Abstract
Migraine is a primary headache disorder which involves both genetic and environmental components. Since angiotensin-converting enzyme (ACE) and matrix metalloproteinase (MMP) share the same homology, we investigated whether the MMP-3 and ACE I/D gene variants are involved in migraine risk and whether the ACE variant might act in combination with the MMP-3 genetic variant in patients with migraine. This is the first study to evaluate the association between MMP-3 and ACE polymorphisms, and migraine. Genotypes were determined by polymerase chain reaction. The frequencies of 5A5A genotypes of the MMP-3 and D allele of ACE were significantly elevated, but II genotypes of the ACE and 6A allele of MMP-3 significantly decreased in all patients. The combined DD/5A5A and ID/5A5A genotypes increased the risk of migraine. Individuals who were homozygous for the deletion (D) allele showed increased ACE activity. Subjects with the 5A5A genotype and/or D allele or with the combined DD/5A5A or ID/5A5A might be more susceptible to migraine development. In contrast, subjects with the II and/or 6A6A genotypes may be protected from migraine development. The greater activity of the 5A5A and DD genotypes might result in vascular reactivity that is more pronounced in migraine. Taken together, our data suggest that numerous genes may influence ACE activity. Discovery of new genes might better clarify the pathogenesis of migraine and open an avenue to therapeutic strategies against migraine.
Introduction
Migraine is a highly prevalent primary headache which affects approximately 10% of the white population worldwide (1, 2). It is a complex neurovascular disorder and is likely to be influenced by multiple genes, some of which may be capable of causing vascular changes leading to disease onset. There have been numerous case–control studies reporting positive associations of candidate gene polymorphisms and migraine (3–5).
Angiotensin-converting enzyme (ACE), a key part of the renin–angiotensin system, is expressed in vascular endothelial cells and can regulate blood pressure by mediating potent vasoconstrictors and dilators (6). This makes ACE a plausible candidate gene for involvement in migraine pathophysiology (7). The ACE (I/D) polymorphism has been identified as a risk factor for migraine (8). A polymorphism in the ACE gene has been described, consisting of an insertion or deletion (I/D) of a 287-bp fragment in intron 16 (9).
ACE is a bivalent dipeptidyl carboxyl metalloproteinase. This enzyme originates from a large evolutionary family of metallopeptidases, the majority of which, ACE included, contain zinc and employ zinc binding motif at their active site. Other enzymes in this family structurally similar to ACE include the interstitial collagens, members of the matrix metalloproteinase (MMP) family (10, 11). The extracellular matrix (ECM) in the vascular wall provides an essential supporting, structural framework necessary for the structural and functional properties of the vascular wall. ECM macromolecules are synthesized by the three vascular cell types: intimal endothelial cells, medial smooth muscle cells and adventitial fibroblast (12). MMPs are responsible for degradation of ECM in a variety of physiological and pathological processes (13). All of the cell types that exist in the central nervous system are potential sources of MMPs. In vitro, neurons, astrocytes, microglia and oligodendrocytes express various MMP family members (14, 15). MMP-3 (stromelysin), an important member of the MMP family, is able to degrade collagen types II, V, IX and X and other ECM proteins (16). The functional promoter polymorphism of MMP-3 has been associated with some diseases (17, 18). A common variant in the promoter region of human MMP-3 gene with one allele has a run of adenosines (5A) and the other has six adenosines (6A) (19).
Given that ACE and the MMPs share ancestry, structural homology and mechanism of inhibition, it is conceivable that drugs designed to inhibit ACE may have inhibitory activity against the MMPs. Indeed, recent in vitro and in vivo studies have confirmed the inhibition of mammalian MMPs by ACE inhibitors (20–23).
Since ACE and MMP share the same homology, we investigated whether the MMP-3 and ACE I/D gene variants are involved in migraine risk and whether the ACE variant might act in combination with the MMP-3 genetic variant in patients with migraine.
Materials and methods
Subjects
This study consisted of 59 patients suffering from migraine with aura (MA), 109 from migraine without aura (MoA) and 10 from basilar migraine and two from complicated migraine (Table 1). The patients and controls were recruited from the western part of Turkey. Participants were diagnosed with typical migraine by a detailed questionnaire and a clinical neurologist in accordance with International Headache Society (IHS) criteria (24). The control group was also interviewed and subjected to a questionnaire by specialized neurologists. Subjects receiving ACE inhibitors or patients who had non-migraine headaches that could not be distinguished from migraine were excluded from the study. Two hundred and ten normal healthy volunteers composed the control group, who had not suffered from migraine or tension-type headache. The Ethics Committee of Haydarpasa Numune Education and Research Hospital approved this research and all participants gave their informed consent.
