Abstract
Interictal evoked central nervous system responses are characterized in migraineurs by a deficit of habituation, at both cortical and subcortical levels. The click-evoked vestibulo-collic reflex (VCR) allows the assessment of otolith function and an oligosynaptic pathway linking receptors in the saccular macula to motoneurons of neck muscles. Three blocks of 75 averaged responses to monaural 95-dB normal hearing level 3-Hz clicks were recorded over the contracted ipsilateral sternocleidomastoid muscle in 25 migraineurs between attacks and 20 healthy subjects, without vestibular symptoms. Amplitudes, raw and corrected for baseline electromyography, were significantly smaller in migraine patients. Whereas in healthy volunteers the VCR habituated during stimulus repetition (-4.96% ± 14.3), potentiation was found in migraineurs (4.34% ± 15.3; P = 0.04). The combination with a reduced mean amplitude does not favour vestibular hyperexcitability as an explanation for the habituation deficit in migraine, but rather an abnormal processing of repeated stimuli in the reflex circuit.
Introduction
Neurophysiological studies performed in migraine patients between attacks have repeatedly demonstrated a lack of the physiological habituation, or even potentiation, in evoked cortical potentials during uninterrupted repetitive stimulation (1). In a few studies, such a habituation deficit has also been found in brain stem-generated activities such as subcortical components of somatosensory evoked potentials (2) and the nociceptive blink reflex (3, 4). The precise cause of the habituation deficit is not known. The fact that the amplitude of evoked responses is usually low in migraine after low numbers of stimulations suggests that it could be due to a decreased preactivation level in the stimulated neuronal circuits (1), but hypoactivity of inhibitory interneurons cannot be excluded (5).
Vertigo and dizziness are frequent symptoms in migraine (6) and abnormal vestibular laboratory tests have been reported in many migraineurs complaining of dizziness (7). However, even in the absence of vestibular symptoms, neurotological tests (8, 9) and optoelectronic 3D movement analysis (10) may be abnormal interictally, suggesting subclinical vestibular and cerebellar dysfunction in migraine.
The vestibulo-collic reflex (VCR) consists of myogenic responses evoked in the tonically contracted ipsilateral sternocleidomastoid muscle by brief monaural clicks (11). The VCR, also called vestibular evoked myogenic potential (VEMP), permits the assessment of a rapidly conducting oligosynaptic brainstem pathway between vestibular nuclei and motor neurons of the accessory nerve nucleus. It originates in the otolith organs, in particular the saccule, as suggested by studies in patients with sensorineural hearing loss or selective vestibular nerve section (12, 13). It has been found to be abnormal in various vestibular disorders such as Ménière's disease, acoustic neuroma, vestibular neuritis and multiple sclerosis (14–18). In a recent study, Liao and Young (19) found delayed or absent VCR responses in 10 out of 20 patients with basilar-type migraine.
Against this background, it is of interest to study the VCR, in particular its amplitude changes over sequential averagings, in patients suffering from the common forms of migraine with or without aura between attacks. Because of our previous experience showing that neurophysiological tests are able to unravel subclinical dysfunctions in migraine (10, 20), it was decided to focuse first on patients without vestibular symptoms or complaints.
Methods
Twenty-five out-patients suffering from migraine were enrolled from our headache clinic (eight male and 17 female, mean age 32.5 ± 9.7 years). Based on the criteria of the International Classification of Headache Disorders, 2nd edition (ICHD-II 2004) (21), 15 patients had exclusively migraine without aura (MoA) (code 1.1; mean duration of disease 10.37 ± 9.17 years, mean attack frequency 3.47 ± 1.7/month) and 10 migraine with typical aura (MA) (code 1.2.1; mean duration of disease 18.6 ± 13.12 years, mean attack frequency 1.02 ± 0.7/month). All had been without preventive antimigraine therapy for at least 3 months and recorded interictally at least 3 days after the last and before the next migraine attack.
A control group of 20 healthy volunteers of comparable age and gender distribution (nine male and 11 female, mean age 30.4 ± 8.8 years) without personal or family history of migraine or other recurrent primary headaches was recruited from the hospital staff and students.
None of the subjects had hearing loss or recurrent vertigo, or a history of vestibular disease.
Written informed consent in accordance with the Declaration of Helsinki 1964 and its 1975 Tokyo amendment was obtained from all subjects, and the study was approved by the local Ethics Committee of the Faculty of Medicine, University of Liège, Belgium.
