Abstract
Retinal migraine is usually characterized by attacks of fully reversible monocular visual loss associated with migraine headache. Herein we summarize the clinical features and prognosis of 46 patients (six new cases and 40 from the literature) with retinal migraine based upon the International Classification of Headache Disorders-2 (ICHD-2) criteria. In our review, retinal migraine is most common in women in the second to third decade of life. Contrary to ICHD-2 criteria, most have a history of migraine with aura. In the typical attack monocular visual features consist of partial or complete visual loss lasting <1 h, ipsilateral to the headache. Nearly half of reported cases with recurrent transient monocular visual loss subsequently experienced permanent monocular visual loss. Although the ICHD-2 diagnostic criteria for retinal migraine require reversible visual loss, our findings suggest that irreversible visual loss is part of the retinal migraine spectrum, perhaps representing an ocular form of migrainous infarction. Based on this observation, the authors recommend migraine prophylactic treatment in an attempt to prevent permanent visual loss, even if attacks are infrequent. We also propose a revision to the ICHD-2 diagnostic criteria for retinal migraine.
Introduction
Retinal migraine is an uncommon cause of transient monocular visual loss. Since the first description by Galezowski (1) in 1882, there have been scattered case reports of monocular visual impairment associated with migraine. The 1988 International Classification of Headache Disorders (ICHD) defined criteria for retinal migraine, which were revised in the 2004 edition (ICHD-2) (2). ICHD-2 criteria for retinal migraine require at least two attacks of fully reversible monocular positive and/or negative visual phenomena associated with a headache which fulfills criteria for migraine without aura (Table 1). Herein, we report six new cases of retinal migraine and review the 40 prior cases that are based on ICHD-2 criteria for retinal migraine (but including both migraine with and without aura) (3–25). We describe its demographic and clinical features, including progression to irreversible monocular visual loss, and propose revised criteria and nomenclature for this entity.
2004 International Headache Society criteria for the diagnosis of retinal migraine (2)

Methods
We identified six new patients with retinal migraine treated at the Montefiore Headache Center from 1994 to 2005 (four were previously presented in abstract form and noted in a review article) (26, 27). Patients had a present or prior history of migraine associated with reversible monocular visual disturbances. In addition, we conducted a systematic search of the English medical literature via Medline using the keywords ‘retinal’, ‘ocular’, ‘monocular scotoma’,‘monocular blindness’, with ‘headache’ and ‘migraine’. Reviewing references in the articles found as well as in textbooks identified additional reports. To be included in this study, patients had to fulfill the ICHD-2 criterion of fully reversible monocular visual phenomena occurring during or within 60 min of an attack of migraine. Histories were also reviewed to determine if ICHD-2 criteria for migraine without aura, migraine with aura, or probable migraine were met. The vast majority of cases were reported before the ICHD-2 criteria were published. Diagnostic work-ups to exclude alternative causes of monocular visual loss were variable. We included patients with incomplete evaluations of monocular visual loss only when the episodes occurred solely in the context of migraine headaches and when other diseases did not occur during follow-up. Reports of transient monocular visual loss that were not temporally associated with an attack of migraine headache, were potentially attributable to other causes or appeared in languages other than English were not included in this review.
A number of patients with retinal migraine experienced transient monocular visual loss and later in their course developed permanent monocular visual loss. Based on this finding, the authors divided the clinical patterns of retinal migraine into transient monocular visual loss only (transient group) and transient monocular visual loss followed by permanent monocular visual loss (permanent group).
Results
We present six new cases of retinal migraine and then summarize the 40 previous reports in the literature.
Case reports
Patient 1
A 42-year-old woman described an 18-year history of migraine without aura associated with recurrent bouts of monocular visual impairment. The headaches were severe, pulsating and left-sided. Associated features included photophobia, phonophobia, osmophobia, nausea, vomiting and ipsilateral nasal congestion. Attacks occurred six times monthly, lasting 24–72 h. One-third of attacks began with transient spells of monocular visual impairment consisting of black spots and flashing lights ipsilateral to the headache. The visual phenomena always began in the periphery of the temporal quadrant of her left eye and expanded to engulf the entire field of that eye in 2–3 min. Complete visual loss of the left eye lasted for 5 min, fully resolved, and was immediately followed by her migraine headache. The patient confirmed the monocular nature of her attacks by alternately covering each eye during the attacks. There was a strong paternal family history of migraine.
