Abstract
Background
Many studies report a habituation deficit of visual evoked potentials (VEP) and/or increased intensity dependence of auditory evoked cortical potentials (IDAP) in episodic migraine patients between attacks. These findings have a pathophysiological interest, but their diagnostic utility is not known.
Aims
To perform an audit on a large database of interictal VEP and IDAP recordings in episodic migraine patients and evaluate their diagnostic accuracy.
Methods
We pooled data for VEP habituation and IDAP measured in 624 episodic migraineurs (EM) and 360 healthy volunteers (HV) from three centers. Thresholds were calculated by Receiver Operating Curve analysis and used to calculate sensitivity, specificity, positive and negative likelihood ratios (LR+ and LR-) and the accuracy of each test, using ICHD diagnostic criteria as the gold standard.
Results
In EM, VEP habituation was significantly lower than in HV, and IDAP slopes were significantly steeper. VEP (five blocks of 50 responses), VEP (six blocks of 100 responses) and IDAP had respectively 61.0%, 61.4% and 45.7% sensitivity, and 77.9%, 77.9% and 87.2% specificity. Their positive (LR+) and negative (LR-) likelihood ratios were respectively 2.760, 2.778, 3.570 and 0.500, 0.495, 0.623, with diagnostic accuracies of 65.3%, 69.0% and 54.3%. In combined VEP + IDAP recordings, an abnormality of at least one test had 83.4% sensitivity, 66.7% specificity, 2.504 LR+, 0.249 LR− and 81.1% accuracy.
Conclusions
In this large database, VEP habituation is significantly reduced and IDAP increased in episodic migraine patients between attacks. Taken alone, neither VEP nor IDAP has sufficient diagnostic accuracy. However, when both tests are performed in the same patient, an abnormality of at least one of them is highly predictive of interictal episodic migraine.
Keywords
Introduction
Migraine has no reliable biomarker and is diagnosed by medical history (1). The International Classification of Headache Disorders (2) proposes diagnostic criteria that define episodic migraine as recurrent attacks lasting 4 to 72 hours, where the headache has at least two out of four characteristics (unilateral, pulsating, medium or severe intensity, increased by routine physical activity) and is accompanied by photophobia and phonophobia, and/or nausea/vomiting. Patients not fulfilling all these criteria may have probable migraine (ICHD 3-beta code 1.5) that can be difficult to distinguish from probable tension-type headache (ICHD 3-beta code 2.4). Moreover, early use of symptomatic drugs can halt full development of a migraine attack and all diagnostic criteria may not be fulfilled, as treatment response is not part of them. In countries where headaches are recognized for disability compensation, a rare perverse effect can be that some patients exaggerate their symptoms to be categorized as “chronic”. In such patients, there is a need for biomarkers that can confirm the diagnosis.
As part of the international headache classification (criterion E), neurological examination and ancillary investigations have to be normal in migraine patients, which chiefly serves to exclude secondary headaches. The search for biological or instrumental tests that are able to discriminate migraine from other headaches has been going on for a long time. The most studied and reliable is the nitro-glycerine test, which has 82.1% sensitivity, 96.2% specificity and 85.5% accuracy in migraine without aura, but is much less sensitive in migraine with aura (sensitivity 13.6%, specificity 96.2% and accuracy 72%) (3).
No electrophysiological test appears sufficiently reliable in individual subjects for the diagnosis of migraine (4), but many studies have found that migraineurs are characterized between attacks by a habituation deficit of visual evoked potentials (VEP) and/or increased intensity dependence of auditory evoked potentials (IDAP) (5). Some researchers were unable to reproduce these findings (6–12), which was tentatively attributed to methodological issues (11–12) or differences in migraine phenotypes (13).
To answer some of these questions in a large subject group, we analyzed pooled VEP and IDAP recordings in episodic migraine patients (EM) and healthy volunteers (HV) from one Belgian and two Italian centres. We assessed the diagnostic accuracy of the electrophysiological tests, taking the ICHD diagnostic criteria as the gold standard.
