Abstract
In order to explore possible interictal brainstem dysfunctions in migraine, we have studied the R2 component of the nociceptive specific blink reflex (nBR) after conditioning by supraorbital or index finger stimuli in 14 untreated migraine without aura patients (MO) between attacks and in 15 healthy volunteers. We determined the R2 recovery curve at increasing inter-stimulus intervals between 50 and 600 ms. The nBR was conditioned by a paired supraorbital stimulus and, in another session, by an ipsilateral electrical shock delivered to the index finger. The R2 nBR recovery curves were normal in MO patients for both the supraorbital and peripheral conditioning. These results do not favour persistent interictal sensitization in the spinal trigeminal sensory system. They also suggest that the control exerted by descending brainstem pathways on medullary R2 interneurones is normal in migraine between attacks.
Introduction
Current knowledge on migraine pathophysiology assigns an important role to different neural structures, such as sensory cortices (1), the brain stem (periaqueductal grey, locus coeruleus, raphe nuclei) (2, 3), and both peripheral and central portions of the trigeminovascular system (4). It is generally accepted that the migraine headache is associated with activation of the latter system. Cutaneous allodynia and decreased pain thresholds in the V1 territory on the headache side (4), as well as the finding of an increased area of the nociception specific blink reflex R2 (nBR) (5) during migraine attacks, are in favour of an ictal sensitization in the spinal trigeminal pain system. There is also circumstantial evidence that such sensitization may persist between attacks (6).
We found recently that the interictal habituation deficit of the nBR described by Katsarava et al. (7) in migraineurs, can also be demonstrated during long lasting stimulations with a custom-built electrode activating Aδ fibres (8). This habituation deficit is in line with similar findings for cortical evoked potentials (see Schoenen et al. (9), for a review). As far as the nBR is concerned, it could be related to peripheral or central sensitization in the trigeminal system.
The R2 blink reflex component is known to undergo both segmental and heterosegmental influences via interneuronal networks (10). The R2 response can be suppressed by a preceding conditioning stimulation of the supraorbital nerve and its recovery curve after paired stimuli is thought to reflect the excitability of the trigemino-facial circuit (10, 11). Moreover, the R2 response can also be suppressed by a preceding peripheral stimulus (12), probably via activation of inhibiting reticular nuclei (13). In a study of supraorbital conditioning of the R2 blink reflex elicited by conventional electrical stimulation, there was a significant difference between migraineurs and healthy volunteers for two interstimulus intervals (ISIs) (14). By contrast, Aktekin et al. (15) found normal recovery curves in migraineurs with a similar protocol. We have previously shown that cluster headache patients, during a bout, have a faster recovery curve of the classical R2 after both supraorbital and peripheral conditioning, which we interpreted as reflecting hyperexcitability of the trigeminal reflex pathway (16).
In order to further analyse the interictal brainstem dysfunction in migraine, we thought it would be of interest to study in migraine the recovery curves of the nBR R2 component after a conditioning supraorbital or peripheral stimulus delivered at varying inter-stimulus intervals. We reasoned that a faster recovery curve might be expected in the case of a persistent state of sensitization at the segmental or suprasegmental levels and compared therefore migraineurs without aura between attacks (MO) and healthy volunteers (HV).
Materials and methods
Subjects
We recorded 14 migraine without aura patients (MO; ICHD-II (17) code 1.1; mean age, 30.7 ± 9.3; nine women and five men; attack frequency, 3.5 ± 2.5). We included only data from MO patients who had an interval of at least 3 days from a migraine attack. They were compared with 15 healthy volunteers of comparable age and gender distribution (HV; mean age, 29.5 ± 9.8; eight women and seven men) without personal or familial history of migraine and devoid of any detectable medical condition. Subjects taking medications on a regular basis except for the contraceptive pill were excluded, as were smokers. Caffeine or alcohol consumption less than 12 h before the recording session was not allowed.
All participants signed an informed consent in accordance with the declaration of Helsinki and the study was approved by the Ethics Committee of the Faculty of Medicine, University of Liège.
