Abstract
This study analyses the differences in the relative cross-sectional area (rCSA) of several cervical extensor muscles, assessed by magnetic resonance imaging (MRI), between patients with chronic tension-type headache (CTTH) and healthy controls. MRI of the cervical spine was performed on 15 CTTH females and 15 matched controls. The rCSA values for the rectus capitis posterior minor (RCPmin), rectus capitis posterior major (RCPmaj), semispinalis capitis and splenius capitis muscles were measured from axial T1-weighted images using axial MR slices aligned parallel to the C2/3 intervertebral disc. A headache diary was kept for 4 weeks in order to substantiate the diagnosis and record the pain history. CTTH patients showed reduced rCSA for both RCPmin and RCPmaj muscles (P < 0.01), but not for semispinalis and splenius capitis muscles, compared with controls. Headache intensity, duration or frequency and rCSA in both RCPmin and RCPmaj muscles were negatively correlated (P < 0.05): the greater the headache intensity, duration or frequency, the smaller the rCSA in the RCPmin and RCPmaj muscles. CTTH patients demonstrate muscle atrophy of the rectus capitis posterior muscles. Whether this selective muscle atrophy is a primary or secondary phenomenon remains unclear. In any case, muscle atrophy could possibly account for a reduction of proprioceptive output from these muscles, and thus contribute to the perpetuation of pain.
Introduction
Tension-type headache (TTH) is the most common type of headache, with 1-year prevalence rates of 38.3% for the episodic form and 2.2% for the chronic form (CTTH) (1). Despite some advances, the pathogenesis of this primary headache is poorly understood. TTH is a prototypical headache in which the cervical musculature might play an important aetiological or pathogenic role (2). Several authors have postulated that TTH could be the manifestation of referred pain from several cervical muscles, e.g. suboccipital, semispinalis capitis, splenius capitis, upper trapezius, etc (3–5). In fact, in a previous study we found that the referred pain evoked from the suboccipital muscles in CTTH patients was related to headache clinical parameters (6). However, the role of the extensor cervical muscles in CTTH remains unclear.
Magnetic resonance imaging (MRI) can be regarded as the gold standard for muscle imaging and it has been used to measure and identify changes in muscle morphology, e.g. reductions in muscle cross-sectional area (CSA) or fatty infiltration, in patients with back (7) and neck pain (8, 9). It has been proposed that MRI analysis may be of value to detect muscle changes in the cervical muscles in chronic pain patients. McPartland et al. qualitatively found marked atrophy in the suboccipital muscles, i.e. rectus capitis posterior minor and major muscles, in chronic neck pain patients compared with healthy subjects (10). Elliott et al. have recently published preliminary data of normal morphometry of the cervical extensor muscles in a cohort of healthy females within a defined age range (18–45 years) (11). Objective observations of measurable differences in muscle size may have diagnostic and therapeutic implications for rehabilitation of patients with chronic pain.
Based on previous observations, it seems that the cervical extensor musculature, particularly the suboccipital muscles, might play an important role in the genesis of TTH. However, we are unaware of other works that have analysed the morphometrical features of the cervical extensor muscles in CTTH. Our aim in this study was to analyse the differences in the morphometry of four cranio-cervical extensor muscles (i.e. rectus capitis posterior minor, rectus capitis posterior major, semispinalis capitis and splenius capitis muscles) between CTTH patients and healthy controls. In addition, we aimed to assess the relationship between muscle size and several clinical variables concerning the intensity and the temporal profile of headache.
Materials and methods
Subjects
A total of 15 CTTH females, aged 24–56 years (mean 43 ± 12 years) and 15 healthy females, aged 25–56 (mean 40 ± 10 years) participated in this study. Patients with CTTH were recruited from the Neurology Department of Fundación Hospital Alcorcón, whereas healthy controls were recruited from the hospital staff. All subjects were right-handed. CTTH was diagnosed by an experienced neurologist according to the criteria of the International Headache Society (IHS). Key elements of headache history were ascertained, including family history, headache features, temporal profile and present and past medications. To be included in this study, patients had to describe all the characteristics typical of TTH: bilateral location, pressing or tightening (non-pulsating) pain, mild or moderate intensity [no more than 7 on a 10-cm visual analogue scale (VAS)] and lack of aggravation during routine physical activity. In addition, none of them reported photophobia, phonophobia, vomiting or evident nausea during headache. All cases had evolved from episodic TTH, but had to have headache on at least 15 days per month for >3 months to be considered chronic. A headache diary was kept for 4 weeks in order to substantiate the diagnosis (13). None of the patients fulfilled current criteria for other primary headaches. Otherwise, there were no indications of any secondary headache by history, physical and neurological examination, and appropriate tests including neuroimaging (computed tomography or MRI). By the time the study was completed, patients did not received either physical therapy or prophylactic drug therapy. They reported to have used only analgesics or anti-inflammatory drugs when pain was most intense, but medication-overuse headache as defined by the IHS (12) was ruled out in all cases. The health status of all patients was clinically stable, without any evidence of other concomitant chronic disease.