The click-evoked VCR was elicited according to the method described by Colebatch et al. (12). Subjects were seated in an armchair with adjustable backrest in a semireclining position at an angle of 45° to the horizontal plane. They were asked to turn their head to one side by about 80° in order to activate the contralateral sternocleidomastoid muscle (SCM). Using CEDTM 1902 preamplifiers (Cambridge Electronic Design, Cambridge, UK), surface EMG activity was recorded from electrodes placed over the upper half of each SCM, with a reference electrode on the medial portion of the clavicle and a ground electrode over the sternum. To ensure regular muscle activation, subjects received visual feedback via an oscilloscope screen which was placed in front of them and displayed the EMG. They were instructed to maintain during the whole recording session a constant muscle activity of 30–40 µV, which is considered to be the optimal target EMG level to elicit the VCR (22).
The acoustic stimulus was a brief (0.1 ms) click presented to the ear ipsilateral to the contracted SCM muscle and delivered via earphones at a repetition rate of three per second. A fixed click intensity of 95 dB normal hearing level was used in all subjects. Rectified and unrectified EMG activity was collected from 50 ms before to 50 ms after the stimulus and off-line filtered (2–1.5 kHz). Using a CEDTM 1401 signal averager and the SignalTM software package version 2.15 (Cambridge Electronic Design), a total of 225 responses were averaged in three sequential blocks of 75 stimuli (23).
The first positive (p13) and the first negative peak (n23) of the unrectified VCR were measured in terms of latency and peak-to-peak amplitude ipsilateral to the stimulated ear. Only the first p13-n23 component was considered, which depends specifically on the integrity of vestibular afferents and not the later components (n34, p44) because they are inconsistent and probably of cochlear origin (13). Because the amplitude of the averaged responses increases in direct proportion to the mean level of tonic muscle contraction (24), differences in activation between subjects were corrected for by dividing the raw peak-to-peak amplitude by the mean rectified EMG activity during the 20 ms prior to the stimulus (‘corrected amplitude’). In this study, both raw and corrected VCR amplitudes are reported.
Habituation of the response was expressed as the percentage change of corrected and raw amplitudes between the first and third block of averagings.
VCR measurements were made off-line by one investigator (M.A.) blinded to diagnosis.
Measured variables were expressed as means ± SD. In order to ascertain if a given variable had a normal distribution, the Kolmorov–Smirnoff test was used. For comparison between groups, data with a normal distribution were analysed with an unpaired t-test. Right and left side values were compared with the paired t-test. Pearson's test was used to search for a correlation between amplitude of the first VCR block and habituation over three blocks. Results were considered significant at P < 0.05. Statistical calculations were carried out using version 3.00 of the GraphPad InStat program for Windows (GraphPad Software, San Diego, CA, USA).
Results
The VCR was successfully recorded in all subjects as a biphasic, initially positive, p13-n23 waveform (Fig. 1). We first searched for a difference in VCR latency, amplitude and habituation between right and left sides. Since there were no significant side differences, the mean value calculated from both sides of each subject was used for further analysis. The results are summarized in Table 1, with the levels of significance found for the various measures taken in healthy volunteers and in migraine patients interictally.
