Abstract
Studies in experimental animals have suggested that the hypocretin/orexin system may be involved in migraine pathophysiology. Using a case-control design study, we genotyped 246 migraine patients and 239 healthy controls for the 1246G→A polymorphism of the hypocretin receptor 2 (HCRTR2) gene. Genotypic and allelic frequencies of the examined polymorphism were similarly distributed between cases and controls (χ2 = 2.22, P = 0.14 and χ2 = 2.45, P = 0.29, respectively). When different migraine subgroups were compared (migraine with aura vs. migraine without aura and episodic vs. chronic migraine) no significant difference was found. Comparison of the clinical features of the disease with the 1246G→A genotypes showed no significant difference. Our data suggest that the HCRTR2 gene is not a genetic risk factor in migraine.
Introduction
Migraine is a paroxysmal neurovascular disorder that affects approximately 12% of the western population (1). Several studies have suggested that genetic factors play an important role in migraine. At present, only a rare subtype of migraine, familial hemiplegic migraine, has been associated with mutations in the CACNA1A, ATP1A2 and SCN1A genes (2–4). Although several genes have been supposed to be involved in the pathophysiology of the disease, no major gene related to migraine has yet been found.
In the late 1990s, two independent research groups discovered a novel neuropeptide system in the hypothalamus (5, 6). The new system, which has been called the hypocretinergic or orexinergic system, influences a wide range of physiological and behavioural processes in mammals (7, 8). This system encompasses two peptides, hypocretin-1 and -2 (orexin-A and -B) that are encoded by a common precursor, preprohypocretin, and two G-protein coupled receptors (HCRTR1 and HCRTR2). Hypocretin-1 and -2 binding to its receptors results in elevation of cytoplasmic calcium concentrations (9). The peptides of the hypocretinergic system may influence feeding, the sleep–wake cycle, cardiovascular function, autonomic function, hormone secretion and memory (10). This system is therefore crucial in the regulation of motivational and adaptative behaviour to metabolic and environmental stimuli. Recent studies have revealed a new role for hypocretins in reward seeking, drug relapse and drug addiction (11, 12).
Several of the functions regulated by the hypocretinergic system are significantly impaired in patients with migraine and this could be relevant to the pathophysiology of the disease. The hypothalamus is known to play an important role in migraine (13). Premonitory symptoms preceding a migraine attack, such as fatigue, yawning, excessive sleepiness and craving for certain food, indicate an involvement of different hypothalamic nuclei. Recently, a number of experimental studies have provided strong evidence that hypocretins are also involved in pain modulation within the central nervous system. Bingham et al. have reported that intracerebral orexin injection produces analgesia in experimental animals (14). The injection of orexin-A reduces mechanical allodynia in a neuropathic pain model (15). Finally, hypocretin-1 receptor activation attenuates neurogenic dural vasodilation in an animal model of trigeminovascular nociception (16). The results of these studies suggest a link between the hypocretinergic system and the nociceptive phenomena observed in primary headache disorders.
We have previously reported that the 1246G→A polymorphism of the hypocretin receptor 2 (HCRTR2) gene is significantly associated with cluster headache (17). This association has been confirmed in a large study from Germay, showing that homozygous carriers of the G-allele have a twofold increase in disease risk (18). The 1246G→A polymorphism of the HCRTR2 gene induces an amino acid substitution of valine to isoleucine at position 308, which could engender an altered receptor function. Migraine and cluster headache are sometimes comorbid and it has been suggested that common genetic factors or functional alterations in the same central neurological circuits may play a role in the pathogenesis of both disorders (19). The aim of the present study was to evaluate the association between migraine and the HCRTR2 gene. We performed a case–control association study in a cohort of migraine patients, referring to a Headache Centre, and in healthy controls, in order to evaluate the effects of the 1246G→A polymorphism of the HCRTR2 gene on the occurrence and clinical features of migraine.