Demographic and clinical characteristics of migraine patients and controls

P < 0.05 compared with healthy controls.
P < 0.01 compared with healthy controls.
Mean ± SD.
Determination of ACE activity
ACE activity was measured on an Hitachi 902 auto analyser (Roche Diagnostics, Basel, Switzerland) using reagents, standards, controls and calibrators provided by the manufacturer of the Sigma Diagnostics ACE kit. ACE activity in the samples was determined by comparing the sample reaction rate to that obtained with the ACE calibrator. ACE activity was expressed in U/l. The results were given as mean ± SD (25).
Determination of genotypes
Genomic DNA was extracted from white blood cells using a salting-out protocol (26). Intron 16 of the ACE gene was amplified by polymerase chain reaction (PCR) according to the method of Rigat et al. (9) using 5′-CTGGAGACCACTCCCATCCTTTCT-3′ as the sense primer and 5′-GATGTGGCCATCACATTCGTCAGAT-3′ as the antisense primer. To avoid mistyping, a second independent PCR amplification was performed using a primer pair which recognizes the insertion-specific sequence (I allele).
The insertion/deletion (−1171 5A→6A) polymorphisms in the promoter region of MMP-3 were studied by PCR with the use of mutagenic primers (27). A 130-bp fragment was amplified from genomic DNA using the following primers. The forward primer (−1201 to −1172, mismatch at nucleotide −1173) and reverse primer (−1072 to −1101) were 5′-GGTTCTCCATTCCTTTGATGGGGGGAAAgA-3′ and 5′-CTTCCTGGAATTCACATCAC-TGCCACCACT-3′, respectively. Thus, a recognition site for Tth111I enzyme (GACN/NNGTC) is created in the case of a 5A allele. The PCR product was digested with Tth111I for 3 h at 65°C. All PCR products were separated on 2% agarose gels with ethidium bromide staining and visualized under ultraviolet light. Two independent individuals assessed genotypes, blinding control genotypes to those of the assay. All genotype discrepancies were rectified or excluded from data analysis.
Statistical analysis
Analysis of data was performed using the computer software SPSS for windows (version 12; SPSS Inc., Chicago, IL, USA). Frequencies of genotype and allele distributions were assessed by χ2 test. ACE activity was presented as mean ± SD. Comparison of ACE activity among genotypes and study groups was analysed by non-parametric Kruskal–Wallis test. Non-parametric Kruskal–Wallis analyses were also used to test for differences in migraine clinical characteristics (i.e. frequency of attack, duration of disease and response to drug) among genotypes. Multivariate logistic regression analysis was performed to examine the possibility of combined effects of the ACE and MMP genes on migraine susceptibility.
Results
Table 1 shows the demographic characteristic of subjects. Controls and patients were adjusted for age. As expected, our patients were predominantly female. Patients were divided into subtypes according to IHS criteria. The frequencies of genotypes were according to Hardy–Weinberg equilibrium (Table 2). We observed no significant differences in the distribution of genotypes and alleles of ACE and MMP-3 among subtypes of migraine patients compared with controls. In contrast, when genotypes of all patients (without dividing into subgroups) were compared with those of controls, the frequency of the 5A5A genotype (P < 0.001) of MMP-3 and D allele (P < 0.01) of ACE was significantly elevated, but II genotype (P < 0.001) of ACE and the 6A allele (P < 0.01) of MMP-3 significantly decreased in all patients. In the total patient group, the frequency of combined genotypes of DD/5A5A (P < 0.05) and ID/5A5A (P < 0.01) was significantly higher, but that of II/6A6A (P < 0.05) was significantly lower compared with the control group (Table 3). ACE and MMP-3 genotypes were not correlated with frequency of migraine attacks, duration of disease or response to drugs in all migraineurs. ACE activity in serum was highest in DD, intermediate in ID and lowest in II genotypes in patients as well as in controls (P < 0.001) (Fig. 1). The mean activity of ACE was significantly lower in patients with MoA and MA than in controls (P < 0.01). One limitation of our study is that we could not measure the activity of MMP-3.

Comparison of angiotensin-converting enzyme (ACE) activity with ACE genotype in all groups. Mean activity of ACE expressed as U/l.
