Abstract
Background
Lack of habituation of visual evoked potentials (VEP) is a common finding in migraine patients between attacks. Previous studies have suggested an electrophysiological familial aggregation pattern associated with migraine. The aim of this study was to evaluate the influence of a positive familial history of migraine on VEP amplitude and habituation.
Methods
We recorded six blocks of 100 VEP during continuous pattern-reversal stimulation in 30 patients with migraine between attacks (MO) and in 30 healthy volunteers, of whom 15 had a first-degree relative suffering from migraine (HVm) and 15 had not (HV).
Results
Both MO and HVm had a significant deficit of VEP habituation and similarly reduced N1-P1 first block amplitudes, compared to HV (habituation slope: MO = 0.033, HVm = 0.021, HV = −0.025, HV vs. MO p = 0.002, HV vs. HVm p = 0.036; mean N1-P1 amplitude in the first block: MO = 9.08 µV, HVm = 9.29 µV, HV = 12.19 µV. HV vs. MO p = 0.041, HV vs. HVm p = 0.076). The first block N1-P1 amplitude was negatively correlated with the habituation slope for both MO (ρ = −.44, p = 0.015) and HVm (ρ = −.56, p = 0.031) while no significant correlation was found in HV (ρ = .17, p = 0.53). There were no differences in VEP latencies between the groups.
Conclusions
Our study suggests that lack of habituation of visual evoked potentials is probably a genetically determined endophenotypic trait that is associated with both migraine and migraine susceptibility. We hypothesize that genetic diversity of populations could account for some of the discrepancies between electrophysiological studies performed in migraine and for interindividual variations among the subgroups.
Introduction
Migraine is a disabling primary headache disorder that actively affects around 11% of the population worldwide (1–4). As a result of the high prevalence of this condition, it has been estimated that a positive family history of migraine could be found simply by chance in > 65% of families (5). In routine clinical practice, however, identifying a positive family history of migraine by interviewing the patient could be unreliable and lead towards an underestimation (6,7). Apart from the three types of familial hemiplegic migraine – rare monogenic subtypes of migraine – the complex polygenic inheritance of the common forms of migraine has not yet been fully elucidated (8). Nonetheless, results from clinical studies show that the risk of developing the disease in first-degree relatives of migraine patients is almost doubled compared to controls (9). In addition, this risk is increased depending on clinical features in the proband, such as younger age at onset and higher pain severity (9), as well as diagnosis of migraine with aura (5). Electrophysiological testing is a minimally invasive and riskless approach that offers the advantage of providing an insight into sensory processing for several stimulation modalities in human beings. Familiar aggregation in migraine has also been found using electrophysiological measures at subcortical (nociception specific blink reflex) and cortical levels (10–12). In clinic-based studies, a habituation deficit to repetitive stimuli has been the most reproducible neurophysiological finding in migraine patients between attacks (13). Moreover, the responses obtained in migraineurs and their first-degree relatives in prior studies have shown a similar habituation profile (10,11). These findings were also corroborated in a study that compared the habituation response of visual evoked potentials (VEP) in pairs of migraineurs made up of parents and their children (12). In addition, this study demonstrated that the correlation within related pairs outweighed the correlation of non-related individuals (12). However, to our best knowledge, the influence of a positive family history of migraine on habituation of visual evoked potentials has not yet been evaluated in healthy adults. Thus, in order to better understand migraine pathophysiology and inheritance, we investigated the habituation response of visual evoked potentials in migraineurs and healthy volunteers with and without first-degree relatives suffering from migraine.
Methods
Subjects
Sixty subjects participated in this study: 30 migraine without aura patients (MO) (14), 15 healthy volunteers with at least one first degree relative who suffers from migraine (HVm), and 15 healthy volunteers without any personal or family history of migraine or any other recurrent headache (HV). Participants were recruited within staff members of the clinic (Clínica Universitaria Reina Fabiola, Córdoba, Argentina) and their family. All subjects were personally interviewed for diagnostic purposes and family history assessment. ICHD-III Beta 2013 criteria were used for migraine diagnosis (14). None of the patients had pharmacological prophylactic migraine treatment, and/or other pharmacological treatments other than the contraceptive pill. All subjects had a normal or corrected visual acuity and were devoid of any other pathology known to affect VEP results. A written informed consent was obtained from all participants. The study protocol was approved by the Institution’s Ethics in health research committee (Clínica Universitaria Reina Fabiola).
Experimental design and recordings
Recordings in patients were performed interictally, within a headache free interval of at least 72 hours before and after a migraine attack. Pattern-reversal visual evoked potentials (VEP) were performed in a quiet room with dimmed light. Subjects were seated in an armchair, 1 m in front of a computer monitor. Based on previous recommendations (15), stimuli were presented as a checkerboard pattern of black and white squares subtending 14’ of arc at a reversal frequency of 3.1 Hz. With the left eye patched, subjects were instructed to fixate on the center of the screen. Surface scalp electrodes were placed at Oz (active electrode) and Fz (reference electrode) according to the 10–20 electroencephalography system. The ground electrode was placed on the forearm. During repetitive stimulation, six consecutive blocks of 100 responses were recorded (Sweep duration: 200msec, bandpass: 1–100 Hz) and averaged online. The stimulation and recordings were performed using a Pentatek® device (Advantek® S.R.L. Argentina).
