Abstract
Efficacy of 5–day treatment with oral frovatriptan 2.5 mg/die for the prophylaxis of post-dural puncture headache (PDPH) was tested in 50 in-patients. A mild headache occurred in 7 (14%) patients for a total of 9 days (p < 0.01 vs. no-PDPH). Most episodes of PDPH occurred in the first days of treatment (only 1 patient had headache at dismissal): 5 patients had only 1 episode, while 2 had headache for 2 consecutive days. No other symptoms were recorded. Occurrence of PDPH in a subgroup of 6 (12%) patients previously submitted to a diagnostic lumbar puncture was also examined: 4 of them reported a PDPH on the previous lumbar puncture in absence of triptans. In only 1 of these 4 patients PDPH recurred under treatment with frovatriptan. In conclusion, our non-randomized open-label study suggests efficacy of oral frovatriptan for PDPH prevention. These results need to be confirmed in a randomized, controlled, double-blind study.
Introduction
Post-dural puncture headache (PDPH) occurs in up to 40% of patients undergoing diagnostic lumbar puncture (DLP), its incidence varying depending upon how the procedure is performed (e.g. replacement of the stylet or insertion plane of the bevel with the Quincke needle), the needle used (atraumatic vs. cutting Quincke and diameter) and the demographics of the patients (age and gender) (1, 2). Its origin has been related to the decrease in cerebrospinal fluid volume or pressure, or both, through the hole made by the spinal or epidural needle. This loss leads to PDPH due to shifts of intracranial contents and traction on pain-sensitive structures in the upright position, activation of adenosine receptors and consequent induction of arterial and venous dilation in the central nervous system (1, 3). Several measures have been proposed for treatment of PDPH, but very few of them have proved to be effective (e.g. epidural blood patch in up to 98% of patients), while most have not been shown to work with certainty (1).
Although a few case reports have suggested that subcutaneous injections of sumatriptan, a serotonin agonist acting on 5-HT1B/1D receptors and used for treatment of migraine, may be successfully employed for treatment of PDPH (4–6), a small controlled trial found no evidence of benefit from sumatriptan for the treatment of PDPH (7). Frovatriptan is an oral 5-HT1B/1D receptor agonist, used for the treatment and prophylaxis of migraine, whose high selectivity for cerebral vasculature, long elimination half-life and high persistence of therapeutic action make it a potential effective preventive for PDPH (8). It acts on extracerebral, intracranial arteries by inhibiting excessive dilation of these vessels occurring during migraine attacks and it is better tolerated than other triptans (9).
The present study aimed at assessing the efficacy of frovatriptan given orally as a prophylactic treatment of PDPH.
Methods
This was a non-randomized, open-label, non-placebo-controlled, pilot study. Eligible patients were considered those of either gender, aged 18–65 years and needing a DLP. Exclusion criteria included: (i) moderate or severe arterial hypertension or mild uncontrolled arterial hypertension; (ii) positive medical history of ischaemic heart disease, myocardial infarction, peripheral vascular disease, severe liver failure; (iii) treatment with ergot medications, other 5-HT agonists or monoamine oxidase inhibitors at the time of enrolment; (iv) known hypersensitivity to frovatriptan; and (v) pregnant women or those with childbearing potential but not practising an effective method of birth control. The study protocol was reviewed and approved by the Institutional Review Board and all patients provided written informed consent prior to inclusion.
All patients underwent a DLP according to procedures indicated by the American Academy of Neurology. Briefly, a 20-G Quincke needle was inserted in the dura with the bevel parallel to the dura fibres and stylet was replaced before needle withdrawal (10, 11).
Following the DLP patients were treated with frovatriptan 2.5 mg orally once daily for 5 days. Occurrence of PDPH was recorded through a patient's diary. PDPH was diagnosed according to the International Classification of Headache Disorders (ICHD)-II (12) as a headache (i) worsening within 15 min after sitting or standing and improving within 15 min after lying, with at least one symptom among neck stiffness, tinnitus, hypacusia, photophobia and nausea, (ii) developing within 5 days after dural puncture, and (iii) resolving either spontaneously within 1 week or within 48 h after effective treatment of the spinal fluid leak (usually by epidural blood patch). In addition to frovatriptan, patients could be treated conservatively (fluid hydration and bed rest), provided that no drugs interfering with study treatment were administered during the study (e.g. triptans, cortisone, analgesics, etc.).