Distributions of genotype and allele frequencies of angiotensin-converting enzyme (ACE) and matrix metalloproteinase (MMP)-3 polymorphisms in patients and controls
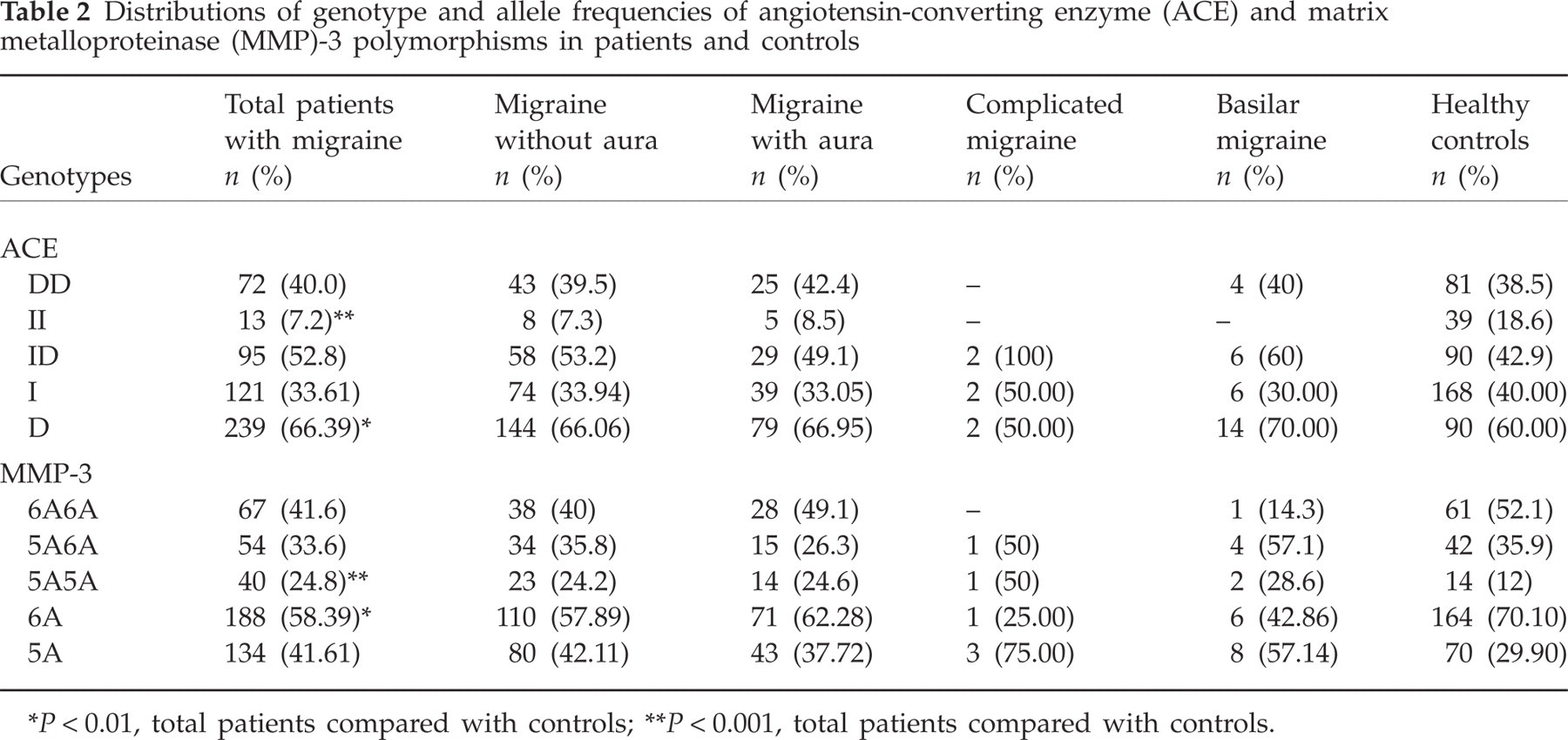
P < 0.01, total patients compared with controls;
P < 0.001, total patients compared with controls.
Combined genotypes of angiotensin-converting enzyme and matrix metalloproteinase-3 polymorphism in patients and controls

P < 0.05, total patients compared with controls;
P < 0.01, total patients compared with controls.
Discussion
Migraine is considered to be a polygenic disorder with a number of susceptibility genes having a minor but nonetheless significant impact on susceptibility (28, 29). In the present study, MMP-3 and ACE I/D polymorphisms were correlated with migraine. In previous ACE I/D polymorphism studies, there has been inconsistency between investigations. Some recent studies have indicated an association between DD genotype or D allele and migraine, but others have not (8, 30). As for MMP-3 polymorphism, to the best our knowledge there is no study that shows an association of MMP-3 polymorphism with migraine, so this is the first to evaluate the association between MMP-3 and ACE polymorphisms, and migraine.
