Abstract
We have previously shown decreased, nocturnal lipolysis in both phases of cluster headache (CH). Lipolysis is stimulated by noradrenaline (NA), growth hormone (GH) and cortisol, and inhibited by insulin, hormones which are directly or indirectly regulated by the hypothalamus. Our aim was to investigate the nocturnal secretion of NA, GH, cortisol and insulin in nine CH patients in remission and 10 healthy controls. Nocturnal venous blood samples were collected in hourly intervals for analysis of NA, cortisol and insulin and in 30-min intervals for GH. We found a reduced increase in GH between 24.00 h and 01.00 h (ANOVA, P < 0.05) in CH patients. Nocturnal secretion of NA, cortisol and insulin did not differ significantly between the groups. The altered nocturnal GH pattern that was seen in CH patients in remission might in part explain the altered nocturnal lipolysis previously found and further indicate a permanent hypothalamic disturbance in CH.
Introduction
The aetiology of cluster headache (CH) is still not known, but findings from neuroimaging studies (1, 2), as well as the effect of deep brain stimulation (3), have demonstrated involvement of the hypothalamus, particularly in the active headache period. The hypothalamus is important for regulation of pituitary hormone secretion and regulation of circadian rhythms. Altered hormonal regulation in the active CH period has been reported in a number of studies. There are also a few reports of melatonin, prolactin and cortisol release in the remission phase (4–7), which indicate a persistent alteration of the secretory patterns, possibly due to permanent hypothalamic disturbance.
In a previous study we have shown diminished nocturnal lipolysis in CH patients, both in the active period and in remission, reflecting permanent metabolic disturbance in CH. In addition to reduced lipolysis, CH patients in remission show an altered temporal pattern of the nocturnal lipolysis rhythm (8). We have also shown that noradrenaline (NA)-induced lipolysis is increased in CH remission, indicating up-regulated receptors, and that the altered pattern of lipolysis is not due to defective β-receptors (9). Lipolysis is acutely stimulated by activation of the sympathetic nervous system and release of catecholamines (10–12). In contrast, insulin has a potent antilipolytic effect (13, 14). Growth hormone (GH) stimulates lipolysis also, but in a slower modulating way (15, 16). The effect of cortisol on adipose tissue metabolism is still controversial and studies have shown a stimulating as well as an inhibiting effect on lipolysis (17, 18). The hypothalamus is directly involved in the regulation of sympathetic nervous system activity and release of NA as well as GH and cortisol release from the anterior pituitary, and indirectly involved in the regulation of glucose homeostasis and insulin secretion. Altered secretion of one or more of these hormones in CH remission might explain the altered lipolysis in CH and may also further indicate permanent hypothalamic disturbance. However, nocturnal levels of NA, GH and insulin have never been measured in CH remission.
The aim of this study was to investigate the nocturnal secretion of NA, GH, cortisol and insulin, hormones with potential effect on lipolysis and of which the secretion at least partly is regulated by hypothalamic activity. Hormone concentrations were measured in venous blood and the results compared between CH patients in remission and healthy subjects.
Methods and materials
Subjects
Nine CH patients in the remission phase, suffering from CH according to the 2nd edition of the International Headache Society criteria (19), participated. All CH patients except two were otherwise healthy and took no chronic medication. One CH patient had mild hypertension and medicated with hydrochlorotiazid + amilorid and one had allergy, which was treated with a histamine-1 antagonist and nasal glucocorticoids. In addition, 10 healthy volunteers matched for sex and smoking habits were included, none of them suffering from headache (Table 1). The Ethics Committee of Karolinska University Hospital approved the study and written informed consent was obtained from all subjects.
Clinical characteristics of CH patients and healthy subjects

Cluster headache (CH) patients and healthy subjects were matched with regard to sex and smoking habits. As regards age, CH patients were found to be significantly older than healthy controls.
P < 0.01 (Mann–Whitney U-test).
