and sensory disturbances, they could represent a restricted form of complex regional pain syndrome. This should be taken as a possible evolution of the underlying morbid process of NH.
Introduction
Nummular headache (NH) presents a clear-cut clinical picture characterized by chronic pain that is exclusively felt in a round or oval area of the head surface, typically 2–6 cm in diameter. The symptomatic area remains unchanged in both shape and size, and may show a variable combination of symptoms and signs of sensory dysfunction (1–10). The aetiology and pathogenesis of NH are largely unknown, although it has been regarded as an epicrania, i.e. an ‘in situ’ headache probably stemming from epicranial tissues (4, 11).
We have recently observed five patients fulfilling the diagnostic criteria of NH, who in addition developed some marked trophic changes within the symptomatic area. Since this new feature occurred in otherwise typical NH patients, it may be viewed as a potential trait of NH, and this finding may have important consequences. On the one hand, local trophic changes provide valuable clinical signs. On the other hand, the knowledge of the process that produces such trophic changes might throw light on the underlying mechanisms of NH. Here we describe the details of this latest clinical finding, and discuss possible diagnostic and pathogenic implications.
Methods
Through a 3-year period we have examined all the patients with NH, searching for any trophic changes in the symptomatic area. Diagnosis of NH was made according to the diagnostic criteria of the International Headache Society (3). Patients had to outline the painful area, which was subsequently mapped by palpation and by means of a pointed probe. Those cases with local trophic changes including thinness of the skin were selected (n = 5), and the margins of the skin signs were also depicted in each of them. The diameters of both the area of pain and the area of skin depression were exactly measured. Other dermatological features, such as hair loss, colour variations or local temperature changes, were recorded whenever they were evident upon examination. Skin colour and skin temperature were assessed before any pressure was applied on the symptomatic zone. When the patient could recall the progression of symptoms in detail, the time of onset of both pain and trophic changes was specified.
Various signs of sensory dysfunction—hypoaesthesia, hyperalgesia or allodynia—were detected in the symptomatic area. Otherwise the neurological examination was normal in all patients but one (no. 5), who had moderate weakness in girdle muscles due to a long-standing myopathy. All patients were examined by a dermatologist, who failed to find any skin lesion apart from the above-mentioned trophic changes.
An X-ray examination of the skull and a computed tomography scan or magnetic resonance imaging of the head were taken in all cases, with normal results. Routine blood work-up with erythrocyte sedimentation rate, and the screening for immunological disorders were also normal. A skin biopsy of the symptomatic area was performed in three patients, and a biopsy of the symmetrical asymptomatic area was also obtained in two of them. The procedures were always in agreement with the dictates of the local ethics committee. All patients signed an informed consent prior to their inclusion.
