Abstract
The purpose of this study was to assess the sensitivity of 5-HT1D receptors in patients with episodic cluster headache using sumatriptan as a pharmacological probe. The drug, a selective 5-HT1B/1D agonist, stimulates the secretion of growth hormone and inhibits the release of prolactin, adrenocorticotropic hormone (ACTH) and cortisol. These effects may be used to explore the function of serotonergic systems in vivo. We administered subcutaneous sumatriptan and placebo to 20 patients with cluster headache (10 in the active phase and 10 in the remission period) and to 12 controls. The sumatriptan-induced increase of growth hormone concentrations was significantly (P < 0.05) blunted in patients with active cluster headache. Prolactin and ACTH responses to the drug were significantly (P < 0.05) reduced in patients with cluster headache, both in the active and in the remission period. Our results suggest that cerebral serotonergic functions mediated by 5-HT1D receptors are altered in patients with episodic cluster headache.
Introduction
The aetiological and pathophysiological mechanisms of cluster headache (CH), one of the most severe primary headache syndromes, are still largely unknown (1). The signature feature of this disorder is its rhythmicity: the disease displays both a circadian periodicity in pain attacks and a circannual periodicity in cluster periods. The cyclic occurrence of the headache attacks suggested a dysfunction of the circadian timing system within the central nervous system (2, 3).
Several endocrine studies have shown alterations in the cyclic secretion of melatonin, cortisol, testosterone, gonadotrophins, prolactin, and thyrotropin, both in the active period and in the remission phase of the disease, suggesting evidence for hypothalamic involvement in CH (4–6). Recent studies with positron emission tomography and with voxel-based morphometry provided additional evidence for hypothalamic activation in the pathogenesis of CH attacks (7, 8).
To investigate further hypothalamic involvement in CH, we decided to use the neuroendocrine challenge paradigm, a strategy that provides a ‘window’ on central neurotransmitter function in vivo (9). A previous challenge study, performed with m-CPP, showed impaired central serotonergic function in patients with episodic CH (10). However, m-CPP is a drug characterized by a large variability in pharmacokinetic and pharmacodynamic parameters and it is doubtful whether it is a useful compound for challenge tests (11). Sumatriptan is a selective 5HT1B/1D receptor agonist effective in the treatment of CH attacks (12). Several studies in healthy volunteers have shown that the drug induces a significant increase in growth hormone (GH) plasmatic concentrations and a reduction of prolactin (PRL), adrenocorticotropic hormone (ACTH) and cortisol secretion (13–15). Studies in experimental animals have shown these effects are mediated through the activation of hypothalamic 5-HT1D receptors (16). Sumatriptan does not easily cross the blood–brain barrier but may reach hypothalamic nuclei trough circumventricular organs (17). The neuroendocrine effects induced by sumatriptan administration have been used as a pharmacological tool in several psychiatric and neurological diseases in order to explore cerebral serotonergic functions in vivo(18–22).
The purpose of our study was to assess the sensitivity of 5HT1D receptors in patients with episodic CH using subcutaneous sumatriptan as a pharmacological probe. We administered the drug and a placebo in CH patients, both in the active phase and in the remission period of the disease, to verify the hypothesis of an abnormal sensitivity of cerebral 5-HT1D receptors in the disease.