Protocol
All subjects were fasting from 18.00 h, but they were allowed to drink water. They were not allowed to smoke. Subjects arrived at the hospital at 20.30 h and stayed overnight. At 21.00 h, a cannula was inserted into an antecubital vein. Subjects went to bed at 23.00 h and during the night there was a continuous dim light in the room. Venous blood samples were collected between 23.00 and 07.00 h, at hourly intervals for NA, cortisol and insulin and at 30-min intervals for GH. Blood samples for GH, cortisol and insulin were centrifuged and serum was stored at −20°C until analysis. Blood samples for analysis of NA were collected in heparinized ice-chilled tubes, centrifuged immediately at 4°C and plasma was stored at −70°C.
Assays
All samples were analysed at the Laboratory of Clinical Chemistry at Karolinska University Hospital, Huddinge by accredited, commercial methods. The analyses were carried out at two time points. Serum GH was determined by FluoroImmunoAssay (Wallac Oy, Turku, Finland) with a sensitivity of 0.01 µg/l. The intra-assay coefficients of variation (%CV) were 4.00% and 3.55% (time points 1 and 2), respectively, and the interassay %CVs were 2.77% and 1.86%. Serum insulin and serum cortisol were determined by an immunometric method (ECLIA) (Roche Diagnostics GmbH, Mannheim, Germany). For insulin the sensitivity of the assay was 1.39 p
Statistics
Data was analysed using
Results
The nocturnal profiles for GH, NA, cortisol and insulin for CH patients and healthy controls are shown in Fig. 1a–d.

The nocturnal profiles for growth hormone (GH) (a), noradrenaline (NA) (b), cortisol (c) and insulin (d) are shown for cluster headache (CH) patients in remission (▪) and healthy subjects (○). Venous blood samples were collected between 23.00 and 07.00 h, at hourly intervals, for NA, cortisol and insulin, and at 30-min intervals for GH. The nocturnal pattern for GH was different in CH patients compared with healthy subjects (P = 0.03) (
Growth hormone
GH varied significantly over time (P = 0.01). The nocturnal pattern was different in CH patients compared with healthy subjects (P = 0.03). This was shown statistically by the interaction effect between time and group, and clinically observed as a smaller increase between 24.00 and 01.00 h in CH patients, reaching a level of 0.41 µg/l (0.22–2.14) (median and interquartile range) compared with 2.83 µg/l (1.93, 3.90) in controls. The maximum GH peak did not differ significantly between the groups, nor did the nocturnal GH concentrations. In CH patients, the maximum GH peak occurred between 24.00 and 02.30 h in six subjects, at 23.00 h in two subjects and at 05.00 h in one subject. In healthy controls, the maximum peak occurred between 24.00 and 02.30 h in eight subjects and between 06.00 and 07.00 h in two subjects. Time to the maximum peak did not differ significantly between the groups (Table 2). Moreover, regression analysis was carried out between the variable time from the previous bout and the variables nocturnal mean concentration (R = 0.27, P = 0.49), maximum GH value (R = 0.35, P = 0.36) and time to the maximum GH value (R = 0.15, P = 0.69). No significant correlations were found.