Results
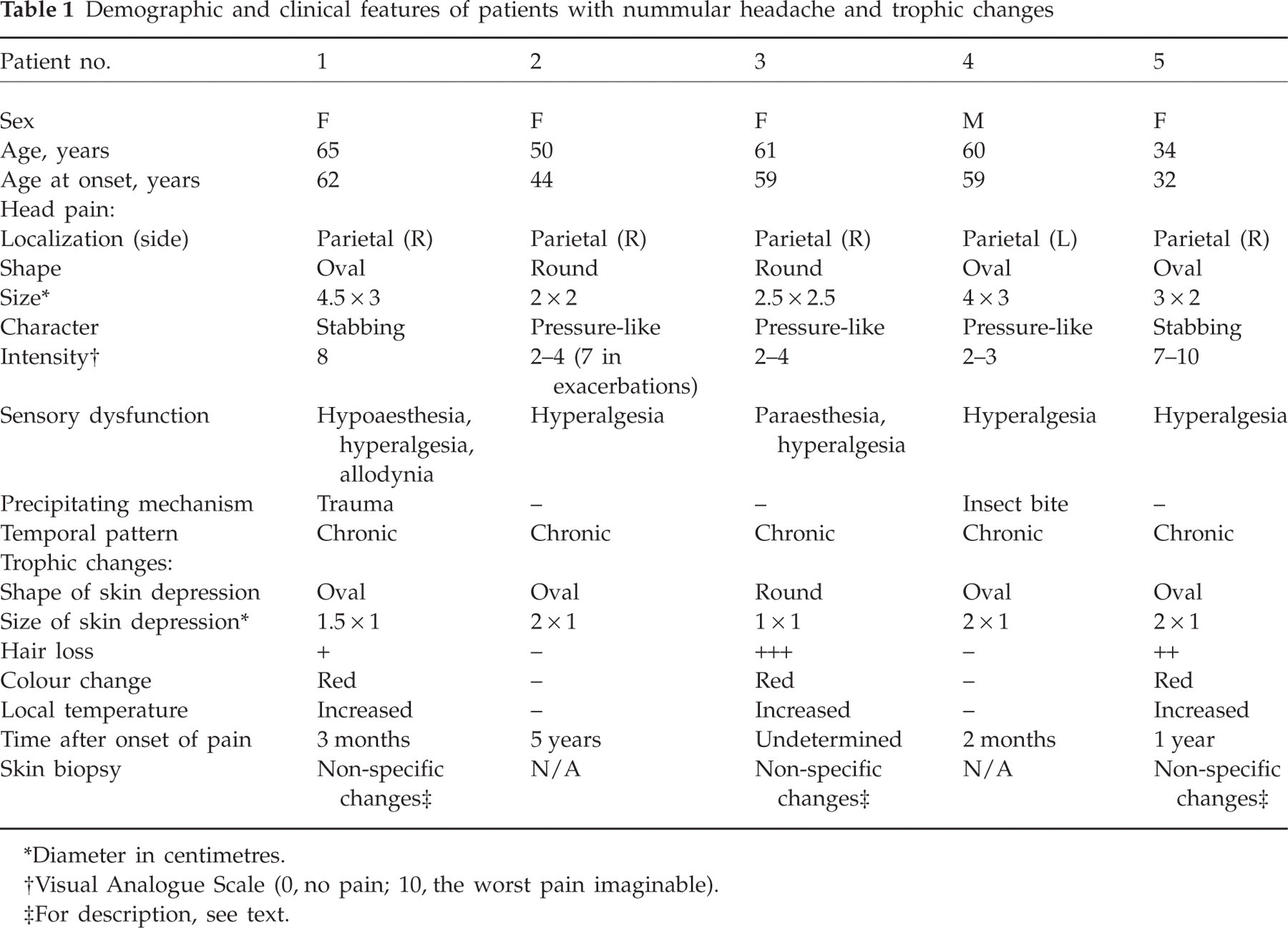
Among a total of 59 NH cases seen over the last 3 years, five had associated trophic changes. Demographic and clinical characteristics of these five patients are shown in Table 1. Two of them could identify a close temporal relationship between the symptoms and a precipitating event (a cranial trauma and an insect bite, respectively). Head pain had started spontaneously in the rest of the patients. In four patients the trophic changes clearly followed the onset of pain after a variable period of time (months to years). The remaining patient was unable to ascertain the chronological sequence. Patient 5 had started to show trophic changes 1 year after the onset of pain, and both pain and trophic changes had finally vanished in unison 1 year later. None of the patients had been treated with local injections of anaesthetics, steroids, or botulinum toxin.
Demographic and clinical features of patients with nummular headache and trophic changes
Diameter in centimetres.
Visual Analogue Scale (0, no pain; 10, the worst pain imaginable).
For description, see text.
In all five patients the area of trophic changes fell within the limits of the painful area, and was either oval or round (see Table 1). The diameters of the area of skin depression (1–2 cm) were consistently smaller than those of the area of pain (2–4.5 cm). In addition to a patch of skin depression inside the painful area, three patients showed hair loss, reddish colour, and local increased temperature in the same territory (Fig. 1). The sensitivity to pain stimuli was always enhanced in the whole symptomatic region, but mainly within the atrophic zone. Skin biopsies were performed in three patients (nos 1, 3 and 5). Histopathological findings documented the trophic changes, but were never specific to a particular dermatological disease:

Patient no. 3. A rounded patch of complete hair loss in the scalp is observed. Careful inspection also revealed that the skin over the area of alopecia was slightly erythematous, while palpation demonstrated skin depression and toughness plus a local increase of temperature. Alopecia and skin depression are confined within the symptomatic painful area, which exceeds the borders of the trophic changes (skin depression, 1 × 1 cm; painful area, 2.5 × 2.5 cm).
Patient no. 1
A biopsy specimen in vertical sections showed fibrosis of the follicles, and only minimal perivascular superficial mononuclear cell infiltrates could be observed in epidermic and infundibular hair regions. No remarkable changes were detected in epidermis, sweat glands, collagen bundles or elastic fibres. In particular, there was no increase of mucin. Fungal infections or other types of infection were ruled out by appropriate techniques including periodic acid–Schiff (PAS) and Grocott stains.
Patient no. 3
A biopsy specimen in vertical sections showed some flattening of the epidermal rete ridges. There was absence of all adnexal structures, including follicles and sweat glands. Collagen bundles were randomly oriented, but thickened and sclerotic. Elastic fibres showed solar elastosis. Neither inflammation nor mucin increase could be observed. Mycotic and other infections were ruled out by appropriate techniques including PAS and Grocott stains.