Methods
Subjects
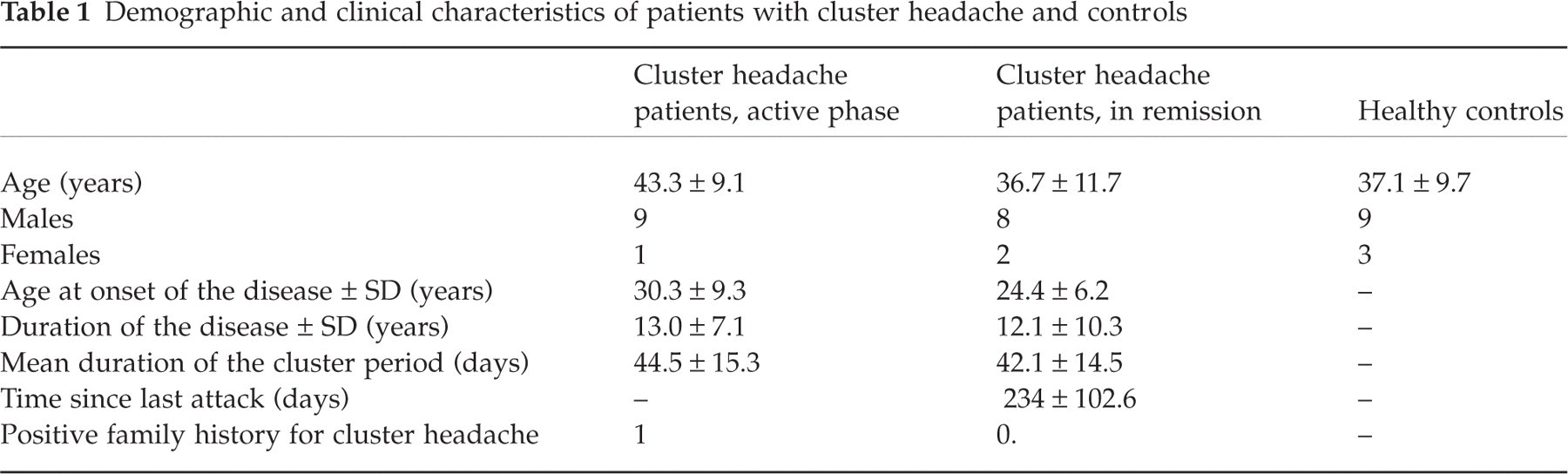
Twenty patients with episodic CH (three females, 17 males; mean age ± SD 40.0 ± 10.9 years) were recruited from the out-patient population at the Headache Centre of the University of Torino, Italy. The diagnosis of episodic CH was made according to the International Headache Society (IHS) criteria (23). Ten patients (one female, nine males; mean age ± SD 43.3 ± 9.1 years) were in the active cluster period while 10 patients (two females and eight males; mean age ± SD 36.7 ± 11.7 years) were in the remission period (at least 2 months after the last headache attack). Patients with mood or anxiety disorders were excluded after administration by a trained psychologist of the State-Trait Anxiety Inventory-X1, -X2 (STAI-X1 and STAI-X2) (24), and the Beck's Depression Inventory (BDI) (25) (cut-off limits STAI-X1 = 45 for women and 42 for men; STAI-X2 = 49 for women and 45 for men; BDI = 12 for men and 15 for women). Patients did not take any prophylactic drug for at least 4 weeks prior to testing. CH patients in the active phase were not allowed to use analgesic drugs for a 24-h period prior to drug administration. Six out of 10 patients in the active group and four out of 10 patients in the remission group had never used subcutaneous sumatriptan for treatment of CH.
Twelve healthy volunteers (three females, nine males; mean age ± SD 37.1 ± 9.7 years), matched for age, sex and phase of the menstrual cycle, served as controls. All subjects gave their informed consent to the study. Demographic and clinical characteristics of patients with CH and healthy controls are shown in Table 1.
Demographic and clinical characteristics of patients with cluster headache and controls
Procedures
The study was conducted at the Headache Centre of the University of Torino, Italy, according to a standardized protocol. Each subject underwent two tests, one with subcutaneous sumatriptan (6 mg) and the other with placebo (2 ml subcutaneous saline), in a double-blind fashion. In each subject the tests were separated by 3–6 days. Testing began at 09.00 h after overnight fasting. All subjects were confined to bed and were not allowed to eat or sleep. An indwelling intravenous catheter was inserted in a forearm vein and kept patent with a slow infusion of 0.9% NaCl. A 45-min period was allowed for relaxation. Two baseline blood samples (−15 and 0 min) were taken. Subsequent blood samples were taken at +15, +30, +45, +60 and +90 min after injection.
