Abstract
The objective of the study was to find out what kind of neck pain (NP) is associated with headache (HA) and with various headache variables: frequency, type, intensity, disturbance, and relief with analgesics. A population-based sample of 12-year-olds with and without HA (n = 304) was followed for 4 years. At the age of 16 years, NP was evaluated on the basis of self-reported symptoms and a thorough physical examination of the neck region. Both self-reported and measured NP were associated with HA variables. Co-occurrent NP was found in adolescents with migraine as often as in those with tension-type HA. Especially, muscle pain and intensive, frequent NP were associated with disturbing HA unresponsive to analgesics. The study indicates that concomitant NP should be considered in adolescent HA sufferers, and a thorough cervical and muscle evaluation is recommended when planning the treatment of HA.
Introduction
In adults, various features of neck pain (NP) such as self-reported NP, pericranial muscle tenderness, myofascial referred pain from the neck muscles, and the dysfunction of the joints of the upper cervical spine have been associated with headache (HA) (1–5). In adolescents, NP and HA are often concomitant (6–9), but few data are available on features of NP associated with HA (10–12). To our knowledge, no studies have been published to date on the associations of various self-reported and measured NP features with specific features of adolescent HA.
Adolescent HA is often episodic (13,14) but may be a ‘pre-stage’ of chronic HA in adults (15). It is therefore important to identify the treatable components of adolescent HA to arrest the pain sensitization process (16, 17). NP may be one of these treatable components contributing to central sensitization underlying frequent HA. Persistent unaddressed headache triggers, such as NP, may also limit the effectiveness of headache treatment (18).
The purpose of the present study was to find out what features of NP are associated with adolescent HA measured using seven HA outcome variables.
Methods
The present study is part of a population-based study of HA in school children. The original source population covered all 12-year-old school children in the city of Turku (total population 170 931) in south-western Finland. The details of the study design have been published previously (11, 13). A flow chart shows the participation rates of the follow-up studies (Fig. 1). In the present study, the cross-sectional results of 16-year-old subjects will be presented.
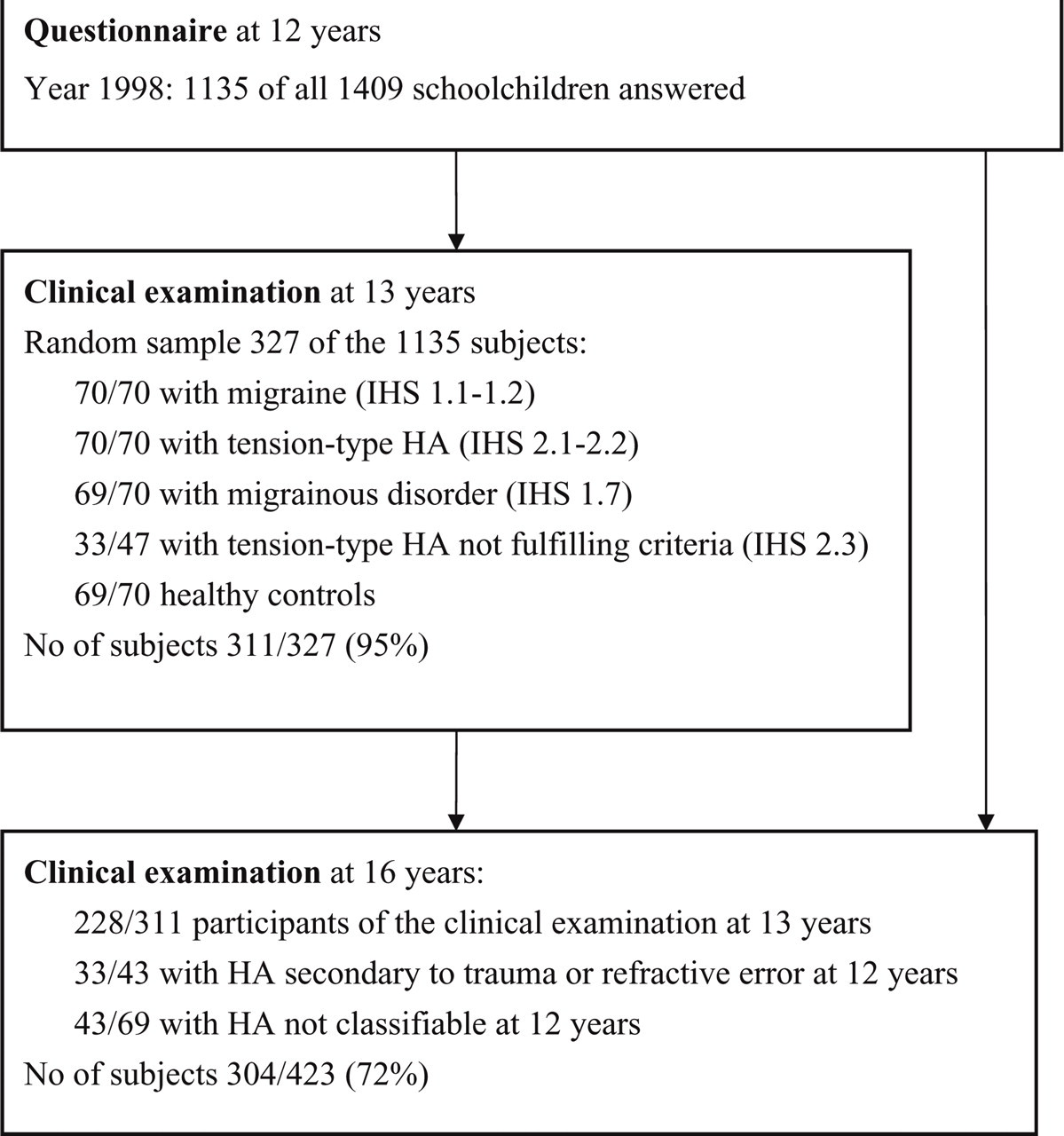
Flow chart of the study population.
Outcome variables