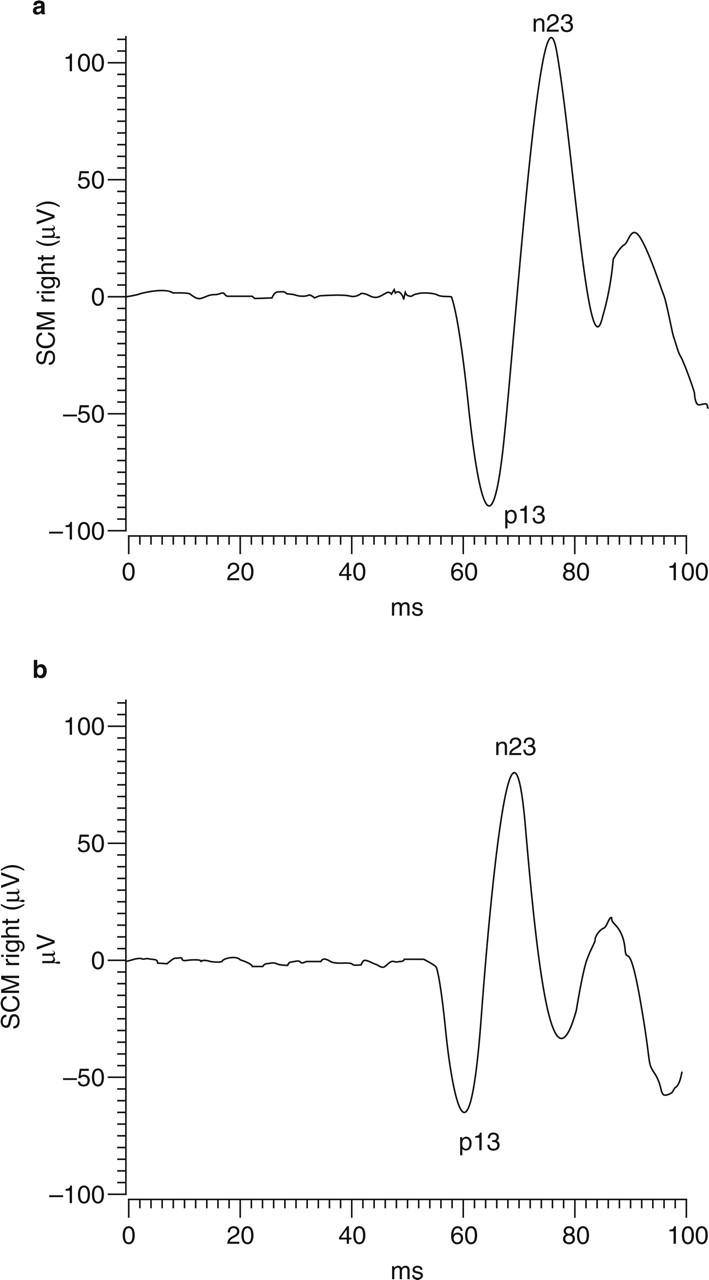
Illustrative non-rectified traces of the vestibulo-collic reflex obtained in right sternocleidomastoid muscle (SCM) after ispilateral monaural clicks in a healthy subject (a) and a migraine without aura patient (b). The acoustic stimulus starts at 50 ms.
Synopsis of vestibulo-collic reflex measures (mean ± SD) in migraineurs and healthy subjects.
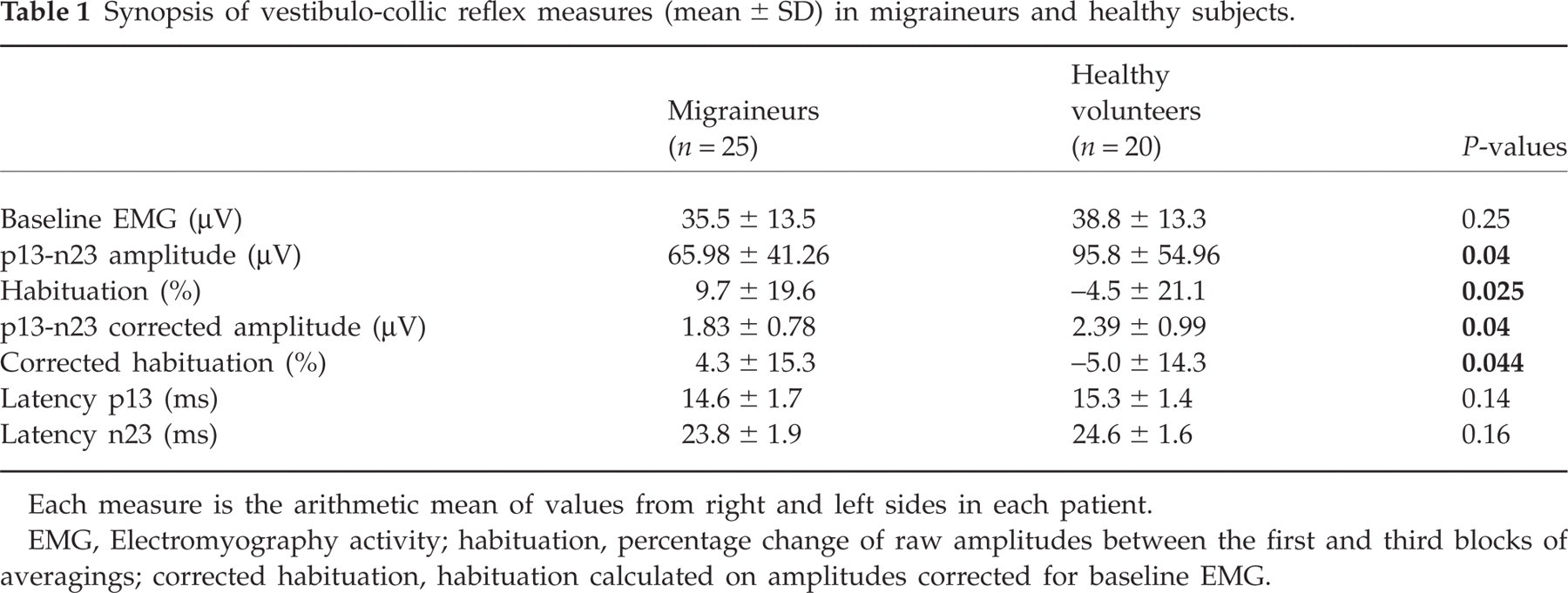
Each measure is the arithmetic mean of values from right and left sides in each patient.