Nociception specific blink reflex
Subjects were seated relaxed on a comfortable chair in an illuminated room and asked to stay with eyes open. During the entire duration of the recording session we checked at regular intervals that their level of attention and vigilance remained normal.
The nociception specific blink reflex (nBR) was elicited according to the methods described by Kaube et al. (18) and by Di Clemente et al. (8). A custom-built planar concentric electrode providing a high current density at low intensities via a central cathode of 1 mm2 (total area, 20 mm2) was used to stimulate the supraorbital region with monopolar square pulses of 0.3 ms duration. Pain threshold was determined on the right side of the forehead with an ascending and a descending sequence of 0.2 mA intensity steps. We chose to limit the study to a right-sided stimulation in order to avoid a lengthy, uncomfortable procedure and because none of the migraineurs studied had a higher attack prevalence on one side. The stimulus intensity was set at 1.5 times the individual pain threshold (1.9 ± 0.6 mA for HV, and 1.6 ± 0.8 mA for MO patients). Stimuli were delivered at pseudorandom 20–21 s inter-stimulus intervals. Recording electrodes were placed infraorbitally (active) over the orbicularis oculi muscle and latero-orbitally (reference) on both sides. A ground electrode was placed on the nose. The signal was amplified by CEDTM 1902 preamplifiers and recorded by a CEDTM 1401 device (Cambridge Electronic Design Ltd, Cambridge, UK). All recordings were averaged off-line using the SignalTM software package version 2.15 (Cambridge Electronic Design Ltd). Five nBR responses occurring over a 150-ms post-stimulus epoch, were rectified and averaged to determine the R2 area under the curve (AUC) and onset latency.
Homotopic conditioning of the nBR R2 was obtained by delivering paired stimuli to the supraorbital nerve at inter-stimulus intervals (ISI) from 50 to 200 ms increased in steps of 50 ms and from 200 ms to 600 ms in steps of 100 ms.
In another set of recordings randomly preceding or following the previous one, heterotopic conditioning of the nBR R2 was studied by an ipsilateral index finger stimulation preceding the supraorbital stimulus at the same ISIs as described above. The index finger stimulus was delivered through ring electrodes to phalanxes I and II on the same side as the supraorbital stimulation. Its intensity was set at five times the sensory threshold, it was not painful and it never elicited a blink reflex itself.
For each conditioning session, five nBR R2 responses were rectified and averaged as described above.
The nBR R2 recovery curves were computed off-line by calculating the percentage R2 AUC change at each ISI respective to that of the unconditioned response (‘test response’) considered as 100%.
Statistical analyses
One-way analysis of variance (
Results
The mean values of stimulation intensity, latency and response area of the unconditioned nBR R2 are shown in Table 1. We found no difference between migraine patients and healthy volunteers for stimulus intensity (F[1,27] = 1.248, P = 0.274) and thus pain threshold, onset latency (F[1,27] = 0.357, P = 0.555) or AUC (ipsilateral, F[1,27] = 0.927, P = 0.344; contralateral, F[1,27] = 0.808, P = 0.377) of the nBR test R2.
Unconditioned (‘test response’) nBR R2 parameters in healthy volunteers and in migraine patients (mean

Similarly, the nBR R2 recovery curves did not differ significantly between migraineurs and healthy controls, neither after supraorbital conditioning (ISIs∗group: ipsilateral, d.f. = 7, F = 0.290, P = 0.950; contralateral, d.f. = 7, F = 0.981, P = 0.470) nor after conditioning by an index finger stimulus (ISIs∗group: ipsilateral, d.f. = 7, F = 0.707, P = 0.667; contralateral, d.f. = 7, F = 0.550, P = 0.787) (Figure 1).

The nBR recovery curve after conditioning by (a) a paired supraorbital or (b) a preceding electrical stimulus applied at the index finger. Ordinate: conditioned R2 response expressed as a percentage of the unconditioned (test) response. Abscissa: intervals in ms between conditioning and subsequent stimuli.
Discussion
We found no significant difference between healthy volunteers and migraineurs for the unconditioned nBR R2 response with respect to onset latency or AUC, although, ipsilaterally to the stimulation, the latter tended to be larger in MO than in HV. Also, regarding the main objective of our study, the nBR R2 recovery curves after supraorbital or index finger conditioning were similar in both groups of subjects.
