Abstract
Interictally, migraineurs have on average a reduction in habituation of pattern-reversal visual evoked potentials (PR-VEP) and in mitochondrial energy reserve. 5,10-Methylenetetrahydrofolate reductase (MTHFR) is involved in folate metabolism and its C677T polymorphism may be more prevalent in migraine. The aim of this study was to search in migraineurs for a correlation between the MTHFR C677T polymorphism and the PR-VEP profile. PR-VEP were recorded in 52 genotyped migraine patients: 40 female, 24 without (MoA), 28 with aura (MA). Among them 21 had a normal genotype (CC), 18 were heterozygous (CT) and 13 homozygous (TT) for the MTHFR C677T polymorphism. Mean PR-VEP N1-P1 amplitude was significantly lower in CT compared with CC, and tended to be lower in TT with increasing age. The habituation deficit was significantly greater in CC compared with TT subjects. The correlation between the cortical preactivation level, as reflected by the VEP amplitude in the first block of averages, and habituation was stronger in CC than in CT or TT.
The MTHFR C677T polymorphism could thus have an ambiguous role in migraine. On one hand, the better VEP habituation which is associated with its homozygosity, and possibly mediated by homocysteine derivatives increasing serotoninergic transmission, may protect the brain against overstimulation. On the other hand, MTHFR C677T homozygosity is linked to a reduction of grand average VEP amplitude with illness duration, which has been attributed to brain damage.
Keywords
Introduction
During the last decade a number of single nucleotide polymorphisms have been found more prevalent in the common forms of migraine with or without aura (1). These polymorphisms concern a variety of genes coding for proteins involved in neurotransmission, blood pressure control or vasoregulation, inflammation or glucose metabolism. This genetic heterogeneity, further illustrated by linkage to more than a dozen chromosomal loci, contrasts with the relatively homogeneous clinical pattern of migraine such as defined by the International Headache Society classification (2). An obvious aim of migraine research is thus to analyse genotype–phenotype correlations in more detail. The phenotype can be characterized by the relative weight of clinical features, e.g. by using latent class analysis (3). However, peculiar electrophysiological patterns, mainly involving cortical evoked potentials, have also been identified in migraineurs (4) and, as in healthy subjects (5), they are likely to be genetically determined (6–8).
Several studies have suggested that the C677T polymorphism of the 5,10-methylenetetrahydrofolate reductase (MTHFR) gene on chromosome 1p36.3 is more prevalent in migraine, chiefly in migraine with aura (MA) (9–13). However, the mechanism underlying the relationship between the C677T variant and MA remains unknown. The MTHFR enzyme catalyses the conversion of 5,10-methylenetetrahydrofolate (CH2THF) to 5-methyltetrahydrofolate (CH3THF), a cosubstrate for homocysteine remethylation to methionine. It plays a pivotal role in the folate and homocysteine metabolisms. MTHFR competes with thymidylate synthase (TS) and methenyltetrahydrofolate cyclohydrolase formyltetrahydrofolate synthase (MTHFD1) for the substrate CH2THF. The C677T mutation involves the coding region of MTHFR gene, leading to the substitution of valine by alanine. It is frequent in Whites (5–16% of TT homozygosity) and associated with decreased activity of the enzyme due to thermolability, which can lead to mild hyperhomocysteinaemia in some, but not all homozygous patients (14). The impact of the C677T mutation on blood homocysteine varies with endogenous factors, such as age or gender, and with environmental conditions, such as high folate, B12 (14) or riboflavin (15) intake. Impaired function of MTHFR is also responsible for a higher substrate availability, i.e. CH2THF, and may lead to enhanced purine and pyrimidine synthesis through the concurrent pathways of TS and MTHFD1 (16). Functional polymorphisms affecting the genes coding for the latter enzymes were found to modulate the risk for migraine aura conferred by the MTHFR 677T allele (17).