Assays
Blood samples were collected into sterile tubes. Tubes were immediately centrifuged at 4°C and the plasma was stored at −80°C until assayed. Plasma GH, PRL, ACTH and cortisol concentrations were measured using commercially available radioimmunoassay and immunoradiometric assay kits (HGH-CTK and PRL-CTK, Sorin, Saluggia, Italy; ACTH Allegro, Nichols Institute, San Juan Capistrano, CA, USA; CORT-CTK-125, Sorin). The sensitivity of GH, PRL, ACTH and cortisol assays were 0.15 µg/l, 0.49 µg/l, 1 pg/ml, and 0.5 ng/ml, respectively. The intra-assay and interassay coefficients of variation were 5.1% and 5.4% for GH, 2.5% and 7.7% for PRL, 3% and 7.8% for ACTH, and 3.8% and 5.7% for cortisol, respectively. All samples from each subject were analysed in the same assay.
Statistical analysis
Statistical evaluation was performed using SigmaStat, version 1.0 (Jandel Corp., San Rafael, CA, USA 1994). The hormonal responses to drug administration were evaluated calculating both the means of delta (Δ) values (point value − T0 value) and Δ peaks (maximum change from T0 value). For statistical analyses,
Results
Adverse events after sumatriptan injection were reported by both healthy controls (58.3%) and CH patients (65%). All the adverse events were described as mild in severity and were transient. The most frequently reported were heaviness and pressure on the chest (62.5%), warmth (37.5%) and dizziness (9.4%).
Table 2 shows both the absolute hormonal concentrations (±SEM) and the Δ AUC (area under the curve after subtraction of basal values) in patients and in healthy controls. Basal GH, PRL, ACTH and cortisol concentrations were not significantly different in controls and in patients with CH, either in the active phase or in the remission period.
Growth hormone (GH), prolactin (PRL), adrenocorticotropic hormone (ACTH) and cortisol (CORT) concentrations in patients with cluster headache and controls
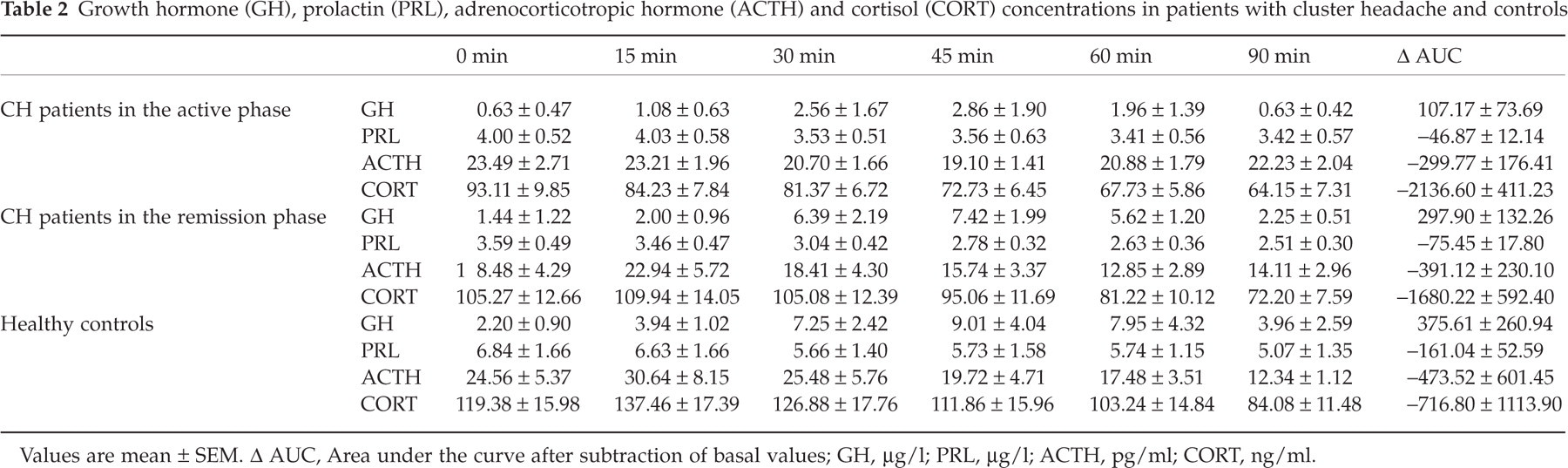
Values are mean ± SEM. Δ AUC, Area under the curve after subtraction of basal values; GH, µg/l; PRL, µg/l; ACTH, pg/ml; CORT, ng/ml.