HA at the age of 16 years was described on the basis of seven outcome variables: (i) overall HA, (ii) HA type, (iii) HA frequency, (iv) intensity of HA measured using the visual analogue scale (VAS), (v) HA disturbing leisure time, (vi) HA disturbing school work, and (vii) HA relief with analgesics. Table 1 shows detailed definitions of the seven outcome variables. The classification of HA type was based on a structured interview, a neurological examination and the criteria of the Committee of the International Headache Society available at the time of data collection (19).
Detailed definitions of seven outcome headache variables

The HA type of subjects with two types of HA (n = 16) was classified according to the most frequent type of HA.
Neck pain variables
NP at the age of 16 years was described on the basis of: subjective, self-reported NP variables, (i) NP frequency, (ii) daily NP, (iii) VAS of NP, (iv) HA from the neck, and (v) current NP; and measured NP variables, (vi) general nuchal palpation pain (PP), (vii) PP of the sternocleidomastoid, (viii) PP of the face, (ix) PP of the masseter, (x) PP of the upper trapezius muscles, (xi) PP of the posterior neck, (xii) referred PP, (xiii) pain in the dolorimeter, (xiv) pain on neck motion, (xv) decrease in the range of motion (ROM) of the spine, (xvi) PP of the spine, and (xvii) dysfunction of the upper neck. Table 2 shows detailed definitions of predictive NP variables.
Detailed definitions of neck pain variables

PP = palpation pain.
The physical examination of the neck was done by a trained specialist in physical medicine and rehabilitation (K.L.) blinded to the subjects' pain history. The examination was performed in the sitting position with the low back against the backrest, forearms on hand rests. Only the palpation and the dolorimeter measurements of the posterior neck and upper trapezius muscles were done in the prone position to permit muscle relaxation. A structured manual palpation test was carried out bilaterally on tenderness and on referred pain of muscles receiving their sensory innervation from levels C1 to C3. The masseter, sternal division of the sternocleidomastoid and upper trapezius muscles were examined using pincer palpation, grasping the belly of the muscle between the thumb and the index finger, and other muscles using flat palpation with the tip of the index finger at a force of 1 kg. The force was calibrated by exercising with a pressure dolorimeter between each participant. In addition to local palpation pain, referred pain on muscle palpation to the head was recorded. This referred pain was regarded as possible myofascial pain of the head. To reach this diagnosis, the spot tenderness of trigger points (3) of the muscles was palpated and subjects were advised to report referred pain on palpation. For palpation, a muscle map from the trigger point manual (3) was used, and only muscles with a possible myofascial referred pattern to the head were palpated (muscles listed in Table 2).