Methods
Subjects
The electrophysiological recordings were performed in episodic migraine patients (EM) and healthy volunteers (HV) at the Headache Research Unit of the University of Liège (Belgium, Centre 1), the Headache Clinic of the Sapienza University of Rome – Polo Pontino (Latina, Italy, Centre 2) and the Headache Clinic of the IRCCS Neuromed (Pozzilli, Italy, Centre 3). Migraine diagnoses were made according to ICHD editions 1 and 2 in use at the time of recruitment (14–15). Data from these centres were pooled retrospectively. Some patients were included in more than one stimulation protocol (Figure 1). We excluded subjects with non-corrected visual or auditory deficits, or with drug intake on a regular basis, or with an attack within three days before the recordings. Migraine patients were recorded at least three days after or before an attack. HV had no personal or family history of recurrent headaches. Local Ethics Committees approved the studies and, in accordance with the 1964 Declaration of Helsinki, informed consent was obtained from each subject.
Distribution of healthy volunteers (HV) and patients (EM) for all types of EP recordings.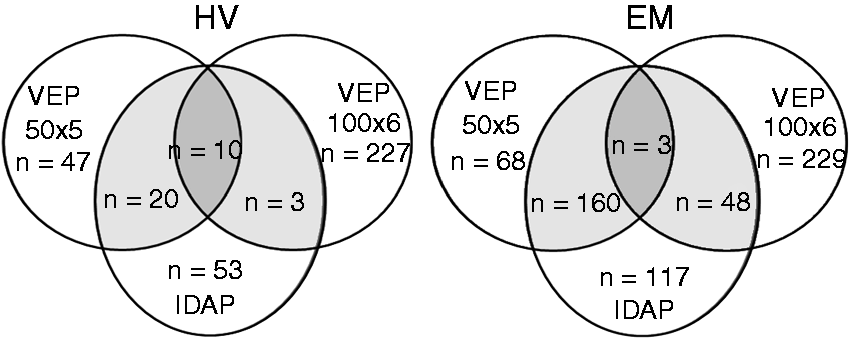
Recordings
VEP50 × 5 recordings and analyses
Number, age and gender distribution of subjects who underwent VEP100 × 6, VEP50 × 5 and IDAP recordings (HV = healthy volunteers; MIG = total migraine patients; MO = migraine without aura; MA = migraine with aura). In Centre 1 some subjects underwent more than one test.
All recordings were performed in Centre 1. Subjects were sitting in an armchair in a dark room with one eye patched and the other fixed on a red dot in the middle of a television screen placed at a distance of 100 cm. When corrected vision was normal in both eyes, the right eye was stimulated, which was the case in the majority of subjects. When there was an inter-ocular difference in visual acuity, the eye with the better-corrected visual acuity was stimulated. After adaptation for 10 minutes, a checkerboard pattern of black and white squares (1 degree and 8’ of arc, contrast 80%) was presented at a reversal frequency of 3.1 Hz. EEG signals were recorded with needle electrodes (active electrode at Oz; reference at Fz; 10–20 reference system). Five sequential blocks of 50 sweeps were collected and on-line averaged using a Cadwell 8400 device (band-pass 1–100 Hz, analysis time 300 ms). The N1 component was identified as the most negative point between 60 and 90 ms after the stimulus and the P1 component as the most positive point between 80 and 120 ms. In each block we measured N1 and P1 latencies and peak-to-peak N1–P1 amplitude. To assess habituation, the change in N1–P1 amplitude between the fifth and the first block was expressed in percentages (negative values = habituation, positive values = potentiation).
VEP100 × 6 recordings and analyses
These recordings were performed in Centres 1, 2 and 3. In the first two Centres, data were amplified with a Digitimer D360 amplifier (band-pass 0.05–2000 Hz, gain 1000) and recorded with a CED™ power 1401 device (Cambridge Electronic Design Ltd, Cambridge, UK). Responses in each block were averaged off-line using the Signal™ software package version 4.08 (CED Ltd). In Centre 3, signals were recorded and averaged on-line with a Medelec Synergy, PLINTH, CareFusion device (Middleton, Wisconsin, USA). Recording conditions were similar to those for VEP50 × 5, except for a 15’ of arc check size. Six sequential blocks of 100 sweeps were collected and VEP components were identified as described above. Percentages of habituation were measured between the sixth and the first block of averaged sweeps.