Figure 1 shows the effects of sumatriptan administration on GH concentrations in controls and in patients with CH. The administration of sumatriptan induced an increase of GH concentrations both in controls and in patients with CH. However, in patients with active CH, the GH increase after sumatriptan administration was significantly blunted in comparison with controls (P < 0.05 at time +15 min and +30 min as Δ values, left of the figure; P < 0.05 as Δ peak, right of the figure) and with patients with CH in the remission period (P < 0.05 at time +45 min, +60 min and +90 min as Δ values, left of the figure; P < 0.05 as Δ peak, right of the figure).
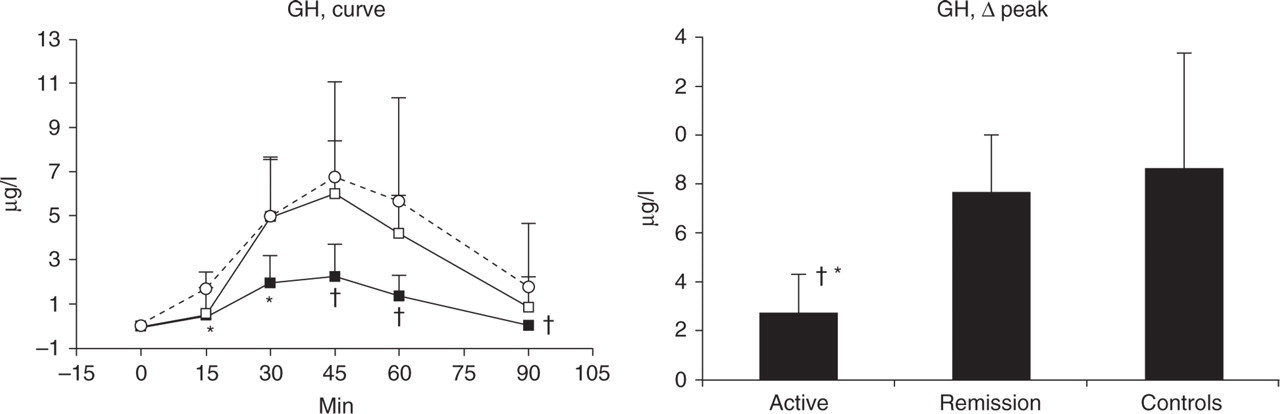
Mean changes in growth hormone (GH) secretion after sumatriptan (6 mg, s.c., at 0 min) in healthy controls and in patients with episodic cluster headache during the active phase and in remission. On the left, values are expressed as Δ values (▪, active; □, remission; ○, control), on the right as Δ peaks. Vertical bars indicate SEM. ∗P < 0.05 in comparison with controls; †P < 0.05 in comparison with cluster headache patients in the remission phase.
Figure 2 shows the effects of sumatriptan administration on PRL secretion. As expected, the drug induced a reduction of hormone plasma concentrations. The inhibition of PRL secretion, in comparison with controls, was significantly less marked both in patients with active CH (P < 0.05 at time +60 min as Δ values, left of the figure; P < 0.05 as Δ peak, right of the figure) and in patients with CH in the remission period (P < 0.05 as Δ peak, right of the figure).

Mean changes in prolactin (PRL) secretion after sumatriptan (6 mg, s.c., at 0 min) in healthy controls and in patients with episodic cluster headache during the active phase and in remission. On the left, values are expressed as Δ (▪, active; □, remission; ○, control), on the right as Δ peaks. Vertical bars indicate SEM. ∗P < 0.05 in comparison with controls.