Data analysis
The six blocks of 100 VEP responses were analysed in terms of peak latencies and peak-to-peak amplitudes by visual inspection. The N1 peak was defined as the most negative point between 60 and 90 ms after the stimulus, P1 as the most positive point following N1 between 80 and 120 ms and N2 as the most negative point following P1 between 90 and 200 ms. The least squares slope of the N1-P1 amplitude ratios between the first and successive blocks was used to determine habituation.
Statistical analyses
Gender proportions between groups were compared using a Z test. Quantitative variables distributions were assessed by the Shapiro–Wilks test. For normally distributed data an ANOVA with a post-hoc Tukey’s Multiple Comparison Test was performed. Variables with a non-Gaussian distribution were compared using the Mann–Whitney U-test with a Bonferroni correction. Habituation between groups was compared using a generalised linear model for repeated measures (within subject factor: N1-P1 amplitude proportion to first block, between subject factor: group) with a post-hoc Tukey HSD test. Two tailed P values were calculated with the significance level set at p < 0.05. Statistical calculations and figures were obtained using the IBM Statistical Package for the Social Sciences (SPSS) for Windows (Version 20.0, IBM Corp, Armonk, NY) and Prism version 6.00 for Windows (GraphPad Software, La Jolla, California, USA).
Results
Population characteristics and VEP results.

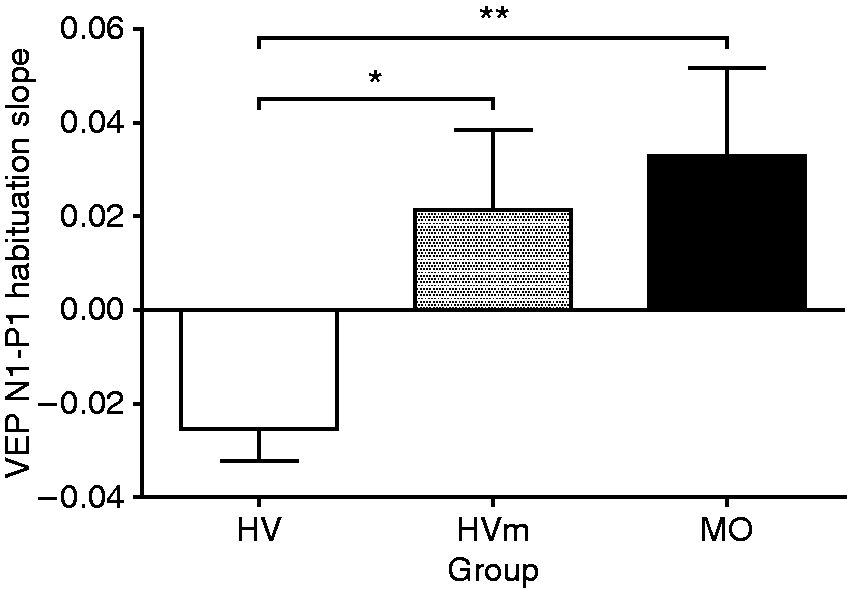
VEP N1-P1 habituation slopes over six blocks of 100 averaged responses (mean ± sem) in HV, HVm and MO groups. (*) p < 0.05 (**)< 0.01.

N1-P1 amplitude variations in blocks two to six, relative to first block amplitude (mean ± sem) in migraine without aura patients (MO: n = 30, dots), healthy volunteers with a first degree relative suffering from migraine (HVm: n = 15, squares) and healthy volunteers without a family history of migraine (HV: n = 15, triangles). (*) p < 0.05; (**) p < 0.01 ANOVA with post-hoc Tukey’s test.

Amplitude of first VEP N1-P1 block in HV, HVm and MO (mean ± sem). (*) p < 0.05 (HV vs MO).

Correlation between habituation slope and VEP N1-P1 first block amplitude in MO, HVm and HV. (*) p < 0.05 (**) p < 0.01.
Discussion
We confirm the results of several previous studies showing that lack of VEP habituation and, in some instances, reduced first block amplitude, characterise migraine patients between attacks (see (13) for review). More importantly, data from our study demonstrate for the first time that the cortical response to repeated visual stimulation in healthy controls with a positive family history of migraine is similar to the response found in migraine patients. This is in line with the results of prior studies on adults in whom nociception-specific blink reflex (10) and contingent negative variation (11) had been evaluated. In a study that investigated VEP habituation and intensity dependence of auditory evoked cortical potentials (IDAP) in 20 pairs of migraineurs made up of parents and their children using a Monte Carlo approach, Sándor et al. had found that although most migraineurs responded similarly to electrophysiological testing, the correlation was stronger within related pairs (12). Furthermore, a review of electrophysiological tests in twins and siblings of the same family showed a strong familial aggregation for both EEG patterns and event-related potentials, though stronger for the former (16). Altogether with our findings, the results of the abovementioned studies suggest that lack of habituation could be a subclinical trait associated with migraine, with a significant genetic contribution.