Study efficacy variables were the occurrence (percentage of patients and number of days), intensity (mild, moderate or severe) and duration of PDPH and related symptoms assessed during the 5-day in-hospital treatment period following DLP. Differences in the percentages of patients with and without PDPH were assessed by χ2 test. Comparison of the number of days with and without PDPH was made by Student's t-test. P < 0.05 was taken as the minimum level of statistical significance.
Results
A total of 50 patients were enrolled. Their main demographic and clinical data are shown in Table 1. Most of the patients had suspected multiple sclerosis or myelitis or encephalitis at entry, these diagnoses being confirmed at final visit after cerebrospinal fluid examination (Table 1). No patients reported headache prior to the procedure. Symptoms associated with encephalitis were fever and ataxia, with no patient showing an altered mental status.
Demographic and clinical data of the study population

CNS, Central nervous system.
Only seven (14%) of the 50 patients studied reported PDPH. The rate of patients with PDPH was significantly (P < 0.01) lower than that without PDPH (Fig. 1a). In all cases PDPH was of mild intensity.
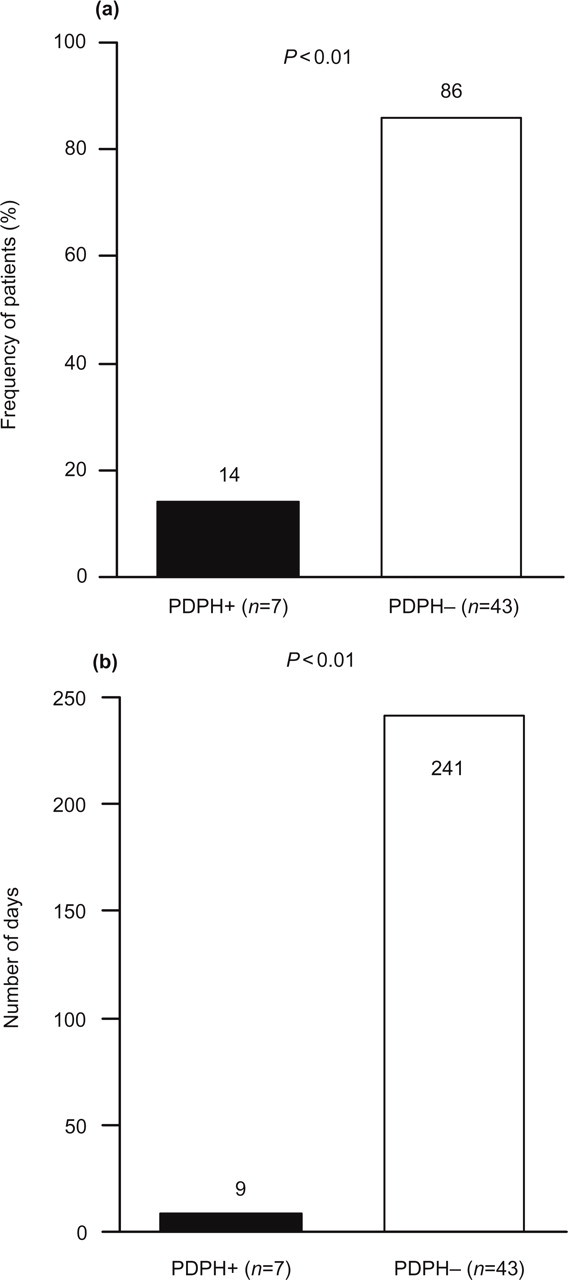
Frequency (%) of patients (a) and total number of days (b) with (+) or without (–) a postdural puncture headache (PDPH).
A total of 9 days of PDPH out of 250 days of treatment were recorded (P < 0.01 vs. number of days without PDPH) (Fig. 1b).