The precise pathophysiological role of homocysteine in migraine patients with the C677T polymorphism remains unclear (13, 18, 19). In an animal model of craniovascular pain, DL-homocysteic acid, an oxidized derivative of homocysteine mimicking its effects on arteries, increased the firing of trigeminal nociceptors (20), suggesting that hyperhomocysteinaemia might favour sensitization in the trigeminovascular system. Riboflavin (vitamin B2), whose efficacy in migraine prophylaxis is thought to be mediated by an effect on mitochondrial metabolism (21), is also able to reduce homocysteine levels, particularly in subjects with the MTHFR C677T mutation (15). A high homocysteine plasma level is considered to be a risk factor for cardiovascular disease through its deleterious action on endothelial function, coagulation and oxidative reactions (14). The high prevalence of the C677T variant has been proposed as an explanation for the increased stroke risk in migraine (10). However, a recent large study has suggested that the effect of C677T on MA is only partially related to increased homocysteine levels (13).
Interictally, migraineurs have on average a reduction in habituation of evoked cortical potentials such as pattern-reversal visual evoked potentials (PR-VEP) (4). This habituation deficit has a strong familial influence in migraineurs, which suggests that it is genetically determined (6). Its underlying mechanisms are incompletely understood, but they may involve a lower cortical preactivation level due to reduced activity in raphe-cortical serotoninergic pathways (4). This hypothesis is supported by the finding of a high-intensity dependence of middle latency auditory evoked cortical potentials in migraineurs (22). Interestingly, DL-homocysteic acid, a metabolite of homocysteine (see above), is able to increase the activation of serotoninergic raphe neurons leading to synaptic facilitation in the rat hippocampus (23).
In light of the above-mentioned data and hypothesis, we searched for a possible correlation between the interictal features of PR-VEP and the MTHFR C677T polymorphism in a clinical population of migraineurs.
Materials and methods
Subjects
We studied 52 consecutive eligible patients attending our Headache Clinic (40 female, mean age 32.36 ± 14.42 years). Based on International Classification of Headache Disorders II criteria (2), 24 patients had migraine without aura (MoA) and 28 MA (mean attack frequency per month 3.49 ± 2.65). They gave written informed consent, underwent the visual evoked potential recording and thereafter blood sampling for genetic analysis. The study was approved by the Ethics Committee of the Faculty of Medicine, Liège University.
Genotyping
The search for the MTHFR C677T polymorphism was performed in the Molecular Biology Department of our hospital according to the method described by Dubreuil et al. for the prothrombin gene 20210 mutation (24).
After genotyping, patients were separated into three subgroups according to the MTHFR C677T polymorphism: no mutation (CC, N = 21), heterozygous (CT, N = 18) and homozygous (TT, N = 13).
Visual evoked potentials
The PR-VEP were recorded and analysed as previously described (25), with the investigator blinded to the patient's genotype. The patients had to have been without antimigraine prophylaxis since at least 3 months and to have normal visual acuity. Only recordings made at least 72 h after or before an attack were taken into consideration. Briefly, subjects sat in a comfortable armchair in a room with dimmed light at a 1-m distance from the monitor. They were asked to relax and to fix a red sticker in the centre of the screen (Nicolet™; 24 × 18 cm) with their right eye, the left eye being covered by a patch. The visual stimulus was a checkerboard pattern of black and white squares (15 mm side, 80% contrast) subtending 51 min and 33 s of arc and alternating at a frequency of 3.1 Hz. Pin-electrodes were used to record the signal: the active electrode was inserted at Oz and was referenced to Fz according to the 10–20 system. The ground electrode was fixed to the right forearm. During uninterrupted stimulation, 600 sweeps were recorded (CED™ 1902 preamplifier and CED™ Micro1401 converter; Cambridge Electronic Design Ltd, Cambridge, UK), which were averaged off-line into six blocks of 100 responses using Signal™ software version 2.15 (Cambridge Electronic Design Ltd). Two hundred milliseconds of the poststimulus period were sampled at a rate of 4000 Hz. The peak-to-peak N1–P1 amplitude was measured, N1 being the most negative point around 70 ms latency (range 60–90), P1 the most positive around 100 ms latency (range 90–120). Habituation was defined as the percentage change of the N1–P1 amplitude between the 1st and 6th blocks of averaged responses.