To our knowledge, this is the first time that the habituation response of VEP has been evaluated in a Latin American cohort. The finding of a deficient habituation in Latin American migraine patients is in line with the results of most previous studies performed in central and southern Europe (13). Conversely, electrophysiological studies from northern Europe addressing this phenomenon did not describe any VEP habituation deficit in their migraine population, despite several methodological adjustments (17). The authors suggested that discordances might be due to a lack of blinding. However, a recent blinded case-control VEP study does not support this hypothesis (18). Genetic-environmental interactions are responsible for neurobiological phenotypes, and are thus likely to determine the electrophysiological and clinical behavior of migraineurs (19). Hence, the genetic background of the participants altogether with environmental factors could account for discrepancies in the results of studies performed worldwide, which cannot so far be explained on the sole basis of methodological differences (20). Despite being recently questioned (17), lack of VEP habituation remains the more reproducible electrophysiological finding in interictal migraine patients. It is well known, however, that this feature is a group and not an individual trait (not all migraineurs have a lack of habituation), and cannot be used therefore as a diagnostic criterion per se (20). Habituation of cortical responses is implicated in learning processes, but it is also a protective mechanism against overactivation (21). Up to now, the precise pathophysiological mechanisms underlying deficient habituation in migraine have not been completely elucidated. Its presence in subsets of healthy volunteers (22) and its absence in some migraineurs (13) suggest that additional neurophysiological modifications are necessary to develop the disease. From all the widespread mechanisms hypothetically associated with deficient habituation in migraine pathogenesis, there are two that hold remarkable interest: reduction of metabolic substrates in the cortex and menstrual-related hormonal fluctuations. Although individually discussed here, these two mechanisms might indeed be tightly correlated since estrogens can regulate many of the key enzymes involved in mitochondrial bioenergetics (23).
Former studies using 31P-MRS have demonstrated a reduction in high-energy phosphates in the cortex of migraine without aura patients (24). This finding allowed the suggestion that they might find themselves compromised in dealing with metabolic stress under certain conditions (25). In fact, contrary to the subsequent diminution in lactate levels in the visual cortex of healthy volunteers (26), lactate concentration has been shown to increase during prolonged visual stimulation in specific migraine subgroups (27). More recently, an electrophysiologically-based study conducted by Gantenbein et al. led to the hypothesis that cortical sensory processing could be energetically more demanding in migraineurs (28). In this scenario, augmented metabolic demands associated to reduced offers could produce a metabolic strain and subsequently disrupt cortical homeostasis. Further studies controlling for both factors could test this hypothesis.
A better known factor associated to migraine is menstrual-related fluctuation of sexual hormones (29). Migraine is close to three times more prevalent in women than in men (30). Results from animal studies lead to the assumption that hormone-related changes in cortical excitability may contribute to the sexual dimorphism in migraine (31). Additionally, GABA mediated inhibitory mechanisms are affected by hormonal fluctuations during the menstrual cycle (32). In our study, while most migraine participants were female, men outnumbered women in the subgroup of healthy volunteers with a positive family history of migraine. Nonetheless, this limitation in our study may shed a light on the possibility that menstrual-related hormonal fluctuations could increase migraine susceptibility in individuals with deficient habituation.
Another possible limitation of this study is the smaller number of subjects in the healthy volunteers’ sub-groups in comparison with the group of patients. In a similar way to our results, VEP habituation studies occasionally report smaller first block amplitudes in patients’ groups, a finding that has been attributed to a reduced level of cortical preactivation (33). The probability exists that with larger sample sizes the difference in first block amplitudes between HV and HVm subgroups found in our study could reach statistical significance.
Finally, even if recent findings suggest that the results of VEP habituation studies are unlikely to be biased by non-blinding analysis (18), the lack of a blinded experiment design might be an additional limitation of our study. All taken into account, it is possible that future investigations addressing this hypothesis in larger, sex-balanced samples may be required.
Conclusions
To conclude, our study shows that healthy subjects with a first-degree family history of migraine have a deficit of habituation of visual evoked potentials like episodic migraine patients between attacks. This abnormality in sensory processing could probably be a genetically determined endophenotypic marker of migraine or migraine susceptibility. Other pathogenic factors, like the influence of ovarian hormones, seem necessary to develop clinically overt migraine.
Article highlights
The visual evoked potential habituation response in healthy volunteers with a positive family history of migraine resembles the response observed in episodic migraine patients in the interictal phase. This finding suggests that lack of habituation of visual evoked potentials is probably a genetically determined endophenotypic trait associated with both migraine and migraine susceptibility.
Footnotes
Acknowledgements
Euroheadpain (FP7-602633) to Marco Lisicki, Kevin D'Ostilio, Jean Schoenen and Delphine Magis.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: the Seventh Framework Programme (10.13039/501100004963), Euroheadpain (FP7-602633).