Her general medical, ophthalmological and neurological examinations were normal. Magnetic resonance imaging (MRI) of the brain was unremarkable, as were extensive haematological studies. The patient was treated with a gradually escalated dose of nortriptyline to 75 mg at night. At this dose, the patient had complete cessation of her recurrent bouts of monocular visual impairment as well as a significant reduction of headaches (one per month). After 8 months of follow-up her visual acuity and fields remained normal.
Patient 2
A 35-year-old male neurologist described a 15-year history of migraine with aura associated with recurrent bouts of monocular visual impairment. The pain was severe and throbbing, located in the temples and associated with photophobia, phonophobia, osmophobia, nausea and visual scintillations in both eyes. Attacks occurred two to three times monthly, lasting 24–72 h. Once or twice per year the patient developed left monocular visual loss and left-sided headache. Visual loss lasted for 5 min, fully resolved and was immediately followed by a left-sided headache. Alternately covering each eye during attacks confirmed that visual features were confined to the left eye. There was a strong family history of migraine with aura.
His general medical, ophthalmological and neurological examinations were normal. Investigations included a computed tomographic (CT) angiogram of the brain, MRI and magnetic resonance angiogram (MRA) of the brain and extensive haematological tests; all were within normal limits. Treatment with flunarizine 10 mg and topirimate 100 mg was followed by complete cessation of headaches but had the untoward effect of weight gain and fatigue, respectively. The medications were changed to magnesium 400 mg twice daily and riboflavin 200 mg twice daily with a reduction in headache frequency (one per 6 weeks) and cessation of retinal migraine attacks. After 6 months of follow-up, visual acuity and fields were preserved.
Patient 3
A 33-year-old woman described a 4-year history of migraine with and without aura. Her headaches began above the left temple, lasted 4–5 h and occurred on average once every 3 months. Associated features included phonophobia and nausea. Occasionally, her headaches were accompanied by a visual aura manifested by an enlarging pattern of coloured squares, which always began 1 h before the headache in the periphery of her left eye and engulfed the left homonymous visual field. Five days before presentation the patient experienced daily attacks of her otherwise typical migraine headache preceded by recurrent bouts of monocular visual impairment ipsilateral to the headache. Visual loss resolved after 1 h and was immediately followed by her migraine headache. The patient confirmed that the visual changes were exclusively in her left eye by alternately covering each eye during attacks. There was no family history of migraine. There was a past history of hyperthyroidism. The patient smoked cigarettes but was not taking an oral contraceptive pill.
She was evaluated by her neurologist during one of the headaches associated with visual blurring of the left eye. The general physical and neurological examinations were normal. Visual acuity was 20/50 in the left eye and 20/20 in the right eye. The visual fields were full to confrontation. Funduscopic examination was normal. The patient's visual symptom cleared completely in 1 h and repeat ophthalmological examination demonstrated a visual acuity of 20/20 in her left eye. Investigations included MRI and MRA studies of the brain, as well as extensive haematological tests; all were within normal limits. She was treated with one aspirin 325-mg tablet daily. With the exception of a 7-day period of headaches in which she experienced four bouts of monocular visual impairment, her vision was fully preserved as of 10 months of follow-up.
Patient 4
A 44-year-old woman described a 14-year history of migraine without aura. The headaches began in the right nuchal and temple regions, typically lasted 1–2 days and occurred on average once to twice per month in association with her menses. Associated features included photophobia, phonophobia, osmophobia, nausea, vomiting, facial pallor and dysarthria. One year before presentation, the patient experienced one to two attacks per week of her otherwise typical migraine headache associated with recurrent bouts of monocular visual loss ipsilateral to the headache. Complete blindness of the right eye always began 30 min into the headache. The uniocular visual loss lasted the entire duration of the headache, ranging from 8 h to 3 days, and then fully resolved. Alternately covering each eye during attacks confirmed that the visual changes were limited to the right eye. There was a strong family history of migraine. Past history included asthma and abnormal uterine bleeding. The patient drank alcoholic beverages occasionally but denied cigarette and illicit drug use.