IDAP recordings and analyses
All recordings were performed in Centre 1 using either on-line averaging with a Cadwell 8400 device (band-pass 1-150 Hz, analysis time 300 ms) (HV = 65, EM = 273) or off-line averaging using CED™ 1902 pre-amplifiers, a CED™ 1401 device and the Signal™ software package version 1.88 (CED Ltd) (band-pass = 0.001–1000 Hz, analysis time 400 ms) (HV = 30, EM = 78). The EEG was recorded with an active electrode at Cz and linked mastoids as reference. Stimuli (1,000 Hz tones) were delivered binaurally through earphones at four intensities above the sensation level (40, 50, 60, and 70 dB for the Cadwell device and 50, 60, 70 and 80 dB for the CED device) in a pseudo-randomised order. With the Cadwell device (interstimulus interval varying between 1,640 msec and 1,887 msec) 100 artefact-free sweeps were automatically selected for averaging at each stimulus intensity (16). With the CED device (repetition rate 0.474 Hz), 120 trials were collected at the four intensities and at least 100 artefact-free trials were used for averaging (17). After identification of the AEP components N1 (between 60 and 150 ms post-stimulus) and P2 (between 120 and 200 ms), peak-to-peak N1–P2 amplitudes were measured at the various stimulus intensities and IDAP was expressed by the amplitude/stimulus intensity function (ASF) slope in μV/10 dB.
Statistical analyses
The Shapiro-Wilk test was used to test the normality of data distributions. Results were expressed as mean and standard deviations. The significance level was set at p < 0.05.
Comparison between study groups
As our data were not normally distributed, the HV and EM groups were compared with the Bonferroni-corrected Mann-Whitney U test. The same test was used to compare the Belgian and Italian subjects’ subgroups.
Sensitivity and specificity of VEP and IDAP
To obtain cut-off values with the best sensitivity and specificity for each test, a Receiver Operating Curve (ROC) was configured for each model. The area under the ROC (AUC) based on a 15% prevalence value for migraine was used to calculate the thresholds, sensitivity (true positive/all patients), specificity (true negatives/healthy subjects), positive (true positives/all positives) and negative (true negatives/all negatives) predictive values and accuracy (true positives + true negatives/all tests) of each electrophysiological test (18–19), after positive and negative likelihood ratios were calculated. Hilgers’ distribution-free 95% confidence bounds were calculated (20). Different AUCs were compared with MedCalc for Windows, version 10 (MedCalc Software, Ostend, Belgium).
Diagnostic performance was evaluated for each test separately and for the combination of VEP and IDAP in subjects who had both recordings.
Results
In total, 984 subjects were recorded over a time span of 16 years (1997–2013): 360 HV and 624 EM patients, among whom 439 had migraine without aura (ICHD code 1.1) and 185 had migraine with typical aura (ICHD code 1.2.1) (Table 1). Some patients were included in more than one protocol (Figure 1).
VEP habituation and IDAP
Mean percentage habituation for VEP50 × 5 and VEP100 × 6, and mean ASF slopes in healthy volunteers (HV) and episodic migraineurs (EM) recorded interictally (for VEP: negative values = habituation, positive values = potentiation).

IDAP was significantly greater in patients between attacks than in controls.
The VEP habituation deficit in migraineurs remained significant when the two subgroups of subjects from Belgium or Italy were analysed separately. In the Italian EM subgroup there was a trend for a stronger potentiation, i.e. a greater VEP habituation deficit, but this did not reach the level of significance (p = 0.11).
Diagnostic yield of VEP and IDAP
The threshold values for VEP habituation and IDAP slope calculated by ROC analysis were +0.43% for VEP50 × 5, −1.16 % for VEP100 × 6 and +1.19 µV/10 dB for IDAP slope (Figure 2). Using these thresholds, we determined the sensitivity, specificity, predictive values, likelihood ratios as well as the accuracy of each electrophysiological test and of their combination (Table 3). Sensitivity was higher for VEP (61.4%) than for IDAP (45.7%), but specificity was greater for the latter (87.2% vs 77.9%).