Intraexaminer reliability
Before onset of the study, 15–16-year-old school children from two secondary school classes in Turku city were recruited to the reliability tests. Forty-seven children filled in a questionnaire twice and 37 children underwent a physical examination twice at 1 week's interval. Reproducibility between the first and the second examination was analysed using the proportion of agreement (PA) and intraclass correlation coefficient (ICC). Reliability was good for reported NP frequency (PA = 66%), reported VAS of NP (ICC = 0.85), referred PP (PA = 86%), motion pain (ICC = 0.67), PP of the spine (PA = 68%), and the sum of PP (ICC = 0.82). Pain in the dolorimeter (ICC = −0.01) was not repeatable after 1 week's interval.
Statistical analysis
The descriptive values were expressed as means and standard deviations (
The study design and the informed consent procedures were approved by the Joint Ethics Review Committee of the Medical Faculty of Turku University and the University Central Hospital of Turku.
Results
Table 3a shows the number of subjects with HA and NP, and the mean values of analysed variables at the age of 16 years. Seventy-five per cent of 247 HA sufferers and 35% of 57 HA-free subjects reported NP during the previous 6 months; 64% of HA sufferers and 30% of HA-free subjects had muscle palpation pain (PP); 7% of HA sufferers had PP referred to the head.
Univariate analysis of neck pain and overall headache, headache type and headache frequency

P-value < 0.05,
P-value < 0.01,
P-value < 0.001.
P-values evaluated by χ2 or Fisher's exact test between categorical, by the M-Whitney U-test between categorical and continuous variables and by Spearman correlation coefficients between continuous variables. Migraine = IHS 1, TTHA = tension type HA, IHS 2. VAS = visual analogue scale. HA from the neck = pain reported as starting from the motion or posture of the neck. Current NP = NP at the beginning of the physical examination. General nuchal PP = sum of PP of sternocleidomastoid, face (frontalis and temporalis), masseter, upper trapezius muscles and posterior neck (the semispinalis capitis, semispinalis cervicis, splenius capitis, splenius cervicis muscles and the suboccipitalis area at the point of possible entrapment of the greater occipital nerve). Referred PP = referred pain from neck to the head on muscle palpation. ROM = range of motion.
Univariate associations of neck pain with headache variables
Tables 3a and 3b shows the univariate associations of NP variables with HA variables. Of self-reported NP, the visual analogue scale (VAS) of NP and NP frequency were positively associated with all HA outcome variables except the HA type. Daily NP was associated with frequent and intensive HA, but not with variables describing disability. Current NP was associated with frequent HA and HA unresponsive to analgesics.
Univariate analysis of neck pain and intensity, disturbance of headache and headache relief with analgesics

P-value < 0.05,
P-value < 0.01,
P-value < 0.001.
P-values evaluated by the Mann–Whitney U-test between categorical and continuous variables and by the Spearman correlation coefficient between continuous variables. VAS = visual analogue scale. HA from the neck = pain reported as starting from the motion or posture of the neck. Current NP = NP at the beginning of the physical examination. General nuchal PP = sum of PP of the sternocleidomastoid, face (frontalis and temporalis), masseter, upper trapezius muscles and posterior neck (semispinalis capitis, semispinalis cervicis, splenius capitis, splenius cervicis muscles and the suboccipitalis area at the point of possible entrapment of the greater occipital nerve). Referred PP = pain referred from the neck to the head on muscle palpation. ROM = the range of motion.