Patient no. 5
A biopsy specimen in vertical sections showed 11 hairs (seven terminal anagen hairs, two terminal catagen/telogen hairs and two vellous hairs); the terminal:vellous ratio was 4.5 : 1, and the anagen:telogen percentages were 78% : 22%. No miniaturization of hairs, concentric lamellar fibroplasia, naked hair shaft remnants or lymphohistiocytic infiltrates, either in the upper follicles or in the lower follicle/bulbs, were seen. No significant changes were seen in the epidermis, sweat glands, collagen bundles or elastic fibres. Only a slight perivascular superficial mononuclear cell infiltrate could be observed. The deposits of mucin were not increased. Infections by fungi or other microorganisms were ruled out by appropriate techniques including PAS and Grocott stains.
Control biopsies of the symmetrical contralateral areas were obtained in patients 3 and 5, and were normal.
Discussion
The aetiology of NH has not been established, but the assortment of symptoms and signs characteristic of this disorder suggests a peripheral origin of head pain (1–11). The trophic changes in these five patients enlarge the clinical spectrum of NH and strengthen its consideration as a peripheral headache. All of them presented with otherwise typical features of NH and none of them met clinical or histopathological criteria for any particular dermatological disease. Therefore, we were likely to be facing a NH with trophic changes instead of a dermatological disorder with pain. To our knowledge, only two other cases of head pain and colocalized trophic changes have been reported, and they were also atypical for any dermatological entity (12, 13). The spatial link between head pain and trophic signs was clear in all our patients, since the area of skin depression and alopecia was consistently located within the margins of the painful area. A temporal connection was also apparent in those who could recall the chronological sequence, since the trophic changes always followed the onset of pain, although the delay was variable in each of them. Moreover, in one of the patients both the head pain and the scalp abnormalities finally faded away. It is therefore conceivable that these clinical features reflect the same disorder, and that they are related by common pathogenic mechanisms.
We do not know for certain the source of NH, but the combination of confined pain and sensory disturbances in the same area is consistent with neuropathic pain (14). In addition, the associated trophic changes in these five patients strongly suggest a lesion or dysfunction of the peripheral nervous system. It is well known that, in certain instances, the normal innervation has a crucial role in tissue differentiation and in the subsequent maintenance of normal structure. In fact, alterations of the skin, the skin appendages and subcutaneous tissues are common in peripheral neuropathies (15). Specifically, NH with trophic changes might be considered a restricted form of complex regional pain syndrome (CRPS), which would probably be related to nerve injury.
Overall, CRPS consists of spontaneous or stimulus-evoked pain, autonomic abnormalities and trophic signs appearing in an anatomical region after a noxious event (16, 17). The intensity of symptoms and signs may be disproportionate to the precipitating event, and their location may be distant from the site of damage. According to the prevailing notion, CRPS is classified as type I (previously known as reflex sympathetic dystrophy), which is not directly attributed to a nerve lesion, and type II (previously known as causalgia), which is associated with nerve injury (18). However, both types of CRPS might be related to peripheral neuropathic effects (19, 20). The pathophysiological mechanisms have not been elucidated, but there is some evidence of neurogenic inflammation and autonomic dysfunction (21, 22). Even in CRPS II, symptoms and signs commonly spread beyond the innervation territory of the injured peripheral nerve and often occur remote from the site of injury. This expansion could be explained by a reorganization of sensory afferences within the central nervous system (22). However, restriction to the territory of a single peripheral nerve is consistent with the current definition of CRPS II. In fact, CRPS II can include some localized post-traumatic neuralgias (21).
There are suggestions that NH might be a localized neuralgia of pericranial nerves (1–4). If this were the case, NH with trophic changes could be considered a restricted form of CRPS II. This is only a hypothesis, since typical CRPS is not located in the head but in the extremities, and some common features of CRPS, such as burning or throbbing pain and the evidence of local oedema at some stage, were lacking in our patients. Nevertheless, neither the pathogenesis of CRPS nor the pathogenesis of NH has been clarified so far. The origin of NH could be a local lesion or dysfunction of the terminal branches of pericranial nerves, but it could also be placed at a distant and more centripetal level. In fact, anaesthesia of the symptomatic area in NH has not been effective (1, 6, 23). The concept of epicrania resolves this problem to some extent. Epicrania has been conceived as head pain stemming from epicranial tissues, i.e. the internal or external layers of the skull or any layer of the scalp, including the pericranial nerves (4, 11). NH could be provisionally considered an epicrania until its origin is finally revealed.
In conclusion, local trophic changes may be a clinical feature of NH. The pathogenic mechanisms are not yet known, but together with pain and sensory disturbances they could represent a restricted form of CRPS. In this context, CRPS should be taken as a possible evolution of the underlying morbid process of NH.