Her general medical, ophthalmological and neurological examinations were normal. Investigations included a CT scan of the brain, MRI and MRA of the brain, MRA of the neck, echocardiography, and extensive haematological tests; all were within normal limits. The patient was treated with topirimate 300 mg daily with complete cessation of her recurrent bouts of monocular visual loss as well as a reduction of headaches (one per month).
Patient 5
An 18-year-old woman described a 10-year history of migraine with visual, sensory, motor and dysphasic auras. Within the past 3 years, the headaches had increased in frequency, occurring three times per week. The pain was throbbing and stabbing, of severe intensity, and affected the right or left hemicranium for 4–6 h. Associated features included photophobia, phonophobia, osmophobia, nausea, vomiting, allodynia, difficulty with concentration, lightheadedness and vertigo. More than two-thirds of her headaches were accompanied by some form or combination of aura symptoms. Ten percent of these auras were manifested as brief spells of visual impairment consisting of greying out or blurring of one eye on the side of the headache. Complete monocular visual loss lasted for 30 min, fully resolved, and was shortly followed by her migraine headache. The patient confirmed the monocular nature of her attacks by alternately covering each eye during the attacks.
The vast majority of her remaining aura symptoms consisted of other visual, sensory, motor or language deficits. Visual auras manifested as stationary or flickering spots of colour in both eyes, loss of peripheral vision bilaterally, vertical diplopia, or what she described as ‘trails of previous vision’, in which the image of an object persisted after it was removed from her visual field (visual perseveration). Sensory auras consisted of unilateral facial paraesthesias that were either ipsilateral or contralateral to the headache and perioral numbness. Less commonly, the patient experienced motor symptoms such as ‘claw-like’ contractions of her fingers and toes or a heavy feeling in one or both legs. During some attacks, the patient also experienced receptive aphasia.
One month before presentation the patient experienced two attacks of her otherwise typical migraine headache (preceded by a visual aura of spots before both eyes, loss of peripheral vision bilaterally, unilateral facial paraesthesias and perioral numbness) associated with recurrent bouts of witnessed seizures. The seizures were preceded by a copper taste in the mouth and were characterized by tonic-clonic activity of all limbs, with urinary incontinence. Each seizure lasted approximately 30 s, recurred seven to eight times over 1.5 h duration and was followed by postictal confusion lasting 3 min.
Her medications included metoprolol 25 mg for headache prophylaxis, acetaminophen with diphenhydramine, and an oral contraceptive pill. There was no family history of migraine or seizures.
Neurological examinations including visual acuity, visual fields to confrontation, pupillary responses and funduscopy were normal. MRI of the brain was unremarkable, as were an EEG and extensive haematological tests. The patient was treated with a gradually escalated dose of topirimate to 100 mg/day and was advised to discontinue the oral contraceptive pill.
Patient 6
A 59-year-old female nurse had suffered from severe attacks of migraine with aura since the age of 7 years. The pain was maximal behind one eye with radiation into the forequarter of the face. Two-thirds of the attacks were right-sided. The pain was throbbing and pulsating, lasting 4–24 h, and was associated with photophobia, phonophobia, nausea and vomiting. During her teenage years the headaches had occurred once per week but became less frequent in her 30s (one per month). A monocular visual aura consisting of black spots and zigzag lines lasted 15 min and then fully resolved over another 1–2 min, heralding an ipsilateral migraine headache. Alternately covering each eye during attacks confirmed that visual features were confined to one eye.