Healthy subjects were not considered if they had a previous history of headache or neck pain, dizziness or vestibular symptoms, a motor vehicle accident, inflammatory joint disease, cervical radiculopathy, myopathy or any chronic disease. In relation to the MRI scan, no patients or controls were included if they suffered from claustrophobia, had a pacemaker, aneurysm clip, or any small implanted metal hardware or wires that could move or be affected by a magnetic field. Furthermore, participants were not considered if they were pregnant or if, in the absence of an effective form of contraception, they could possibly have conceived since the first day of their last normal menstrual period.
This study was supervised by the Departments of Physical Therapy, Neurology and Radiology of Universidad Rey Juan Carlos and Fundación Hospital Alcorcón, and was also approved by the local human research committee. All subjects signed an informed consent prior to inclusion.
Magnetic resonance procedure
Conventional T1-weighed spin-echo images were chosen as they provide contrasted pictures which allow the distinction of the borders for fat and muscle (14). In this study we analysed the following cervical extensor muscles: rectus capitis posterior minor (RCPmin), rectus capitis posterior major (RCPmaj), semispinalis capitis and splenius capitis. Accurate CSA (mm2) assessment required the creation of a region of interest (ROI) for each muscle. ROIs comprising each muscle were manually depicted on both sides with MRIcro (http://www.mricro.com) (Rorden, Nottingham, UK) software (15).
All images were acquired on a HORIZON LX 1.5-T magnet (General Electric, Milwaukee, WI, USA). Measurement parameters were: slices = 28, with a slice thickness of 4 mm, FOV read = 200 mm, TR = 516.7 ms, TE = 19.0 ms, matrix size = 256 × 256, acquisition time TA = 11:20 s.
The axial scan of the cervical spine was planned from the sagittal scout images. Axial MR slides aligned parallel to the C2–3 intervertebral disc were taken from the base of the occiput through the upper portion of the C5 vertebral body. Since CSA images of the cervical extensor muscles above and below the C2–3 level could include a slight measurement error in both patients and controls, relative CSAs (rCSAs) are reported.
The rCSAs of the neck extensor muscles were calculated from the ROIs that were created on the axial images at certain cervical levels. This reliable procedure has been previously described in detail by Elliott et al. (11, 14). Axial scans at C1 and C2 levels were used to measure the rCSA for the RCPmin and the RCPmaj muscles on both sides, respectively (Fig. 1a,b). The rCSA measures for the superficial extensor muscles, i.e. semispinalis capitis and splenius capitis, were obtained at two segments (C3 and C4) on the axial slices crossing the most cephalad portion of each corresponding vertebral body (Fig. 1c). The deep cervical extensors, i.e. the multifidus and semispinalis cervicis, were not measured due to the large amount of periarticular fatty tissue surrounding these muscles and the lack of identifiable deep muscle fibres at the C2–3 segmental levels (16).
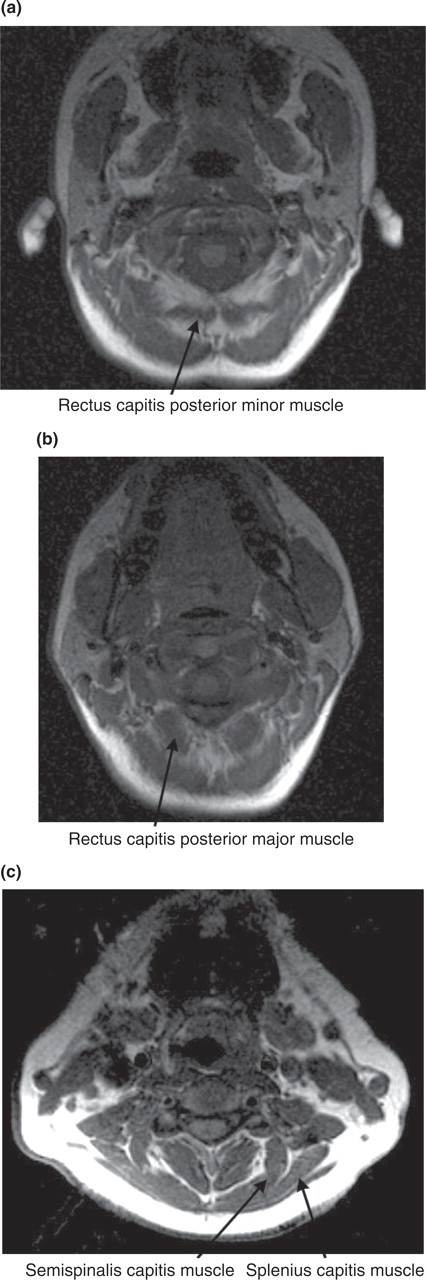
Posterior cervical extensor muscles.