Figure 3 shows the effects of sumatriptan administration on ACTH secretion. After an initial increase of ACTH concentrations, a progressive reduction of hormone secretion was observed. In patients with active CH the reduction of ACTH secretion was significantly reduced in comparison with both controls (P < 0.05 at time +90 min as Δ values, left of the figure) and with CH patients in the remission period (P < 0.05 at time 60 min and 90 min as Δ values, left of the figure). In patients with CH in the remission phase there was a significant reduction of ACTH secretion in comparison with controls (P < 0.05 at time +90 min as Δ values, left of the figure). The administration of sumatriptan induced a reduction in cortisol plasma concentrations. However, multiple comparison of controls and patients with CH showed no significant difference in the response to the challenge drug.

Mean changes in adrenocorticotropic hormone (ACTH) secretion after sumatriptan (6 mg, s.c., at 0 min) in healthy controls and in patients with episodic cluster headache during the active phase and in the remission period. On the left, values are expressed as Δ values (▪, active; □, remission; ○, control), on the right as Δ peaks. Vertical bars indicate SEM. ∗P < 0.05 in comparison with controls; †P < 0.05 in comparison with cluster headache patients in the remission phase.
No significant relation was found between the clinical characteristics of CH and the hormonal responses, expressed as Δ peaks. No significant difference in hormonal responses was found between CH patients who used sumatriptan for the first time and patients who used the drug in previous cluster periods.
Discussion
The aim of our study was to evaluate the sensitivity of cerebral 5-HT1D receptors in patients with episodic CH. In the acute phase of the disease, CH patients showed a significant impairment of GH, PRL and ACTH secretion after sumatriptan injection. During the remission phase, we observed the persistence of an abnormal secretion of both PRL and ACTH. Our data suggest the presence of a significant down-regulation of 5-HT1D receptors in both phases of the disease.
This is the first study to measure 5-HT1D receptor sensitivity in patients with CH using sumatriptan as a pharmacological probe, and the findings of this study must be considered preliminary. However, our data are in accord with the results of previous challenge studies that suggested an impairment of hypothalamic function in the disease. The thyroid-stimulating hormone (TSH) response to thyroid-releasing hormone (TRH) administration was significantly lower in patients with active CH in comparison with controls (26). Using both the insulin tolerance test and the ovine corticotrophin-releasing hormone test, Leone et al. (27, 28) found an altered responsiveness of hypothalamic pituitary–adrenal (HPA) axis in patients with CH, both in active cluster and in the remission phase of the disease. Our study confirms previous studies showing an impairment of both HPA axis and prolactin secretion in CH (4, 5) and provides new data concerning a derangement of the mechanisms regulating GH secretion. All these neuroendocrine findings are in agreement with recent neuroimaging studies that provided convincing data concerning hypothalamic involvement and activation in CH attacks (7, 8).
The main finding of this study is the demonstration of a selective impairment of cerebral 5-HT1D receptor-mediated functions in CH. A previous study with m-CPP (10) suggested impaired central serotonergic function in patients with active CH. However, m-CPP interacts both with several subtypes of serotonin receptors (5-HT2B, 5-HT2C, 5-HT1A) and with α2-adrenergic receptors. In contrast, sumatriptan is a highly selective 5-HT1B/1D receptor agonist and the neuroendocrine effects induced by this drug are strictly related to the activation of cerebral serotonergic systems.
It is well known that serotonergic neurones play an important but complex role in the regulation of pain within the central nervous system (29). 5-HT is a neurotransmitter both in the pathways descending from the brain stem to the spinal dorsal horn and in the ascending antinociceptive pathways (30, 31). The multiple effects of 5-HT on pain transmission are mediated through the activation of both 5-HT1 and 5-HT2 receptor subtypes (32). The serotonergic dysfunction found in patients with CH may explain reduced pain threshold found in these patients (33) In addition, it is of interest to note that lithium, a drug effective in the prophylaxis of CH, significantly increases the activity of the cerebral serotonergic systems (34, 35).
In conclusion, our study provides evidence of a selective involvement of cerebral serotonergic systems in CH patients, both in the active phase and in the remission period. Further studies are needed to evaluate whether this finding is of relevance to pathophysiology and prophylactic therapy of cluster headache.
Footnotes
Acknowledgements
This research was supported by a 2000 grant from M.U.R.S.T., Italy.