Methods
Subjects
A group of 246 consecutive unrelated migraine patients (169 females, 77 males; mean age ± SD 41.5 ± 12.0 years), recruited from the Headache Centre of the University of Torino (Italy), were selected for the study. The diagnosis of migraine was made, according to the International Classification of Headache Disorders-II criteria (20), by a neurologist specialized in headaches. Thirty-eight patients (15%) fulfilled the diagnostic criteria for migraine with aura and 208 (85%) for migraine without aura. The mean age at onset and duration of the disease were 18.6 ± 9.8 and 21.4 ± 15.4 years, respectively. Patients underwent an extensive physical and neurological examination. A standardized record of all the clinical characteristics of headache, suitable for computer analysis, was obtained. A group of 239 age- and sex-matched healthy subjects (152 females, 87 males, mean age ± SD 41.5 ± 13.3 years) were used as controls. The controls were blood donors and were screened to exclude primary headaches. Both patients and controls were of Caucasian origin. Written informed consent was obtained from all participants and the study was approved by the local Ethics Committee. The same populations had previously been used for genetic association studies in migraine (21–24).
DNA extraction and genotyping
Patients and controls were genotyped for the polymorphism 1246G→A of the HCRTR2 gene, located in exon 5 (single nucleotide polymorphism database: http://www.ncbi.nlm.nih.gov). At present, this is the only well-known exonic polymorphism in the aforementioned gene. Genomic DNA was extracted from 200 µl of whole blood using the QIAamp DNA mini Kit (Qiagen, Milan, Italy). Polymerase chain reactions (PCR) were performed in a final volume of 50 µl, with 90 ng of genomic DNA, 0.4 U of Taq Gold DNA polymerase, 250 n
Statistics
Differences in allelic (AF) and genotypic frequencies (GF) between cases and controls were compared using Fisher's exact test or χ2 test, as appropriate. Markov chain Monte Carlo (MCMC) simulations were then performed. Patients' clinical characteristics were analysed with
Results
Hardy–Weinberg equilibrium was verified for all tested populations. Table 1 shows GF and AF of the polymorphism in cases and controls. GF and AF in our control group were remarkably similar to those reported in other European populations (26). GF and AF of the examined polymorphism were not significantly different between cases and controls (χ2 = 2.22, P = 0.14 and χ2 = 2.45, P = 0.29, respectively; MCMC χ2 = 4.35, P = 0.11 and χ2 = 4.27, P = 0.12). Multiple comparisons according to different genotypes (G/G +G/A vs. A/A, G/A + A/A vs. G/G) showed no significant differences. No significant difference between migraine with aura and migraine without aura was found (χ2 = 0.71, P = 0.40 and χ2 = 2.11, P = 0.35). No difference was found between chronic and episodic migraine (χ2 = 0.12, P = 0.73 and χ2 = 0.52, P = 0.77).
Genotypic distribution (GF) and allelic frequencies (AF) of HCRTR2 1246G→A polymorphism

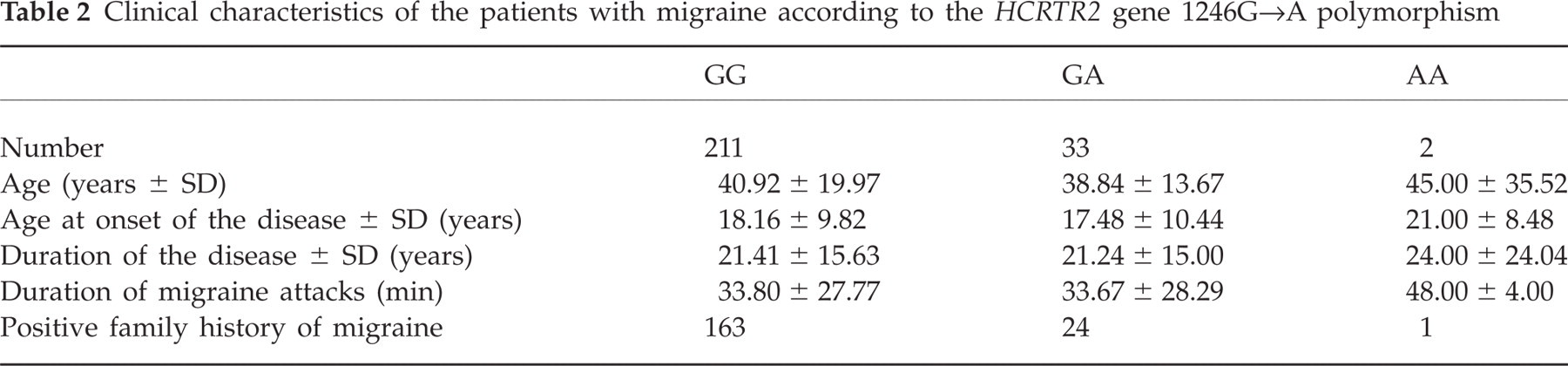
Table 2 compares the clinical characteristics of migraine patients according to different HCRTR2 gene genotypes. Different genotypes had no significant effect on the examined clinical characteristics.