In order to ensure a good degree of reliability for our measurements, two experienced radiologists identified and measured all rCSAs separately. Both of them were blinded to the subjects' condition. The interexaminer agreement was tested for each muscle on each side by means of the repeated measures
Demographic and clinical assessment
Apart from age, sex and handedness, the body mass index (BMI) and activity levels were recorded from each subject to determine potential influences on muscle size. Weight (kg) was measured with a standard electronic weight scale (Seca, Madrid, Spain) and a wall-mounted measuring tape was used to assess the height (m); the BMI was then calculated (kg/m2) and for analysis participants were placed into one of two groups: (i) normal, BMI ≤ 24.9 kg/m2, or (ii) overweight, BMI ≥ 25.0 kg/m2, in accordance with Gallagher et al (17). The activity level of each subject was classified according to the Lipid Research Clinics Questionnaire (LRCQ) as low active, moderately active, or highly active (18).
A headache diary was given to CTTH patients. Each patient registered on this diary the daily headache intensity, on a 10-cm horizontal VAS (range: 0, no pain, to 10, maximum pain) (19), the headache duration (in hours per day) and the days with headache. This headache diary was kept for 4 weeks and was then returned to one assessor, who calculated the following variables: (i) headache intensity, which was calculated from the mean of the VAS of the days with headache; (ii) headache frequency (days per week); and (iii) headache duration (hours per day).
MRI scans of CTTH patients were obtained on days in which headache intensity was <3 points on a 10-cm horizontal VAS.
Statistical analysis
Data were analysed with the SPSS statistical package (v. 13.0; SPSS Inc., Chicago, IL, USA). Group demographic and anthropometric data are presented as mean ±
Results
All CTTH patients described bilateral head pain, of pressing (n = 13) or tightening (n = 10) quality, and mild or moderate intensity (no more than 7 on a 10-cm VAS); in 13 of them (87%) the pain extended bilaterally to the neck. Headache history ranged from 3 to 20 years (mean 10 ± 7 years). Headache frequency ranged from 4 to 6 days per week (mean 4.8;
The interexaminer repeatability of MRI measurements (ICC) ranged from 0.8 to 0.95 in patients and from 0.85 to 0.98 in controls, thus indicating an excellent interexaminer agreement. The standard errors of measurement (SEM) fell between 2.3 and 7.6 mm2 in patients and between 2.1 and 5.8 mm2 in controls.
The average values of the rCSA measures for the RCPmin muscles (C1 level), the RCPmajor muscles (C2 level) and both semispinalis capitis and splenius capitis at C3 and C4 levels are presented in Table 1. No significant differences were found between right and left rCSAs in either patients or controls, except for the splenius capitis muscle at C3 in the control group (P = 0.01). Those three subjects who reported moderate activity levels showed larger rCSAs than those with lower activity levels, but the differences did not reach significance. Finally, neither age (P > 0.2) nor BMI (P > 0.3) influenced rCSA in any of the analysed muscles.
Differences in cross-sectional areas between chronic tension-type headache (CTTH) patients and healthy controls

Values are expressed as mean (
NS, Non-significant.
Significant differences between CTTH and controls (unpaired Student's t-test).
Significant difference compared with the left side (paired Student's t-test).
CTTH patients showed reduced rCSA for both RCPmin and RCPmaj muscles compared with controls (P < 0.01; Figs 2 and 3). On the other hand, rCSAs for semispinalis capitis and splenius capitis at C3 and C4 levels in patients did not differ significantly from those in controls (P > 0.3). Table 1 summarizes the differences in rCSA between patients and controls for each of the analysed muscles.

Rectus capitis posterior minor in chronic tension-type headache patients and healthy subjects.

Rectus capitis posterior major in chronic tension-type headache patients and healthy controls.
Within the CTTH group, there were significant negative correlations between headache intensity, duration or frequency and rCSA for both RCPmin and RCPmaj muscles: the greater the headache intensity, duration or frequency, the smaller the rCSA in the two muscles. Table 2 details the correlations between headache clinical parametersand rCSA of both rectus capitis posterior muscles.
Correlations between headache intensity, frequency, duration and cross-sectional areas in the rectus capitis posterior minor and major muscles within the chronic tension-type headache group

r s, Spearman's rho test; NS, non-significant; VAS, visual analogue scale (0, no pain, to 10, maximum pain).
Statistically significant.
Discussion
This study for the first time provides evidence that both RCPmin and RCPmaj muscles show reduced rCSA, i.e. muscle atrophy, in CTTH female patients compared with healthy controls in the age range 24–56 years, whereas other cervical extensor muscles, i.e. semispinalis capitis and splenius capitis, appear not to be significantly different between patients and controls. Furthermore, headache intensity, frequency and duration were greater in those CTTH patients who showed more reduced rCSA in RCPmin and RCPmaj muscles.