Most episodes of PDPH occurred in the first days of treatment, and only one patient still had headache at dismissal from the hospital. Of the seven patients reporting PDPH, five had only one episode, whereas two had headache for two consecutive days (Fig. 2). No symptoms except headache were reported.

Number of patients/day with a postdural puncture headache (PDPH) in the 5 days of study treatment with frovatriptan.
Occurrence of PDPH in a subgroup of six (12%) patients previously submitted to a DLP was also examined. These patients served as a sort of control group; four of them reported a PDPH on the previous lumbar puncture in absence of triptans. In only one of these four patients did PDPH recur under treatment with frovatriptan (P = NS).
Discusssion
Results of our study report the efficacy of oral frovatriptan at a dose of 2.5 mg for the prevention of PDPH. It also prevented symptoms related to PDPH. The drug reduced the risk but also the duration of PDPH, which occurred in only seven out of 50 patients and lasted only 1 day in the majority (71%) of them. Since headache usually occurs in up to 40% of patients following lumbar puncture (1, 2), our results demonstrate that prophylactic treatment with frovatriptan of patients undergoing DLP may reduce by more than half the chance of having a PDPH.
Although previous case reports of patients treated with subcutaneous sumatriptan seem to support the use of triptans in these patients for the treatment of the acute attack (4–6), there is no previous evidence of such a beneficial effect for the prevention of PDPH by oral formulations. Results achieved in our study with frovatriptan are encouraging. They add to previous evidence of effective prophylactic activity of frovatriptan observed in a different clinical setting (546 women with menstrually associated migraine) by a randomized, double-blind, placebo-controlled study (13). We postulate that the success of headache prevention with frovatriptan may be ascribed to its high affinity for cerebral artery 5-HT1B/1D receptors and to its long elimination half-life (26 h), resulting in a consistent and long duration of action (9).
Few other results need further discussion. First, although we followed currently suggested procedures for DLP (10), we used a larger needle than in the previous randomized controlled study with other triptans (20 G vs. 25 G), potentially increasing the risk of PDPH and reducing the drug preventive efficacy (1). However, we recorded a limited number of cases of PDPH. Second, as expected, PDPH occurred in the first 3 days following lumbar puncture, which strengthens the importance of prophylactic treatment being commenced as soon as possible after the diagnostic procedure. Three, frovatriptan was effective in the oral form, with no need for subcutaneous administration as for other triptans. Four, no adverse events or accompanying symptoms except headache were reported by any patient.
We acknowledge that our study has some limitations, related to its non-randomized, open-label, non-placebo-controlled design, including a relatively limited number of subjects. Although a control group was lacking, we could analyse data from a very small subgroup of patients with a positive history of PDPH. In these patients recurrence of PDPH was low (one out of four patients). It should also be acknowledged that our sample size was much greater than that of the only available randomized controlled study with a triptan (7) or other treatments (14, 15). Finally, one may argue that, even if frovatriptan seems to be an effective preventive treatment for PDPH, we could have reduced the risk of PDPH simply by using an atraumatic lumbar puncture needle such as the Sprotte, which has been proven in randomized trials to reduce the incidence of PDPH to 3–15%, depending upon the study and the diameter of the needle (16–18). However, use of such a needle requires greater attention and expertise by doctors and is thus not easy to apply in all hospitals and situations. It is also true that, even in case of use of a smaller needle, risk of PDPH is not completely abolished and thus frovatriptan may be considered as an effective prophylactic treatment.
In conclusion, our results support the hypothesis that prophylaxis with oral frovatriptan may successfully reduce the risk of PDPH, avoiding the use of blood patch or other aggressive, invasive therapies. Adequately powered, double-blind, randomized, placebo-controlled studies are needed in order to demonstrate definitely the efficacy and safety of oral frovatriptan for the prevention of PDPH.
Acknowledgements
The authors thank Istituto Lusofarmaco d'Italia for the support given to this study. Results of this study have been presented at the 12th International Congress of the International Headache Society held in Kyoto (Japan), 9–12 October 2005.