Seven years before presentation the patient had experienced acute loss of vision in the form of a ‘grey patch’ over the left eye several minutes before an otherwise typical left-sided migraine headache. Gadolinium- enhanced MRI of the brain and orbits showed no lesions. Similarly, MRA of the brain and neck, echocardiography, Holter monitoring, and evaluations for hypercoagulability and autoimmune diseases were also normal. The patient was treated by another physician with dexamethasone 4 mg i.v. q 8 h, sublingual nitroglycerin and prophylactic aspirin 81 mg and verapamil 120 mg. The headache resolved approximately 24 h after onset, but the monocular visual loss persisted for 3 days before returning to baseline. The patient continued taking verapamil and aspirin irregularly for several years, with only one mild headache per month. One month before presentation she had developed a gastric ulcer and aspirin was stopped. Around the same time, the patient stopped the verapamil due to swelling of her left ankle. A deep venous thrombosis was diagnosed by Doppler ultrasound. Aspirin 325 mg was re-started with additional gastrointestinal protection.
Two weeks later the patient experienced permanent monocular visual loss of the right eye in association with a migraine headache. She noted a persistent ‘greying out’ of vision in her right eye, with no colour perception. This particular migraine headache was more severe than her prior attacks, awakening her from sleep at 02.30 h. Neuro-ophthalmological examination 2 weeks later revealed a right afferent pupillary defect with red desaturation and diminished colour vision on the right (Ishihara Plate 6.5/10 right, 10/10 left). There was no optic disc oedema or pallor. Visual acuity with correction was 20/30– in the right eye pinholing to 20/30+. In the left eye, she was 20/25 + 1 pinhole no improvement. Humphrey visual field testing was normal in both eyes.
Repeat MRIs of the brain, neck and orbits as well as extensive haematological studies were normal. Treatment with a gradually escalated dose of nortriptyline to 75 mg/day, verapamil SR 240 mg/day and enteric-coated aspirin 325 mg/day was followed by a complete cessation of headaches. With the exception of a mild residual afferent pupillary defect, the patient's visual acuity and colour vision returned to baseline by 1 month's follow-up.
Literature review
Herein we review the features of 40 previously reported patients as well as the six newly reported patients.
Demographic data
Of the 46 patients with retinal migraine, 28 (61%) were female. Twenty-five of 46 patients experienced only transient monocular visual loss [transient visual symptom (TVS) group, Table 2], while 21 (46%) later developed permanent monocular visual loss in association with otherwise typical attacks of migraine [permanent visual loss (PVL) group, Table 3]. Men and women were equally affected in the transient group (13 women, 12 men), whereas the permanent group showed a female preponderance (15 women, six men).
Cases of retinal migraine and transient monocular visual loss (TMVL) only

Frequency of migraine attacks with TMVL.
Relative to migraine headache.
Based on personal communication with Dr Corbett. R, Right; L, left; MO, migraine without aura; MA, migraine with aura; PM, probable migraine; N, negative; P, positive.
Cases of retinal migraine and transient monocular visual loss (TMVL) followed by permanent monocular visual loss (PMVL)

Frequency of migraine attacks with TMVL.
Eye affected with TMVL.
Relative to migraine headache.
R, Right; L, left; MO, migraine without aura; MA, migraine with aura; PM, probable migraine; N, negative; P, positive.
Age at onset of retinal migraine ranged from 7 to 54 years. Mean age at onset was similar in the two groups (24.7 years for the TVS group, 23 years for the PVL group). In the TVS group, the peak age at onset was the third and fourth decade in women and the second and third decade in men. In the PVL group, peak age at onset was the second and third decade in women and the second decade in men. The duration of retinal migraine before diagnosis ranged from days to several decades. Similarly, the evolution from transient to permanent attacks of monocular visual loss in the PVL group was variable, occurring within the same year of onset of retinal migraine and up to 52 years later. With the exception of one case, in which attacks of transient monocular visual loss ceased after discontinuation of oral contraceptive use, no specific precipitating events were identified. A family history of migraine was documented in 30% (14/46) of patients. Because many cases did not include information on family history, this number may be underestimated. Only two patients had familial retinal migraine.