Of measured NP, especially PP and referred PP were positively associated with HA variables. General nuchal PP was associated with frequent HA disturbing leisure time and HA unresponsive to analgesics. Of the separate muscle groups, PP of the sternocleidomastoid muscles was the only type of PP associated with intensive HA. PP of the sternocleidomastoid, masseter and posterior neck muscles was associated with HA disturbing leisure time or school work. PP of the sternocleidomastoid, masseter and upper trapezius muscles was associated with HA unresponsive to analgesics. Referred PP was associated with frequent HA and HA disturbing leisure time. On the other hand, the dysfunction of the upper neck, pain on motion or decrease in the ROM of the spine, pain measured by dolorimeter, and PP of the spine were not associated with frequent or disturbing HA.
Only a few differences were found between the genders in the association of significant predictive NP variables with the HA variables. Daily NP (P = 0.04) and PP of the masseter muscles (P = 0.004) were associated with overall HA only in boys. When migraine was compared with tension-type HA, spinal pain was associated with tension-type HA only in girls (P = 0.02). Frequent NP (P = 0.04), NP in daily activities (P = 0.01), current NP (P = 0.04), and PP of the sternocleidomastoid (P = 0.02) and upper trapezius muscles (P = 0.02) were associated with frequent HA only in girls. NP variables were associated with HA disturbing leisure time or school work in girls, but not in boys. On the other hand, PP of the sternocleidomastoid (P = 0.04) or masseter muscles (P = 0.01) was associated with HA unresponsive to analgesics only in boys.
Neck pain variables associated with headache variables in multivariate analysis
To find independent NP variables explaining HA outcome variables, the significant NP variables of the univariate analysis were entered into multivariate models (Table 4) except for pain in dolorimeter because of a small number of participants with dolorimeter measurements (n = 117/247). Daily NP was associated with frequent HA. Intensive NP was associated with frequent, intensive and disturbing HA. Of measured NP, spinal pain was associated with overall HA.
Multivariate logistic regression analysis and linear models of significant neck pain variables with headache

OR = odds ratio, 95% CI = 95% confidence interval, β = regression coefficient, SE = standard error of β.
P-value < 0.05,
P-value < 0.01,
P-value < 0.001.
1–3/month and >3/month were combined for logistic regression due to 0 cell frequency.
Difference between sometimes vs often.
VAS = visual analogue scale 0.0–10.0. HA from neck = pain reported as starting from the motion or posture of the neck, Current NP = NP at the beginning of the physical examination. Face muscles = the frontalis and temporalis muscles. Posterior neck = semispinalis capitis, semispinalis cervicis, splenius capitis, splenius cervicis muscles and the suboccipitalis area at the point of possible entrapment of the greater occipital nerve. Referred PP = pain referred from the neck to the head on muscle palpation. ROM = range of motion.
To study which were the most important measured NP variables explaining characteristics and consequences of HA, only significant measured NP variables from the physical examination of the neck were included in the second multivariate analysis. In addition to PP of the spine associating with overall HA, PP of the posterior neck muscles was independently associated with HA disturbing school work, PP of the sternocleidomastoid muscles with intensive HA, and referred PP with frequent HA. When current NP was included in the model, it had no influence on the results.
Discussion
As far as we know, this study is the first investigating associations of self-reported and measured neck pain (NP) with adolescent headache (HA). Both self-reported and measured NP variables were associated with HA, but not with HA type. HA with concomitant NP was more frequent and intensive, disturbing both leisure time and school work and unresponsive to analgesics when compared with HA without NP. Of measured NP, muscle pain was more clearly associated with disturbing HA than spinal pain.
NP is not a uniform symptom resulting from one source of pain with a single uniform aetiology (16,21). The association of NP and HA may also result from various pathophysiological mechanisms. The structures of the neck innervated by the first three cervical nerves can be associated with HA through convergence of nociceptive afferents and sensitization of trigeminocervical neurones (22–25). NP could initiate (4, 26) or maintain HA (27), or NP could be a consequence of HA extended from the head to other parts of the body. Central neuroplastic changes in HA could affect the regulation of peripheral mechanisms and lead to increased pericranial muscle activity (17, 22).