Statistical analysis
We chose to compare the three subgroups (CC, CT and TT) in the main analysis. Since the metabolic effects of the 677T mutation are thought to vary with age, especially in women (see above), a subanalysis according to age category was performed for amplitude. The influence of the presence of at least one T allele was also studied.
Group differences were evaluated by analysis of variance (
Results
Means and standard deviations of all data are shown in Table 1. In all recordings the N1–P1 component of the PR-VEP was clearly identified and no group differences were detected with regard to its latency.
Main demographic variables

Means and standard deviations of major measures.
Demographic variables
TT subjects were on average significantly older than CC patients (P = 0.004). Mean disease duration was significantly longer in CT (P = 0.04) and TT (P = 0.001) than in CC. As regards attack frequency, there was no difference between CC and CT (P = 0.86), or between CC and TT (P = 0.43) or CT and TT (P = 0.34).
VEP amplitude
The grand average VEP amplitude was 7.10 ± 3.00 µV in CC, 4.93 ± 1.89 µV in CT and 6.04 ± 2.25 µV in TT.

Mean visual evoked potential N1–P1 amplitude in the six successive blocks (B1 to B6) of 100 averaged sweeps. Vertical gratings represent standard errors. □, Subjects without mutation (CC, N = 21); ▪, heterozygous patients (CT, N = 18); ○, homozygous subjects (TT, N = 13).

Grand average (600 sweeps) of a visual evoked potential recording in a CC subject (black) and a heterozygous CT subject (grey), matched for age.
There was overall a strong inverse correlation between VEP amplitude and age (r = –0.44; P = 0.001), and disease duration (r = –0.3; P = 0.03). Subanalysis revealed that this was due to a significant inverse correlation in the TT group (r = –0.6; P = 0.03 for both variables), but not in the other groups. No correlation was found between amplitude and attack frequency (r = 0.001).
Because of the age difference between the three phenotypes and the age-dependent metabolic effects of the 677T mutation, the younger and older subjects in each subgroup were subanalysed, taking 35 years of age as a cut-off.
In the ‘younger’ group (N = 16 for CC; 11 for CT and 6 for TT), there was still a clear trend for a lower grand average VEP amplitude in CT compared with CC (P = 0.08), but amplitudes no longer differed between CC and TT (P = 0.82) and in TT the amplitude even tended to be greater than in CT (P = 0.07). In the ‘older’ group of patients (N = 5 for CC; N = 7 for CT; N = 7 for TT), amplitudes tended to be smaller in CT (P = 0.15) and TT (P = 0.21) than in CC. Within-group analysis showed that the difference of amplitude between age categories was significant for the TT (P = 0.006), but not for the CC (P = 0.37) or CT subgroups (P = 0.28).
As expected, the results were similar when a subanalysis was done for disease duration (data not shown).
VEP habituation
There was a trend for a mutation effect on VEP habituation (P = 0.1), which in fact was in favour of a stronger habituation in subjects with the C677T polymorphism. The planned comparisons revealed that the habituation deficit was significantly more marked in CC (−7.47%, i.e. potentiation) compared with TT (8.73%, P = 0.03). In CT (−3.18%), the habituation deficit was less pronounced than in CC, but not significantly so (Fig. 3). When all patients were considered together, a significant correlation was found between the first block amplitude and habituation (r = 0.28, P = 0.04), i.e. the subjects with the lowest amplitude had the greatest habituation deficit. When each genotype was analysed separately, the correlation was not significant, but seemed stronger in CC (P = 0.09) than in CT (P = 0.14) or TT (P = 0.65, Fig. 4).