According to our results, patients with CTTH exhibit muscle reactions (i.e. muscle atrophy) that are typically found in musculoskeletal disorders. Indeed, a reduced rCSA in deep muscles has been previously reported in some musculoskeletal disorders such as chronic neck pain (9, 10) or low back pain (20). Whether the selective atrophy of RCPmin and RCPmaj is a primary or secondary phenomenon to CTTH remains unclear. Future longitudinal studies would be required in order to elucidate if these muscle changes occur as a consequence of chronic headache.
Whether it represents a primary or secondary phenomenon, muscle atrophy could act as a perpetuating factor for chronic pain. McPartland et al. have reported that the atrophy of RCPmin and RCPmaj muscles is related to a comparative decrease in standing balance in patients with neck pain (10). Peck et al. found that the suboccipital muscles had greater concentration of muscle spindles (36 spindles/g for RCPmin; 30.5 spindles/g for RCPmaj) in comparison with other cervical extensor muscles (e.g. 7.6 spindles/g in the splenius capitis muscle) (21). Such high density of muscle spindles in both rectus capitis posterior muscles suggests that they act as ‘proprioceptive monitors’ of the upper cervical spine. Proprioceptive signals stemming from the muscles conveyed by large-diameter Aβ-fibres may serve as a ‘gate’ that blocks nociceptive (C-fibre) transmission into the spinal cord and higher centres of the central nervous system (22). Muscle atrophy could account for a reduction of proprioceptive output from these muscles, and such reduced proprioceptive input may conceivably facilitate the transmission of impulses from wide dynamic range nociceptors (22, 23). In addition, a dysfunction of descending inhibitory pathways has been recently found in CTTH patients (24). Both peripheral and central mechanisms, i.e. facilitation of impulses from peripheral nociceptors, and dysfunction of central inhibitory pathways, are probably involved in the maintenance of pain.
In contrast to the rectus capitis posterior muscles, the most superficial cervical extensor muscles, i.e. semispinalis capitis and splenius capitis, did not show reduced rCSA. One possible explanation could be that muscles with a greater concentration of muscle spindles, such as the RCPmin and RCPmaj, might be more sensitive to muscle atrophy than muscles with lesser concentration of muscle spindles, i.e. semispinalis capitis and splenius capitis. Our results parallel those previously reported in patients with low back pain, in whom muscle atrophy was found only in lumbar multifidus muscles, i.e. muscles of the lumbar spine with the greatest density of muscle spindles (20).
Our study has some limitations. First, only subjects with CTTH were included. Hence, our results cannot be extrapolated to episodic TTH. A potential role of morphological change in the suboccipital muscles might only account for the chronic form of TTH. In addition, other types of headache, such as those attributed to cervical spine disorders (i.e. cervicogenic headache), could also reflect muscle atrophy in these muscles. We have found bilateral muscle atrophy (a musculoskeletal component) in a sample of patients with bilateral headache fulfilling criteria of CTTH. Future studies should address the differences in suboccipital muscle atrophy between TTH and other headaches, including cervicogenic headache. The second limitation was the sample size. In order to extrapolate our results to the general headache population, it would be necessary to repeat the same procedure with a greater number of subjects. Finally, we recognize a methodological limitation relating to the orientation of MRI slices. We incorporated slices parallel to the C2–C3 intervertebral disc, as this orientation provided a perpendicular view of the cervical extensor muscles and had been employed in previous studies (9–11). However, the layout of all the posterior cervical muscles is often not equal across subjects, and the direction of the RCPmin muscle is more tangential than that of the RCPmaj, semispinalis capitis and splenius capitis muscles. However, it is unlikely that a different orientation would have affected our results to a significant degree, since both patients and controls were assessed with the same procedure.
In conclusion, RCPmin and RCPmaj muscles showed reduced rCSA in CTTH patients compared with healthy controls. No differences were observed in rCSA for semispinalis and splenius capitis muscles across the two groups. Headache intensity, frequency and duration were greater in those CTTH patients with more reduced rCSA in both RCPmin and RCPmaj muscles. Headache clinical parameters did not seem to be influenced by the most superficial muscles, i.e. semispinalis and splenius capitis, within the CTTH group. Muscle size was not influenced by the factors of age, activity levels or BMI in either group. Our results point to a musculoskeletal component in CTTH, with the presence of muscle atrophy in the deep cervical extensor muscles. Whether this selective muscle atrophy is a primary or secondary phenomenon to CTTH remains unclear. Further work investigating MRI muscle morphometry in patients suffering from different headache disorders is warranted in order to understand the pathophysiological basis for these muscular changes and their potential clinical and therapeutic implications.