Nocturnal secretion of GH, cortisol, NA and insulin in cluster headache patients in remission and healthy subjects
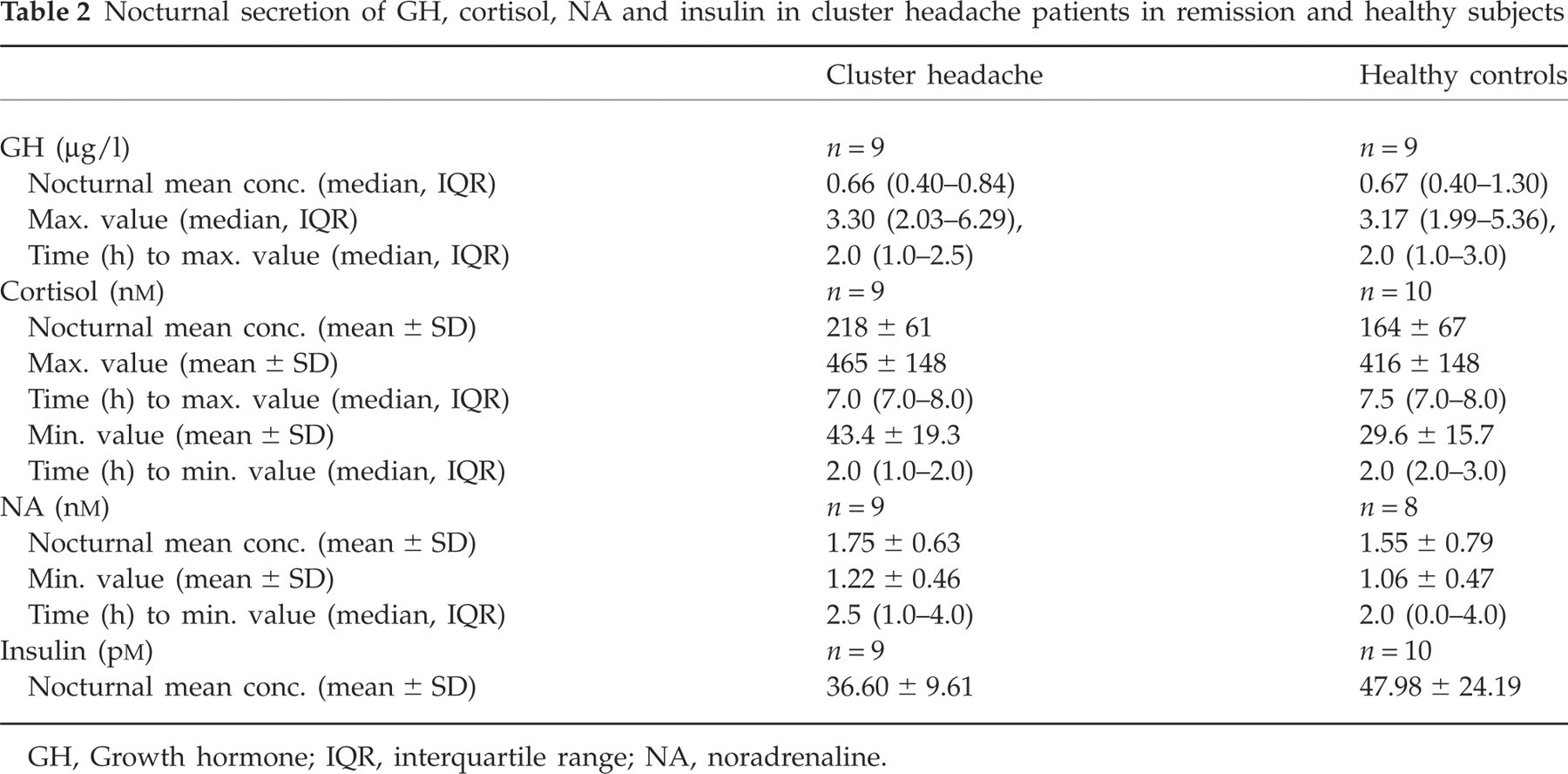
GH, Growth hormone; IQR, interquartile range; NA, noradrenaline.
Noradrenaline
Nocturnal NA concentrations did not differ between CH patients and healthy controls, nor did the nocturnal minimum NA level or time to nadir (Table 2). There were no significant time-related changes in NA concentrations over the night, nor did the nocturnal temporal patterns differ between the groups.
Cortisol
Cortisol concentrations increased significantly during the night (P < 0.00001) and the temporal patterns did not differ between the groups, nor did nocturnal cortisol concentrations, levels of the maximum cortisol peaks, minimum levels, or the time to the maximum and minimum levels, respectively (Table 2).
Insulin
There were no significant time-related changes in insulin concentrations during the night. Temporal patterns did not differ between the groups, nor did the nocturnal insulin concentrations during the night (Table 2).
Discussion
Growth hormone
Time-related changes in GH concentrations were found in CH patients, shown by a reduced increase in GH between 24.00 and 01.00 h. The level of the maximum peak did not differ significantly between CH patients and controls, nor did the time to maximum peak.