Mean visual evoked potential habituation, i.e. percentage of change in amplitude between block 1 and block 6, in the three genetic subgroups (negative habituation means potentiation). Vertical gratings are standard errors.
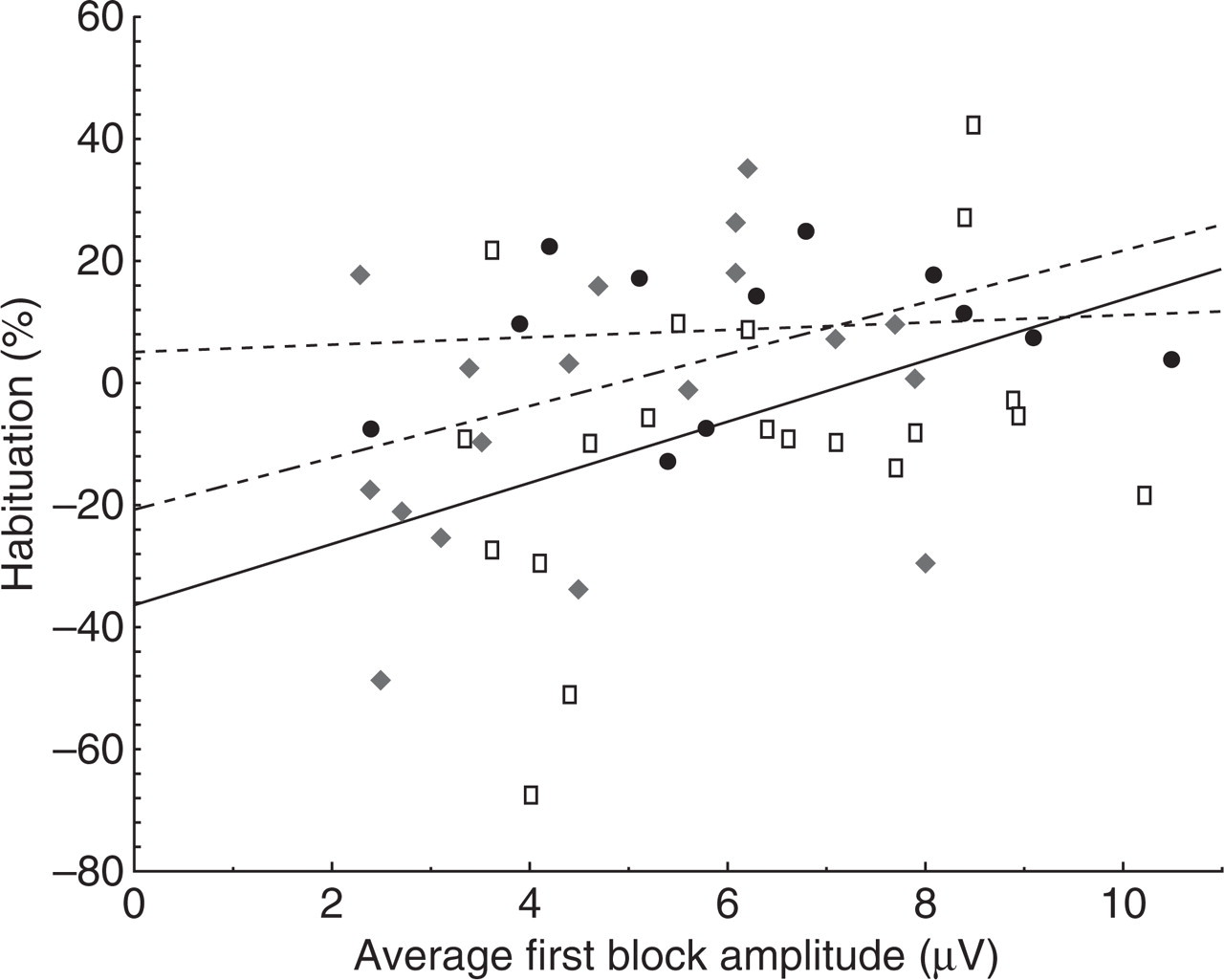
Habituation of visual evoked potential (%) according to first block amplitude. Correlation is stronger for CC subjects (black continuous line) than for CT (black dashed line), and particularly for TT subjects (black dotted line).