The neural network that mediates the polysynaptic R2 component of the blink reflex involves nucleus trigeminalis caudalis, excitatory interneurones of the bulbo-pontine lateral reticular formation and motor neurones in pontine facial nuclei innervating orbicularis oculi muscles (10). The R2 reflex component is modulated by segmental and suprasegmental mechanisms. The paired supraorbital stimulus activates segmental inhibitory interneurones, whereas the index finger stimulus inhibits the excitatory R2 interneurones via spino-reticular afferents to brainstem nuclei such as nucleus reticularis gigantocellularis and nucleus raphe magnus (12, 13, 16, 19). The results of our study suggest that both the segmentally mediated inhibition and the spino-reticular inhibition of the bulbar R2 interneurones are normal in migraine between attacks (Figure 2).
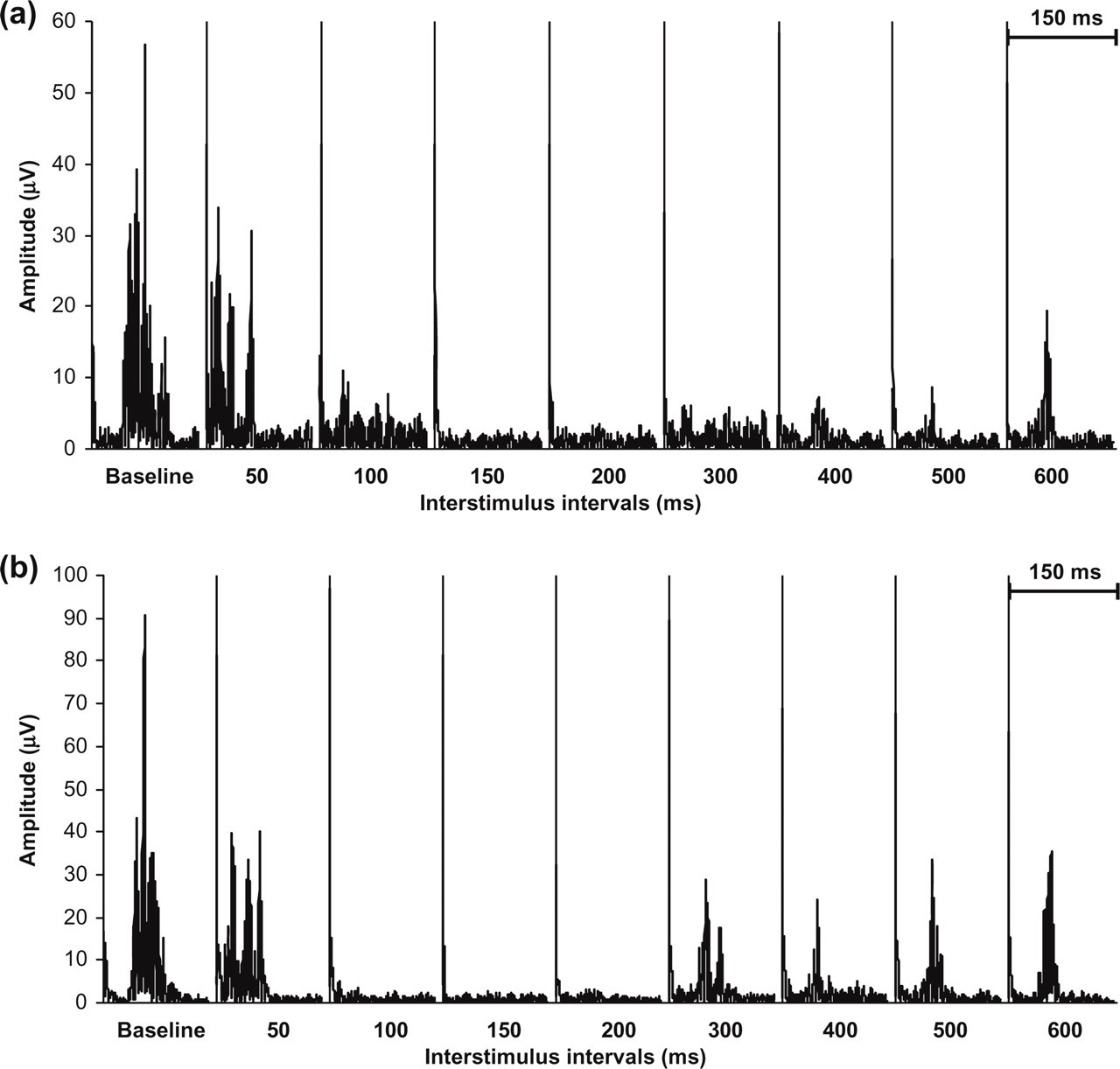
Illustrative traces (healthy volunteer) of nBR R2 recovery at increasing inter-stimulus intervals after conditioning by (a) a paired supraorbital or (b) a preceding index finger stimulation.