Clinical features
All patients reported a history of migraine prior to the onset of monocular visual loss. Subtypes of migraine included migraine with conventional visual aura (50%), migraine without aura (26%) and probable migraine (24%). Non-visual auras such as sensory and language abnormalities were identified in two patients. One patient reported coexisting cluster headache. Headache laterality was reported in 23 patients. The pain was ipsilateral to the transient monocular visual loss in 83% of patients, contralateral in 4% and bilateral in 13%. Attacks of monocular visual disturbances occurred exclusively on one side in 82% of cases, whereas 18% had transient monocular visual loss of either eye in separate attacks. The onset of monocular visual loss preceded the headache in 48%, accompanied the headache in 50% and followed it in 2%. The headache was either preceded by or followed by monocular visual loss in one patient. In eight patients, episodes of reversible monocular visual obscuration occurred from 1 min to 1 h prior to onset of the headache. In two other patients, bouts of temporary monocular visual impairment began 10–30 min after the headache started.
The minimum attack duration of transient monocular visual loss was <5 min in 22% (8/37), between 5 and 15 min in 30% (11/37), between 20 and 30 min in 14% (5/37) and 1 h in 11% (4/37); one other patient had repeated episodes of transient monocular visual loss lasting from 1 to 20 min. Twenty-two percent of cases (8/37) experienced repeated attacks of transient monocular visual loss lasting >1 h. Of these eight patients, four had maximum durations of several hours, two of days and two of weeks. All patients had experienced at least two attacks of transient monocular visual loss. Minimum attack frequency was less than yearly in 17% (4/24), yearly in 13% (3/24), monthly in 21% (5/24) and weekly in 42% (10/24); the remaining two patients had attacks that ranged from a monthly to a weekly basis.
Positive and negative visual phenomena were reported in both the TVS and PVL groups. Teichopsias limited to one eye were reported as flashing rays of light, zigzag lightning-like patterns, bright coloured streaks, or halos. Negative visual symptoms included blurring, ‘grey-outs’ and ‘black-outs’, causing partial or complete blindness. Visual field defects were either altitudinal, quadrantic, central or arcuate. Complex patterns of monocular visual impairment, such as the appearance of ‘black paint dripping down from the upper corner of my left eye’, were noted in one patient. Visual phenomena in both groups were exclusively negative in 65% (30/46), strictly positive in 4% (2/46) and both positive and negative in 31% (14/46). Most patients in the PVL group experienced only negative features. Detailed descriptions of the nature of the monocular visual loss varied from report to report. We classified descriptions of visual loss into five non-mutually exclusive categories: monocular blindness or blurring of the entire visual field (n = 28), quadrantic defects (n = 6), altitudinal defects (n = 4), other specific zones of visual loss (n = 10) and positive or negative scotmata within the monocular visual field but not further localized (n = 24).
Ophthalmological examination
All patients in the TVS group had normal visual acuity between attacks. Five patients who were examined during attacks had reduced visual acuity or some form of visual deficit, which returned to normal with the resolution of the migraine headache. Abnormal vascular lesions on funduscopy were noted in two patients but were not related to the monocular visual loss.
Ophthalmological examinations in the PVL group are listed in Table 4. Half of the patients in the PVL group developed reduced visual acuity between attacks that was not reversible. Relative afferent pupillary defects were noted rarely. Most of these patients (90%) who were examined following an attack had some form of permanent visual field defect. With the exception of two patients who had documented permanent monocular visual loss but no reported funduscopic examinations, all of the patients in the PVL group had abnormal funduscopic examinations.
Cases of retinal migraine and transient monocular visual loss (TMVL) followed by permanent monocular visual loss (PMVL)

APD, Afferent pupillary defect; BRAO, branch retinal artery occlusion; CRAO, central retinal artery occlusion; CRVO, central retinal venous occlusion; LE, left eye; NFB, nerve fibre bundle; RE, right eye; VA, visual acuity; VF, visual fields.
Laboratory testing and neuroradiological findings
A brain MRI or CT was completed in 10 patients from the TVS group and four patients from the PVL group, of which all had normal studies; particular attention was given to the orbits in three of the neuroimaging studies. Cerebral angiography was completed in four patients and was also normal. Laboratory tests in 22 of the entire 45 patients were normal. Extensive haematological studies were rarely reported.
Concomitant illness
Comorbid risk factors for vascular disease were noted in 16% of patients in the TVS group and 15% in the PVL group. The most frequently reported conditions were arterial hypertension and smoking, with three patients in each group. The remaining conditions reported were diabetes mellitus (n = 2), oral contraceptive use (n = 2) and angina (n = 1). No pattern of disease or disorders associated with retinal migraine was noted.