Influence of migraine type
VEP amplitudes and habituation did not differ between migraineurs with or without aura when their genotype was not taken into account (P = 0.85; P = 0.14). The low amplitude observed in CT was slightly more pronounced in subjects suffering from MA [mean (± SD) first block amplitude 4.75 ± 2.29 µV] than in those with MoA (mean amplitude 5.09 ± 1.71 µV), but this did not reach the level of statistical significance (P = 0.73).
Influence of the T allele
As expected from the previous results, grand average VEP amplitude was significantly lower in subjects who had a T allele compared with those who had not (P = 0.02), and VEP habituation tended to be more pronounced in the former (P = 0.1).
Discussion
To the best of our knowledge, this is the first study to search for a correlation between VEP features and the MTHFR C677T polymorphism. As the latter is more prevalent in migraineurs (9–11), chiefly with aura (9–13), in whom VEP abnormalities have been described (4), our study was conducted with a pathophysiological perspective and limited to patients. Differential results were obtained for VEP amplitude and habituation, which will therefore be discussed in sequence.
The presence of the 677T allele is significantly associated with a lower VEP amplitude both for the total of 600 averaged responses and for each of the six blocks of 100 averagings. Low VEP amplitude is not a known feature in migraineurs between attacks (see for a review (4)). In some early studies, especially those using flash stimulation, VEP amplitudes have rather been reported as greater in migraineurs than in healthy volunteers. By contrast, amplitude of the first block of averaged responses is commonly low in migraine (4, 26). The decrease in amplitude in the presence of the T allele could be due to increased homocysteine levels. A number of studies have suggested that hyperhomocysteinaemia can cause or promote central nervous system dysfunction. The mechanisms are heterogeneous and most remain to be proven. They involve direct neurotoxicity (excitotoxicity), decreased resistance to toxic insults, inhibition of methylation reactions and occlusive vascular disease (27). Hyperhomocysteinaemia could be neurotoxic because of its oxidized derivatives (28). DL-homocysteic acid, for example, can act as a glutamate analogue and bind to N-methyl d-aspartate receptors (29). Of direct interest here is the study in rat showing that, after topical application, DL-homocysteic acid is able to induce a depression of somatosensory evoked potentials (30). There are no data available on the potential effects of homocysteine on evoked potentials in humans. However, Poloschek et al. (31) have reported a reduction of flash-evoked VEP amplitude in a patient suffering from methionine synthase deficiency, another genetic cause of elevated plasma homocysteine levels. A possible cytotoxic effect of homocysteine might thus lead to mild subclinical lesions in visual pathways. Because of the methodological constraints necessary to obtain reliable measurements of plasma homocysteine levels (32) and because baseline levels of homocysteine may not reflect long-term changes in central DL-homocysteic acid and thus serotoninergic transmission, it was decided not to determine plasma homocysteine levels in our study. In future studies of MTHFR polymorphisms, however, such levels, and especially their change after an oral methionine load, would be of interest.