Self-reported neck pain
In this study, self-reported NP and measured NP at the level of cervical nerves C1 to C3 were examined. Of the reported NP variables, the intensity of NP by the visual analogue scale (VAS), NP frequency and daily NP were positively associated with the characteristics and consequences of HA. Subjects with current NP at the time of clinical examination reported more HA than those without current NP. The intensity of NP by VAS was the strongest independent factor explaining HA variables. In most headache studies, only the frequency, not intensity, of NP has been examined. The intensity of NP could be a more important indicator of the sensitization process of pain than the frequency of NP alone.
Measured neck pain
At the physical examination of the neck, especially muscle palpation pain (PP) and referred PP were positively associated with HA variables. To identify tools that would be useful to clinicians examining adolescent HA sufferers, we wanted to use the conventional palpation method instead of a pressure-controlled palpometer, although manual palpation has been criticized for being subject to bias (28). To optimize the reliability in our study, the force of manual palpation was calibrated between each participant. In our reliability study in another adolescent population with mainly HA-free subjects before the onset of the study, muscle PP and referred PP were reliable when recorded at 1 week's interval. In adults, the reliability of the physical examination is better in symptomatic subjects (29), and we assume that the reliability of PP in adolescent HA sufferers with more permanent neck symptoms is also good.
Pericranial muscle tenderness distinguished HA sufferers from HA-free subjects, as in previous studies of young women (5) and adults (2), and tenderness increased with increasing frequency of HA as in adults (2). In our previous study, measured muscle tenderness at the age of 13 years did not predict the outcome of HA on follow-up (14), and we suggested that muscle pain at 13 years of age was only a temporary phenomenon with no permanent structural changes in pericranial muscles. However, previous studies have shown that NP and muscle tenderness in 15–18-year-old adolescents predicts NP in adulthood (16, 30), and we presume that the muscle pain seen in our study group at the age of 16 years is also more permanent than it was at the age of 13 years.
In our study population of non-chronic HA sufferers, the tenderness of all neck muscle groups separately (the sternocleidomastoid, upper trapezius, masseter, posterior neck and face muscles) was associated with HA, as in a previous study of female students aged 17–26 years (5). If pericranial muscle tenderness was only a consequence of central neuroplastic changes in HA, it would probably not be associated with non-chronic adolescent HA, and this association of muscle tenderness with infrequent adolescent HA could be a sign of NP as one factor in the aetiology of recurrent HA (5). We speculate that muscle pain is likely to be one cause of disturbing adolescent HA, not only a consequence of a sensitization process, because the present study showed an association of muscle pain, especially PP of the masseter and posterior neck muscles, with disturbing HA, although this PP was not associated with frequent or intensive HA. Of the studied muscles, the influence of the sternocleidomastoid muscles on HA could result from the weakness of these neck flexors when compared with the spine extensors (31), increasing the imbalance of the neck. A sedentary way of life, on the other hand, could induce excessive loading of all studied neck muscles and irritate the muscle-nerve system of the neck with unfavourable positions, changes in posture and difficulties in relaxing muscles. Changes in posture have been seen in adult HA sufferers (32). In our study, muscle pain was associated with HA variables in both genders, but with disturbing HA only in girls and with HA unresponsive to analgesics only in boys. The found associations emphasize the importance of concomitant muscle pain in adolescent HA regardless of gender.
Myofascial pain is a common condition in adults referring pain to distant sites and causing intense and disabling HA (3, 4). Some studies define any soft tissue pain as myofascial pain, but in our study myofascial pain was diagnosed if the palpation of small hyperalgesic trigger points in the neck or face muscles referred pain to the head (3, 4, 33). In adults, especially the sternocleidomastoid and upper trapezius muscles are important in referring myofascial pain inducing HA (3, 4). It has been shown that the stimulation of myofascial trigger points can induce HA (34) and the inactivation of the trigger points can eliminate such headaches (35). In our study, myofascial referred PP was found in HA sufferers, indicating that myofascial pain may also contribute to HA in adolescence.