Association analysis of the migraine subtypes separately was not meaningful due to the small numbers in each group. On the other hand, when patients were not divided into subtypes, the findings were statistically significant. Although he DD genotype was not associated with migraine susceptibility, the D allele seems to be a risk factor for the development of migraine. ACE activity was associated with ACE genotypes. Subjects with the DD genotype had higher ACE activity than subjects with either ID or II genotypes in all groups. Our findings are consistent with those reported by Riga B et al. (31). Unexpectedly, we found reduced ACE activity in patients compared with controls. This finding suggests that migraine is a complex disease involving multiple genes of moderate or small effect. Circulatory levels of ACE are partly under genetic control. It has been shown that more than one gene influences ACE levels (32). I/D polymorphism has been estimated to explain up to 47% of the variance in circulating ACE levels (31). Given that I/D polymorphism is an intronic Alu sequence, a direct influence on circulating ACE levels is not readily apparent. I/D polymorphism is therefore considered to be a genetic marker in linkage disequilibrium with a functional variant influencing ACE level. The association between I/D and ACE levels has been described in Whites, but more recent studies in African populations have shown that other variants of the ACE gene are more closely associated with circulating ACE levels than the I/D polymorphism (33–35). A transition at (nt) 22982, in the sequence AF118569 or 31958, has been found to show the largest phenotypic differences between genotypes (33, 35, 36). Although all markers tested in Europeans showed significant differences in ACE levels between each genotype (probably due to linkage disequilibrium between genotypes), the most marked difference in ACE levels in both Afro-Caribbean and European subjects was found between genotypes at A22982G. Absolute linkage disequilibrium between I/D and 22982 has been shown for White populations (37) and a strong haplotype association has been shown in African populations (33, 35). The I allele at I/D has been shown to be in linkage disequilibrium with the A allele at A22982G and the D with the G (37). Scott et al. (32) have also shown that 22982 polymorphism is more strongly associated with the variation in ACE levels than genotype at I/D. In the present study, reduced ACE levels might result from the other variants of the ACE and 22982 genes or different genes which have not yet been associated with ACE activity. Our data must be interpreted with care because the number of our patients was low, due to including residents from a restricted region. Our study sheds light which future studies may use to explain the relationship between ACE and migraine.
ACE has been reported to show altered activity in patients with neurological disease. Kehoe et al. (38) have suggested that decreased ACE activity may influence susceptibility to Alzheimer's disease by a mechanism involving β-amyloid metabolism. Patients with moderate degrees of senile dementia of the Alzheimer's type and comparably demented patients with Parkinson's disease or progressive supranuclear palsy exhibited mean levels of ACE activity that were decreased by 41, 27 and 53%, respectively, compared with the mean level in an age- and sex-matched group of neurologically intact individuals (39). These results raise the possibility that ACE activity may be an index of neuronal dysfunction in certain central neurodegenerative disorders.
The reduced ACE levels found in the present study do not argue against the important prophylactic effects of ACE inhibitors and angiotensin II receptor blocker on migraine (40). ACE inhibitors have various pharmacological effects that may be relevant in migraine. In addition to blocking the conversion of angiotensin I to angiotensin II, they also alter sympathetic activity, inhibit free radical activity, increase prostacyclin synthesis and block the degradation of bradykinin, encephalin and substance P, all important mechanisms in the modulation of pain. Bender (41) has reported successful treatment of migraine patients with an ACE inhibitor. After this study, lisinopril, an ACE inhibitor, was shown to have a clinically important prophylactic effect in migraine (42). The angiotensin II receptor blocker, candesartan, has also provided effective migraine prophylaxis in a randomized controlled trial (43). These trials suggest that the renin–angiotensin–aldosterone system must be involved, at least in part, in the pathogenesis of migraine.