EMG, Electromyography activity; habituation, percentage change of raw amplitudes between the first and third blocks of averagings; corrected habituation, habituation calculated on amplitudes corrected for baseline EMG.
During baseline voluntary contraction, the mean rectified EMG activity for left and right SCM was not significantly different between migraineurs and healthy subjects. Both p13 and n23 VCR latencies were slightly shorter in patients than in controls, but the difference did not reach the level of statistical significance.
By contrast, mean raw, unrectified VCR amplitude was on average significantly smaller in migraineurs (F(2,43) = 1.775, P = 0.04) than in healthy volunteers (Fig. 2).

Vestibulo-collic reflex raw amplitudes (mean ± SEM) in three sequential blocks of 75 averaged responses in migraine patients (MIG, n = 25) and healthy volunteers (HV, n = 20). ▪, First block;  , second block; □, third block.
, second block; □, third block.
Habituation calculated on the uncorrected VCR amplitude between the first and the third block of 75 stimuli was significantly replaced in migraineurs by potentiation (F(2,43) = 1.157, P = 0.02).
After correction of the VCR for interindividual differences in SCM contraction, healthy subjects still had on average a significantly greater VCR amplitude (F(2,43) = 1.61, P = 0.04) and habituation (F(2,43) = 1.145, P = 0.004).
Raw and corrected VCR first block amplitudes were also compared between groups. Both were smaller in migraineurs than in healthy volunteers (F(2,43) = 1.982, P = 0.05 for raw amplitudes and F(2,43) = 1.246, P = 0.05 for corrected amplitudes).
There was no significant correlation between first block amplitude and habituation and no significant differences were found between MA and MoA.
Discussion
The click-evoked VCR or VEMP has been used in clinical practice for a variety of peripheral and central vestibulopathies (14–18), but not for migraine. As mentioned, its short-onset latency suggests that it is mediated by an oligosynaptic pathway, which probably includes the saccular macula, the inferior vestibular nerve, the lateral (Deiter's) vestibular nucleus in the medulla, the vestibulospinal tract and finally the neck motor neurons innervating the ipsilateral SCM (11, 12). This three-neuron arc which provides a fast link between vestibular receptors and neck muscle has been well studied with electrophysiological methods in the cat (25–27). Electrical stimulation of the saccular nerve evokes chiefly excitatory postsynaptic potentials in the vestibular nuclei, but inhibitory postsynaptic potentials in ispilateral SCM motor neurons, whereas in contralateral SCM motor neurons the stimulation has almost no effect (28). The authors concluded that the saccular–sternocleidomastoid reflex pathway is mainly associated with ipsilateral disynaptic inhibition. Concordantly, Rothwell and Colebatch (24, 29) found in single motor unit EMG recordings of the SCM in humans that intense clicks are followed ipsilaterally by a brief period of inhibition that coincides with the surface positivity of the p13-n23 response. A similar pattern has been found in the vestibulo-masseteric and the trigeminocervical reflexes where, respectively, the p11-n25 and the p19-n31 components correspond to a short silent period in motor unit discharges (30–32). Interestingly, the trigeminocervical reflex is also modified in headache patients (33).
Our study is the first to analyse the habituation pattern of the VCR in healthy subjects and migraine patients. Habituation, which is on average moderate, but clear-cut, in normal volunteers, is replaced in migraineurs by potentiation. This pattern is similar to that described in migraine between attacks for cortical-evoked responses (1). However, a habituation deficit has also been reported for subcortical components of evoked potentials (2) and, more recently, for a brainstem reflex, the nociceptive blink reflex (3, 34). We have in fact shown in a previous study that habituations of the latter reflex and of visual evoked potentials are correlated intraindividually (4).