Although the joints of the upper cervical spine of adult patients have been shown to be a common origin of occipital headaches (1), neither the dysfunction of the upper neck in this study nor the disc degeneration of the spine (36) was associated with adolescent HA. It seems that in this age group the neck muscles play a more important role in HA than spinal changes. In healthy adults, the extreme position of the cervical spine has been shown to cause NP referring to the head (37). As the cervical spine of adolescents is more flexibile than that of adults (38), it could be thought that in extreme positions of the spine in sedentary hobbies the neck muscles in this age group are even more prone to become loaded than in adults.
Cervicogenic HA was not diagnosed in our study population, because this diagnosis requires abolition of HA following successful treatment of the causative disorder (23, 26), and thus the strict diagnostic criteria of IHS do not allow diagnosis of cervicogenic HA in a population-based study. However, as many as 38% of HA sufferers reported that their HA usually began from the posture or movement of the neck, which is one symptom included in the criteria of cervicogenic HA from 1988 (19). It is remarkable that even if this symptom was not associated with frequent or intensive HA, HA starting with the posture or movement of the neck disturbed leisure time and school work more often than HA not beginning from the neck. The cause of disturbing HA with start from the neck could be an unfavourable posture of the neck at school and in sedentary hobbies.
Headache features referring to concomitant neck pain
HA with concomitant NP was more frequent, more intensive, disturbing leisure time and school work, and unresponsive to analgesics when compared with HA without NP. As in our study, adult HA studies have shown that NP is more clearly associated with HA frequency than with HA type (5, 39). Although the classification of HA type is symptom-based and does not allow aetiological conclusions, pericranial tenderness and NP have traditionally been associated with tension-type HA (26, 27). In adolescent HA, muscular factors could, however, play a role in both migraine and tension-type HA. On the other hand, because the fluctuation of HA type is common in this age group (14, 40, 41), we assume that juvenile migraine and tension-type HA are not totally separate entities.
In our previous study, adolescents frequently using analgesics constituted a risk group for a poor outcome of HA (13). In the present study, concomitant NP was associated with HA disturbing leisure time and school work but also with HA unresponsive to analgesics. With the aim of developing the treatment of HA and preventing the sensitization process of pain, adequate treatment of concomitant NP could be important in adolescents with non-chronic HA (18).
As the proportion of chronic HA sufferers was small, our results cannot be directly applied to patients suffering from chronic HA. One simple aetiology and one simple pathophysiological mechanism cannot be expected to explain chronic HA. In studying episodic HA, finding a way to prevent the evolution of episodic HA to chronic HA is a major challenge (2, 17). Prolonged nociceptive stimuli from the neck muscles may have a role in the conversion of episodic HA into chronic HA (2, 17). As NP has been shown to be the most permanent pain syndrome in adolescence, with a high tendency to convert to widespread pain on follow-up (16, 42, 43), prevention of NP in this age group may have an impact on the reduction of pain and disability later on in life.
It is interesting that different NP variables are associated with different features of HA. More intensive NP is associated with HA disturbing school work, probably by causing concentration problems. Muscle pain, on the other hand, is more associated with HA disturbing leisure time, probably because of sedentary hobbies with unfavourable posture of the neck. NP variables associating with frequent or intensive HA can be a cause or a consequence of HA. Most interesting, however, are the NP variables associated only with disabling HA or with HA unresponsive to analgesics, but not associated with frequent, intensive HA. These NP variables (muscle pain and HA beginning from the neck) could represent a type of NP clearly causing HA. NP causing HA, on the other hand, probably requires other treatment than analgesics.
Tenderness in the neck muscles is common in HA patients (44), and muscular neck pain also seems to be clearly associated with disturbing HA in a non-chronic adolescent population. Concomitant NP should be considered in adolescent HA sufferers, and a thorough cervical and muscle evaluation is recommended when planning the treatment of HA.
Acknowledgements
This study was supported by grants from the Turku University Foundation, the City of Turku, and the University Hospital of Turku. Olli Kaleva, BSc, is acknowledged for skilful computation of the statistical analyses, and Mrs Inger Vaihinen for her valuable assistance.