We have shown the combined effect of both ACE and MMP-3 genetic variants on migraine disease. The combined DD/5A5A and I/D 5A5A genotypes increased the risk of migraine (odds ratio 2.876, P = 0.002) compared with those who possess neither the MMP-5A5A nor ACE DD genotypes. The overall risk was greater than with either of these genotypes alone. Lea and colleagues (44) have shown the combined action of ACE I/D and MTHFR C677T genetic variants on migraine in Whites. Cortical spreading depression (CSD) implicated in neurovascular disorders such as stroke, head trauma and migraine has been shown to activate and up-regulate MMP-9 which is activated by MMP-3. The same study has also suggested that CSD may augment MMP up-regulation and vascular permeability changes in migraine (45). In the total patient group, the frequency of 5A5A genotype distribution was significantly higher than that of the 5A6A and 6A6A genotypes. There are only two studies of MMP-3 polymorphisms in neurodegenerative disease. One, conducted by Saarela et al. (46), showed the relation of the 5A5A genotype with Alzheimer's disease. The 5A genotype has been demonstrated to have a higher promoter activity than 6A. Indeed, increased MMP activity has been shown to cause direct cellular damage in disease of the peripheral and central nervous systems (47–49). The DD combined with the 5A5A genotype seems a risk factor for migraine. Patients carrying both the D allele and the 5A5A genotype may have a greater risk than those carrying the 6A allele and II genotype because of the higher activity of these genotypes. This higher activity might result in vascular reactivity in patients with migraine. In contrast, we speculate that subjects with the II genotype or 6A allele might be protected against migraine development because of their lower activity.
Recent reports have demonstrated that ACE inhibitors and angiotensin receptor blockers also have effects on MMP (50, 51). The precise mechanisms underlying the beneficial action of ACE inhibitors on ECM remodelling are unclear. Natural and synthetic inhibitors of MMPs might bind the zinc moiety within the enzyme's active site, which is similar to the mechanism seen with ACE inhibition by ACE inhibitors, such as captopril. It has ben suggested that angiotensin II mediates up-regulation of MMP-9 through nuclear factor-κB activation in cardiomyocytes (52). These studies suggested that MMP and ACE might share the common mechanisms of inhibition and that these enzymes might have role in migraine pathophysiology. In particular, these findings may be clinically important given the effectiveness of ACE inhibitors as a migraine prophylactic.
The three-dimensional organization of ECM molecules synthesized during fetal development is optimal for the functions of the vessel wall. However, early in life the vessel wall is subject to injury (lipid deposition, hypoxia, enzyme secretion and reactive oxygen species production during inflammatory processes, etc.) (12). The expression and activity of MMPs are controlled at many levels, including the transcriptional level, where MMP expression is regulated by growth factors, cytokines and free radicals (53). The altered balance of inflammatory mediators and proteolytic fragments of the ECM under inflammatory conditions changes the levels of MMP required for normal tissue homeostasis and remodelling (54). Kimura et al. (55) have observed the elevated expression of MMP-3 and -20 in cultured granulose cells by bradykinin treatment. It has has been shown in another study that bradykinin enhanced pro-MMP-9 expression.
The bulk of biochemical evidence has suggested that pain in migraine is caused by blood vessels that are dilated and sensitized by circulating pain-producing substances, e.g. bradykinin, serotonin and histamine (sterile inflammation) (56). ACE inactivates bradykinin, substance P and neurokinin A, which are thought to play important roles in the pathogenesis of inflammatory diseases (57). In contrast, there are some contradictory results. Substance P is suggested to be of the neurotransmitters in the ‘trigeminovascular theory’. Few studies have investigated the relationship between the ACE genotype and substance P. Arinami et al. (58) have reported higher substance P levels in brain contents with the D/D genotype of the ACE gene, and this is the opposite tendency, as might be expected. The exact mechanism of the relationship between substance P and ACE genotype remains unknown. The alteration of ACE activity due to the I/D polymorphism may result in changed levels of neurotransmitters and vulnerability to cranial vascular activity. Although the ACE gene is a fundamental determinant of bradykinin peptide level, compensatory mechanisms attenuate the effect of modest changes in ACE gene expression on the levels of these peptides. Identification of these compensatory mechanisms may provide new candidate genes for investigation in humans (59).
In conclusion, we hypothesize that subjects with the 5A5A genotype and/or D allele or with the combined DD/5A5A or ID/5A5A might be more susceptible to migraine development. In contrast, subjects with the II and/or 6A6A genotypes might be protected from migraine development. The higher activities of 5A5A and DD genotypes might result in vascular reactivity that is more pronounced in migraine. Taken together, our data suggest that numerous genes might influence ACE activity. Discovery of new genes might better clarify the pathogenesis of migraine and open up new avenues for developing new therapeutic strategies against migraine.
Acknowledgements
This study was supported by the Research Fund of Istanbul University, Project No. UDP-496/24052005.