The present report is, however, the first to show a habituation deficit of an inhibitory brainstem reflex in migraine. The VCR has been reported abnormal in 10 out of 20 patients with basilar-type migraine (19). The abnormalities were described as delayed or absent responses and disappeared in nine out of the 10 patients after preventive antimigraine treatment. In the present study, the VCR was identified in all patients and there was, in contrast, a tendency for a shortening of p13 and n23 latencies. This difference is probably due to the different patient populations. We studied patients suffering from MoA or MA, i.e. the most prevalent forms of migraine, which may be pathophysiologically (20) and, most likely, genetically different (35) from basilar-type migraine.
Moreover, because our patients had no vestibular symptoms, they did not have electronystagmography (ENG). Mild subclinical abnormalities may occur in migraineurs, but this is the case in only a minority of patients. It was therefore thought most unlikely that subclinical ENG abnormalities could account for the VCR changes found in our study.
Vascular factors are unlikely to explain our findings of reduced VCR amplitude and habituation in migraineurs, because all recordings were performed interictally and patients were selected without a previous history of vertigo, dizziness or imbalance.
One can thus only speculate on other possible underlying mechanisms. Although it has been shown in the cat that the VCR is controlled by descending cortical projection on the vestibular nuclei (36), the abnormal excitability of the cerebral cortex that characterizes migraine between attacks can hardly explain the VCR abnormalities found here. Indeed, cortical control of the VCR remains to be demonstrated in humans. Similarly, there is no evidence that the subcortical abnormalities found in migraine could be induced by a cortical dysfunction. It has been hypothesized by our group (37) and others (38) that the habituation deficit of cortical evoked responses might be due to hypofunctioning subcortical serotonergic projections to the cortex, and an ensuing decrease in preactivation levels (1). Both ascending and descending projections originate in serotonergic raphe nuclei. In particular, there are reciprocal connections between vestibular nuclei, including Deiter's nucleus which is involved in the VCR, and neurons in the dorsal raphe nuclei (39, 40). The serotonergic input to the vestibular nuclei has been emphasized in pathogenic models of migrainous vertigo (7). We propose therefore that reduced activation by serotonergic afferents of the reflex arc underlying the VCR could be responsible both for its reduced amplitude and for deficient habituation in migraineurs.
Vestibular tests may be abnormal in migraineurs complaining of vertigo or dizziness (7). For some tests, including the vestibulo-ocular reflex, this can also be the case in patients without vestibular symptoms (9). We have thus begun to record the VCR in migraineurs with vertigo/dizziness, although the interictal abnormalities found in the present study may not necessarily be more prevalent in this subgroup of patients, for several reasons. First, it seems unlikely that decreased VCR amplitude and habituation are sufficient to produce vestibular symptoms. Second, we have shown that cortical evoked potentials, also characterized by low amplitudes and reduced habituation, normalize during the attack, i.e. when migraine becomes symptomatic (41). This change has been attributed to an ictal increase in brain serotonin (37). A recent study in mice has shown that serotonin is able to produce plasma extravasation in the inner ear, including in the intralabyrinthine vestibular nerves (42). If plasma extravasation is part of migraine pathophysiology, as has been suggested, one may expect that the VCR would undergo attack- or vertigo-related changes other than those reported in our study, but this remains to be determined in a proper clinical study.
To conclude, the VCR, an inhibitory brainstem reflex, is characterized by low amplitude and lack of habituation interictally in migraineurs without vestibular symptoms. These changes are similar to those reported for cortical evoked responses and the nociceptive blink reflex. We postulate that they are due to reduced serotonergic control of the reflex circuit, in particular of the vestibular nuclei, and not necessarily to a dysfunction predisposing to vestibular symptoms in migraine. This hypothesis will be examined in a study of patients with migrainous vertigo/dizziness.
Finally, although auditory brain stem evoked potentials are overall normal in migraine, as far as cortical auditory potentials are concerned, we have reported interictal changes in habituation and intensity dependence, as well as amplitude changes in auditory P3. A future study comparing various potentials would be of interest.
Acknowledgements
This study was in part supported by research grant 3.4.563.04 of the National Fund for Scientific Research (Belgium).