ROC curves displaying sensitivity and specificity for all possible threshold values of VEP habituation and IDAP (bold lines) and Hilgers’ 95% confidence intervals (thin dashed lines). ROC curves of habituation for VEP50 × 5, VEP100 × 6, and IDAP displayed as bold lines. The diagonals indicate the casualness of test distributions, whereas test values in the upper right portion of each graph indicate non-casualness. The threshold values indicated by arrows on each graph correspond to the highest specificity/sensitivity of VEP habituation and IDAP. Sensitivity, specificity, predictive values, likelihood ratios and accuracy of VEP50 × 5, VEP100 × 6, IDAP and the combination of both tests (abnormality of at least one of VEP or IDAP). HV = healthy volunteers. EM = episodic migraine patients.

In subjects for whom both tests were available, when an abnormal value in at least one of the two tests respective to the above-mentioned thresholds was taken into account, sensitivity was better (83.4%) but specificity worse than for one evoked potential recording (66.7%).
Positive likelihood ratios were always superior to 1, and negative likelihood ratios always inferior to 1. Diagnostic accuracy was best when either VEP or IDAP abnormalities were considered in the same subjects (81.1%). When both VEP habituation and IDAP were abnormal, specificity increased to 90.9% at the expense of a low sensitivity (24.6%).
Discussion
This analysis of pooled data from three different headache centres confirms in a large cohort of 624 migraine patients and 360 healthy controls the neurophysiological results previously reported in smaller study samples: habituation of cortical visual evoked potentials (VEP) is decreased and intensity dependence of cortical auditory evoked potentials (IDAP) increased in migraineurs between attacks (16,21). Taken together, this indicates that cortical information processing is modified in most migraine patients between attacks, which can be considered as an endophenotypic biomarker of the disorder. The underlying neurobiological mechanisms are still under discussion, but might involve lowered preactivation levels of sensory cortices due to thalamo-cortical dysrhythmia and low serotonergic tone (5,17,22–23).
Different recording devices, investigators or stimulation protocols yielded comparable VEP results. For instance, there was no significant difference in habituation rates between Belgian and Italian centres or between measurements made on five blocks of 50 or six blocks of 100 averaged responses. This suggests that the results are reproducible in different laboratory settings.
The principal objective of our study was to evaluate the diagnostic usefulness of VEP and IDAP recordings in migraine. Diagnostic accuracy is moderate to fair for VEP habituation (65.3% and 69%) and IDAP (54.3%) taken separately. However, when both tests are combined in the same subject and an abnormal value of one of them is considered for calculations, diagnostic accuracy rises to 81.1% and positive predictive value to 94.1%, which means that a subject with an abnormality in either VEP habituation or IDAP is most likely to have episodic migraine in an interictal state. We have previously shown that there is no significant intra-individual correlation between VEP habituation and IDAP (24). It is thus not surprising that few migraine patients have an abnormality in both tests.
The sensitivity (83.4%) and specificity (66.7%) of having at least one abnormality in combined VEP-IDAP recordings are fair enough to be useful for migraine diagnosis. They are similar or superior to those reported for VEP latency in multiple sclerosis (25) (69% and 62% respectively) or optic neuritis (26) (72% and 25%). For comparison, the P300 latency had a sensitivity of 80% to detect mild cognitive impairment (27). As mentioned above, most migraine patients can be diagnosed by taking their history. The differential diagnosis between tension-type headache and mild migraine may however be problematic (28) and revised criteria for tension-type headache are proposed in the appendix of ICHD-3beta to solve this problem. In a recent field-testing, Chu et al. (29) applied these revised criteria in a cohort of tension-type headache patients and found that 59.3% did not fulfil them: 19.6% of patients fulfilled the criteria for probable migraine and 39.7% were unclassifiable. In such patients, VEP habituation and IDAP studies could be useful, because habituation deficit has not been found in patients suffering from tension-type headache (30).