Our findings are in line with those of Aktekin et al. (20), but differ from those reported by de Tommaso et al. (14). In these studies, however, the blink reflex was elicited with a classical non-selective stimulation electrode and it has been demonstrated that the responses obtained with the concentric electrode have in part a different neuronal substrate. Using a comparable stimulation electrode to ours, which activates mainly αδ fibers, Katsarava et al. (7) found normal latencies and areas under the curve for the nBR R2 in migraineurs interictally, but a reduced habituation, as we did in this and a previous study (8). By contrast, in a study of the corneal reflex, known to depend less on the input from Aβ fibres, a lower reflex threshold was found between attacks in migraineurs compared with HV, which led to the suggestion that sensitization of the trigeminal system may persist interictally (6). During the migraine attack, indeed, the nBR R2 amplitude was shown to be increased, which suggests ictal sensitization of the reflex pathway (21). A similar change was found in a patient with a pontine carvernoma involving the left nucleus raphe magnus of the brainstem, who developed de novo strictly right-sided migraine attacks (22). The dissimilarity between studies could be due to the methodological differences or to differences in the timing of recordings with respect to the last or next migraine attack.
It is known from studies using supraorbital stimulation with a conventional electrical stimulation (14, 15) or with laser stimulation (23) that after supraorbital or peripheral pre-stimuli the inhibited R2 test response recovers progressively from an ISI of 300 ms, but that the baseline amplitude is not reached before ISIs of 1–1.5 s. We have limited our study to a maximal ISI of 600 ms for several reasons. R2 inhibition is maximal with ISIs comprised between 50 and 600 ms. In cluster headache patients within a bout, we have previously found a significantly faster recovery curve of the R2 blink reflex evoked with a standard stimulation electrode as soon as the 300 ms ISI and for all subsequent ISIs up to 600 ms (16). In a study of the classical R2 blink reflex (14) a significant difference between migraine patients and normal controls was reported in the recovery curve after supraorbital conditioning for short ISIs (250 and 400 ms), but not for ISIs of 1 s or more. Moreover, we have increased the ISI by rather small 50 or 100 ms steps compared with other studies (23). Studying longer ISIs would thus have led to a substantial number of supplementary stimuli and discomfort for the subjects. It must therefore be taken into account that our conclusions about the absence of difference in nBR recovery curves between migraineurs and healthy volunteers is valid only for an ISI up to 600 ms. A study of longer ISIs has yet to be performed, although the likelihood that it may come to a different conclusion seems meagre.
Another trigeminal reflex mediated by brainstem (inhibitory) interneurones can be suppressed by preceding index finger stimulation: the second temporalis muscle exteroceptive suppression (ES2) (24). In migraineurs, we found that the peripheral conditioning of ES2 was within normal limits, which contrasted with tension-type headache patients in whom it was markedly enhanced (25).
Taken together, these results suggest that in migraine the excitatory/inhibitory interneurones and their trigeminal target motor neurones have a normal excitability between attacks. The same applies to the spino-reticular pathways that modulate these neurones. Their excitability, however, is likely to change dramatically during an attack, which underlines the interest in ictal studies of the same electrophysiological tests.