GH is secreted in short bursts
Sleep is one of the most important triggers of GH secretion and, in men, approximately 70% of the daily GH output occurs during early sleep (23). There is a relationship between sleep and CH. CH attacks often occur at night and there is a correlation to rapid eye movement sleep (24). Moreover, there are some reports of a disturbed sleep pattern in both phases of CH (25, 26) and it has also been shown that obstructive sleep apnoea is more common in CH patients than in the general population (27, 28). However, in the absence of simultaneous sleep recordings in the present study, we do not know whether altered sleep patterns influenced the GH secretory rhythm.
Nocturnal GH secretion is under the influence of other regulating mechanisms also. For example, the hypothalamus influences GH release via growth hormone-releasing hormone stimulation and somatostatin inhibition. Somatostatin has also been shown to have central pain modulatory properties (29), and octreotide, a somatostatin analogue, has been shown to be effective in the acute treatment of CH attacks (30). However, if there are any alterations in basal plasma somatostatin concentrations in CH patients, is still unclear. Thus, we cannot exclude an alteration in somatostatinergic tone as an explanation of our findings.
A correlation between GH secretion and the time from the previous bout or time to the next bout may indicate that the somatotropic axis is involved in initiating or ending the active period. It was not possible to obtain specific information from the study subjects about the time between the study and the next bout, but a regression analysis of the existing data showed no significant correlations between the time from the previous bout and GH secretion. To evaluate this topic further, another study design would be required.
Obesity and age are factors that are well known to reduce GH levels (31). Obesity is associated with decreased GH pulse frequency, whereas pulse amplitude declines with age. Healthy subjects had slightly higher BMI than CH patients, while CH patients were older than controls. The estimation of GH levels was adjusted for BMI and age, respectively, in separate models. The statistical estimates of GH were not affected by age, thus the variation in GH between groups was not explained by differences in age. BMI was found to correlate with GH pattern and for that reason BMI was included as a covariate in the final
Noradrenaline
The present study is the first to measure nocturnal basal NA concentrations in the remission phase. We did not find any differences in NA nocturnal concentrations or temporal patterns between CH patients and controls. Occasional supine daytime NA levels, as well as response to standing up, have previously been reported to be normal in CH remission (32). This is in contrast to findings in the active period, which demonstrate decreased plasma concentrations of NA in early morning as well as in late evening, as an indication of sympathetic hypofunction (33). Furthermore, CH patients in remission as well as in active period, as an indication of long-term sympathetic function, have shown reduced levels of NA and adrenaline in platelets (34). We have previously found diminished lipolysis in CH patients in remission and in the active period, interpreted as an indication of sympathetic hypofunction and hypothalamic disturbance (8). In CH patients in remission, we have also found increased β-receptor response to NA in subcutaneous adipose tissue, indicating up-regulated β-receptors and also interpreted as a sign of a generalized sympathetic hypofunction (9). The present results do not exclude an autonomic disturbance in the remission phase. The concentration of NA in plasma is a net result of a number of processes, and changes in venous blood may reflect regional as well as global changes. Correcting for age differences in the
Insulin
The hypothalamus is indirectly involved in the regulation of glucose concentrations and insulin secretion, in part through glucose-sensing neurons in the ventero-medial nucleus that trigger neuroendocrine responses to hypoglycaemia (35). There are no previous studies of insulin levels in CH and in the present study we could find no indications of disturbed insulin regulation in CH remission. However, it is possible that insulin-related mechanisms are involved in CH pathophysiology, since it has been reported that eight of nine patients in active cluster period developed a typical CH attack during an insulin tolerance test (6). The patients had typical autonomic signs of hypoglycaemia before the attack and vasodilation due to hypoglycaemia was discussed as an underlying mechanism. Insulin seems to be of importance in migraine pathophysiology. In migraine, impaired insulin sensitivity has been found (36), as well as polymorphisms in the insulin receptor gene, suggesting an involvement of insulin regulation in the pathogenesis of migraine (37). To our knowledge, no such studies have been undertaken in CH.