A limitation of the evoked potential studies is that they are abnormal in migraine patients only between attacks (31). As a consequence, patients recorded in close vicinity to or within an attack may have normal recordings. Similarly, VEP habituation is on average normal in chronic migraine (32) that has therefore been compared to “a never-ending attack” (33). As a corollary, patients who fulfil the criteria for chronic migraine by history and have abnormal evoked potentials might be suspected to overestimate or exaggerate their headache frequency, or to overuse acute anti-migraine medication. In medication overuse headache, habituation of cortical somato-sensory evoked potentials is indeed deficient (34), although this remains to be shown for VEP. Another limitation could be that the evoked potential studies in migraine have been criticised for their non-blinded methodology and poor test-retest repeatability (11,12). Regarding the blinding problem, it is worth mentioning that one IDAP study (17) and four VEP studies (35–38) that confirmed the electrophysiological abnormalities in migraine were blinded for the analyses. In a very recent study (39), the same VEP data were analysed separately by two investigators, one of them blinded to the migraine diagnosis and phase. The analyses by both confirmed interictal VEP habituation deficit in episodic migraine and normal VEP habituation in healthy volunteers and migraine patients during an attack. VEP habituation percentages measured by both investigators were strongly correlated, suggesting that blinding has little influence on the results of VEP studies. Test-rest repeatability for VEP habituation in migraine patients was found to be good in the same study, but moderate in another (40). In future test-retest studies, it would be preferable to use the binary criterion of “normal or abnormal” recordings based on the thresholds determined in the present study.
The trend for greater VEP potentiation in Italian than in Belgian migraine patients found here might nurture the discussion of other causes for discrepancies in the literature. It is indeed likely that neurophysiological phenotypes are influenced by underlying genotypes, and the latter can differ between patients coming from distant geographical areas. In the case of the VEP habituation deficit in migraineurs, there might be a gradient between southern European countries, where it would be greatest (see above) and Nordic populations, where it could be non-significant. Whether this could be due to different genotypes or environmental differences such as illumination remains to be determined. In fact, we have already identified in Belgian patients two different neurophysiological phenotypes when IDAP is sensitized by simultaneous high-frequency flash stimulation (13). Studies of genotype-phenotype correlations and future electrophysiological tests that are more sensitive, for the detection of more subtle abnormalities, are necessary to verify this hypothesis.
To conclude, this study confirms in the largest sample analysed so far, comprising subjects from three different centres, that habituation of visual evoked potentials is on average deficient and intensity dependence of auditory evoked cortical potentials is increased in episodic migraine patients between attacks. Accuracy is not sufficient for diagnostic purposes, if only one evoked potential recording is considered. However, in subjects undergoing both electrophysiological tests, it is satisfactory for diagnosing episodic migraine interictally, suggesting that evoked potential studies could be useful as an additional tool for diagnosing migraine in uncertain cases, although this needs to be confirmed in large, prospective controlled studies.
Footnotes
Clinical implications
This retrospective multicentre audit on a large number of subjects confirms that episodic migraine (EM) patients are characterised between attacks by a habituation deficit of Visual Evoked Potentials and an increased Intensity Dependence of Auditory Evoked Potentials compared to healthy volunteers (HV).
Taken alone, neither VEP nor IDAP have sufficient diagnostic efficacy to distinguish EM from HV, although both have good positive predictive values (89% and 93% respectively). When both tests are combined in the same subject, however, an abnormality of at least one of them is highly predictive of interictal EM (81% efficacy), suggesting that the combination of VEP and IDAP could be a useful additional tool for diagnosing migraine in uncertain cases.
Acknowledgements
The authors are grateful to Drs Afra J, Bohotin V, Bolla M, Cremers J, Di Clemente L, Iezzi E, Proietti Cecchini A, Sandor PS, Sasso D’Elia T, Viganò A, for making some of their recordings available for this study.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: JS was supported by research convention 3.4.650.09 of the National Fund for Scientific Research–Belgium, Special Research Funds and grants from the Fonds Frédéricq of the Faculty of Medicine–University of Liège. AK was partially supported by the Italian Ministry of Health for Institutional Research and by the 5 per mille 2014 fiscal SRP institutional fund. DM and JS's work was supported by the European Union (EUROHEADPAIN, Seventh framework program, Grant agreement no 602633). GC was financially supported by the Italian Ministry of Health and Fondazione Roma.