Clinical characteristics of the patients with migraine according to the HCRTR2 gene 1246G→A polymorphism
Discussion
The findings of our study do not support the hypothesis that genetic variations within the HCRTR2 gene confer increased risk of migraine. Both genotypic and allelic frequencies of the 1246G→A polymorphism were similarly distributed between cases and controls and no evidence was found of an association between the studied polymorphism and the disease. When patients were divided into migraine with aura and without aura subgroups, no significant difference was found. Finally, different HCRTR2 genotypes did notsignificantly modify the clinical characteristics of migraine. It is therefore unlikely that genetic variations within the HCRTR2 gene greatly contribute to disease susceptibility.
This is the first study, to our knowledge, to evaluate the association between the HCRTR2 gene and migraine and our results should be interpreted with caution. There are some limitations to our study that may have influenced the results. Our cases were recruited from a Headache Centre and we examined a population with a high percentage of chronic migraine patients. In addition, the number of migraine with aura patients genotyped may have been too low to detect a genetic association. It is well known that the results of genetic association studies must be interpreted cautiously until they are confirmed by multiple replications and/or by well-designed studies involving large numbers of both cases and controls. Additional studies in different populations are therefore needed to confirm the findings of our study.
The hypocretin-producing neurons of the central nervous system are specifically located to the dorso-lateral hypothalamus and have widespread anatomical projections within the central nervous system, with extensive dense projections to the locus coeruleus, dorsal raphe nuclei, amygdala, suprachiasmatic nucleus, basal forebrain, cholinergic brainstem and spinal cord (27). There are minor projections to the cortical regions, central and anterior amygdaloid nuclei and the olfactory bulb. The hypocretins are thought to act primarily as excitatory neurotransmitters. Systemic and intracerebroventricular administration of hypocretins directly stimulates noradrenergic cells on the locus coeruleus in rats and monkeys, suggesting a role for the hypocretins in various central nervous functions related to noradrenergic innervation (28). Their actions on serotonin, histamine, acetylcholine and dopamine neurotransmission are also thought to be excitatory and a facilitatory role on gamma-aminobutyric acid and glutamate-mediated neurotransmission has been suggested (29, 30). As regards the complex mechanism of action of the hypocretins and the multiple physiological functions that are influenced by these neurotransmitters, further studies are needed in order to exclude the involvement of these neuropeptides in migraine.
In conclusion, we have found no evidence of an association between the HCRTR2 gene and migraine. Our results do not support the hypothesis that the HCRTR2 gene is a genetic risk factor for migraine or might influence the disease characteristics. However, new studies with additional genetic markers are warranted in order to elucidate the possible involvement of the hypocretinergic system in the pathophysiological mechanisms of migraine.
Acknowledgements
The study was supported by grants from the Ministero dell'Università e della Ricerca Scientifica and the Regione Piemonte (Italy).