If the neurotoxic hypothesis is correct, it may seem surprising that the effect on VEP amplitude is more pronounced in the heterozygous CT than in the homozygous TT subgroup. In the latter the reduction in amplitude appears chiefly with advancing age. As mentioned in the Introduction, the metabolic effects of the MTHFR C677T vary with dietary factors and the presence of polymorphisms in genes coding for related enzymes in the folate metabolism. None of these was assessed in our study. It is known, however, that the mutation effect can be counteracted by high folate and vitamin B12 intake. The TT variant could in fact confer a higher responsiveness to folate and B12. This is suggested by an inverse relationship between homocysteine and plasma folate/B12 levels in such patients (33) and by the fact that the plasma homocysteine-lowering effect of supplementation with 5-methyltetrahydrofolate or folic acid is greater in TT subjects (34). This could be an ancestral genetic adaptation to living constraints and explain why the consequences of the MTHFR gene mutation are not necessarily very different between hetero- and homozygous subjects, which is further illustrated by recent study results attributing a similar responsibility to both genotypes in the incidence of neural tube defects (35).
Several studies have shown that blood homocysteine levels increase with age (36, 37). The underlying molecular mechanisms of this aged-related increase remain controversial: decreased activity of homocysteine-metabolizing enzymes or lower vitamin status. Homocysteine serum levels were also much higher in postmenopausal compared with premenopausal women (38), which is relevant to our study, where women outnumbered men. Above 35 years of age, our homozygous TT patients had a clearly reduced VEP amplitude compared with the younger age group and the CC genotypes. This suggests that the relative protection against the neurotoxicity of high homocysteine levels possessed by young TT subjects because of the correcting effect of folate/B12 may be lost with age, perhaps because of a dietary deficit in vitamin intake. Unfortunately, we cannot exclude in our study that part of the decrease in amplitude with age is due to duration of the disorder, which in one study of MA was found to be associated with a decrease of the VEP P100 (39).
Another finding of our study is the significant difference in VEP habituation between TT and CC subjects. In the TT subgroup, the degree of habituation between sixth and first blocks of VEP averages was just below that found in healthy volunteers (26). By contrast, in CC subjects, and to a lesser extent in CT subjects, the normal habituation was replaced by an increase of the N1–P1 component with repetitive checkerboard stimulation, i.e. potentiation. The habituation deficit in migraineurs is usually proportional to the first block amplitude (4, 26). It is unlikely, however, that this accounts for the differences found between the three genotypes, in which case one would expect to find the greatest habituation deficit in the CT subgroup, which is not the result found here (see Table 1). An effect of attack vicinity on VEP habituation which normalizes ictally (40) is unlikely, as because of similar exclusion criteria it should be equally distributed over the three groups of subjects. As mentioned in the Introduction, the interictal habituation deficit in migraine could be due to a decreased cortical preactivation level induced by insufficient activation by raphe-cortical serotoninergic pathways (4). The homocysteine derivate DL-homocysteic acid, which acts as an excitatory amino acid, is able to activate median raphe neurons in the animal (23) and has been found to be elevated in the plasma of migraineurs in one study (41). One may thus speculate that increased levels of DL-homocysteic acid in homozygous and heterozygous MTHFR C677T mutations may stimulate deficient serotonin transmission and improve VEP habituation. This would be more marked in TT than in CT subjects, because in the latter the lower amplitude in the first block (discussed above) would favour a greater dishabituation. In a recent cross-sectional study of individuals aged 50–70 years, the MTHFR TT variant was associated with better cognitive performance compared with CC or CT genotypes, an association which was not mediated by homocysteine concentrations or carotid intima-media thickness (42). Knowing the seminal role of habituation in learning processes (43), the question arises whether subjects with the TT genotype might perform better on certain cognitive tests because they habituate better during repeated sensory stimulation.
To conclude, our study has shown that the MTHFR C677T polymorphism is associated with a decrease in amplitude of VEP, and its homozygous state with a significantly better habituation in migraine patients recorded between attacks. We postulate that the amplitude decrease could be due to a mild neurotoxic effect of homocysteine and the better habituation to an effect of the homocysteine derivative, DL-homocysteic acid, on serotoninergic transmission. Further studies are needed to substantiate these hypotheses; they should include dynamic homocysteine blood level measurements.
Acknowledgement
This work is supported by the EU STREP EUROHEAD (LSHM-CT-2004-5044837).