Cortisol
Cortisol concentrations increased significantly during the night in both groups and the time of the maximum peak did not differ between CH patients in remission and controls, indicating a normal circadian cortisol rhythm in CH remission. We did not find any significant differences in nocturnal concentrations, maximum peak levels or minimum levels between CH patients and controls. Only one previous study has measured nocturnal cortisol in remission, and our results confirm these findings (4). However, basal morning concentrations have been found increased in remission, and corticotropine-releasing hormone test as well as insulin-induced hypoglycaemia have revealed blunted cortisol responses in CH remission (6).
Hormones and lipolysis
In a previous study we have shown diminished lipolysis in CH patients in remission between 02.00 and 06.00 h as well as a change of the nocturnal lipolysis rhythm (8). In CH patients, lipolysis decreased between 24.00 and 04.00 h, in contrast to healthy subjects, in whom lipolysis increases gradually during the night (38). Catecholamines have a pronounced effect on lipolysis. Catecholamines act on β-adrenergic receptors and stimulate lipolysis, predominantly by release of NA from peripheral sympathetic nerve endings, but also by circulating catecholamines from the sympatho-adrenal system (10–12). In the present study we found no differences in circulating nocturnal NA concentrations between CH patients and healthy subjects, but a disturbance of NA release from sympathetic nerve endings in adipose tissue cannot not be excluded. However, the stimulating effect of catecholamines is mainly acute and in a situation of overnight fasting, β-adrenergic stimulation might not be of importance (12).
Trace amines, such as tyramine, octopamine and synephrine, are chemically related to classical monoamines. Octopamine is derived from tyramine metabolism by oxidation through monoamine oxidase (MAO) activity (39). Since migraine patients treated with MAO inhibitors may exhibit headache after ingestion of tyramine-containing food, it has been proposed that trace amines are involved in the pathogenesis of migraine (40, 41). In accordance, MAO activity in platelets has been shown to be reduced in migraine as well as in CH patients (42) and it has also been shown that plasma as well as intraplatelet concentrations of octopamine and tyramine are elevated in both phases of CH as well as in migraine with and without aura (43, 44). Like catecholamines, octopamine influences adipose tissue metabolism. Octopamine directly stimulates lipolysis via activation of β3-adrenergic receptors, an effect that in humans is probably of minor importance, because the expression of β3-receptors in human adipose tissue is low. As a substrate of semicarbazide-sensitive aminooxidase activity in adipose tissue, octopamine has been shown in vitro to have an insulin-like effect as well, such as stimulation of glucose uptake and to some extent also inhibition of lipolysis (45, 46). It cannot be excluded, therefore, that elevated plasma concentrations of octopamine to some extent contribute to the reduced nocturnal lipolysis, previously demonstrated in CH (8). However, further studies are needed.
GH is also known to stimulate lipolysis, but in a slower manner. A given bolus dose of GH results, after a delay of about 2 h, in an increase of lipolysis (16), and suppression of the nocturnal rise in GH has been shown to influence lipolysis the following morning (47). GH stimulates lipolysis, at least in part, by improving the sensitivity to catecholamine action (15, 18). A blunted GH peak at 01.00 h, as was found in the present study, could result in reduced lipolysis later in the night. For that reason, the alteration in lipolysis concentrations and rhythm that was previously found might result from diminished GH secretion or dyssynchronization between GH and catecholamine secretion in CH remission.
Insulin is the major antilipolytic hormone. In the present study, insulin levels were unchanged over time and the temporal patterns did not differ between CH patients and controls. Thus, we have no indication of disturbed insulin regulation in CH in remission and it is not likely that the altered lipolysis is due to alterations in insulin secretion.
The effect of cortisol on lipolysis is not completely clarified and lipolytic as well as antilipolytic effects have been reported (17, 18). We found no differences in nocturnal cortisol secretion between CH patients and healthy subjects, indicating that cortisol is not involved in the altered lipolysis in CH remission.
In conclusion, the altered GH nocturnal pattern seen in CH patients in remission is a further indication of permanent hypothalamic disturbance in CH and might in part explain the altered nocturnal lipolysis previously found in CH remission.
Acknowledgements
This study was supported by grants from VR project K2005-72X-09941-14A, Pfizer AB, AFA forsakring, GlaxoSmithKline AB, Sweden and AstraZeneca AB, Sweden.