Therapy of retinal migraine
Acute medication during the attack of retinal migraine was used in only a few patients. Inhaled carbon dioxide improved vision ‘slightly’ in one patient during one attack, whereas amyl nitrate and isoproterenol via inhaler showed ‘good efficacy’ in improving the visual loss in a few patients. None of these medications helped relieve the headache. Medications such as calcium channel blockers, oral and intravenous corticosteroids, and vasodilators showed no consistent benefit in patients who experienced permanent monocular visual loss.
All classes of migraine prophylactic agents were tried in patients with retinal migraine. Some patients experienced a reduction or cessation of retinal migraine attacks after use of β-blockers, calcium channel blockers, tricyclic antidepressants, antiepileptic drugs or aspirin. However, there are no data to determine the efficacy of any preventative agent for retinal migraine.
Discussion
In this review, patients with retinal migraine had a demographic profile which differed from migraine with typical aura and migraine without aura. The gender prevalence ratio for retinal migraine of 1.4 : 1 female preponderance was lower than the 3 : 1 ratio for more common types of migraine (28). While the onset of migraine with and without aura peaks in adolescence (29), the average age at onset of retinal migraine is substantially later, i.e. in the second or third decade. However, the mean age at onset was similar in both the transient and permanent forms of retinal migraine.
Half of patients in the literature, and four of our six patients, had a history of migraine with aura prior to their attacks of retinal migraine or during attacks of monocular visual loss. In almost all of the cases, the auras were visual. Rare cases of transient monocular visual loss have also been reported with cluster headache (30), idiopathic stabbing headache (31), chronic daily headache (8), cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencephalopathy (32) and an unspecified headache type (33). The visual features and headache characteristics of retinal migraine differed from migraine with aura in several respects. Monocular visual loss of retinal migraine accompanied the headache as often as it preceded it, whereas in migraine with aura, the headache usually (92%) follows the visual aura (34). Although the ICHD-2 criteria require that the visual loss accompany or precede the headache, the monocular visual loss followed the headache in 2% of patients. Some data suggest that only a small percentage of patients with unilateral migraine have ipsilateral aura symptoms (35), while in retinal migraine, attacks of transient monocular visual loss were overwhelmingly (83%) ipsilateral to the headache and occurred exclusively in one eye (82%). Most patients with retinal migraine (77%) and migraine with aura (34) (89%) experienced visual symptoms lasting ≤1 h. The duration of the transient visual symptoms in patients with retinal migraine was as short as a few seconds (approximately 8% of patients), but usually lasted for many minutes to 1 h. A surprisingly large minority (22%) of patients with retinal migraine had attacks that lasted >1 h, in contrast to migraine with aura (34) (8%). Some of these cases had prolonged but fully reversible monocular visual loss, sometimes lasting hours, days, or even weeks, supporting the decision of the ICHD-2 classification to include monocular aura durations of >1 h.
Only a few patients had ophthalmological examinations during an attack. When performed, examinations were usually normal, suggesting retrobulbar involvement. Additionally, 43% of patients in this series ultimately experienced permanent monocular visual loss, but no consistent pattern of visual field defect was noted. We were unable to identify predictors of permanent visual loss. Just as migraine aura sometimes gives rise to migrainous infarction, we believe that irreversible visual loss is part of the spectrum of retinal migraine, perhaps a form of migrainous infarction. We recommend caution in generalizing the findings from this series. For example, patients who permanently lose vision may be more likely to be reported; however, in our six cases only one patient had permanent monocular visual loss.
Retinal migraine is thought to be a rare disorder, but its true prevalence is unknown. Recent studies with automated perimetry have demonstrated subclinical visual field defects in some asymptomatic patients with migraine (36). There was a correlation between these findings and duration of disease and advancing age. The interpretation of these findings is uncertain. Other studies such as kinetic arc perimetry (37), measurements with flickering light stimuli (38), motion coherence perimetry (39) and measurements of contrast thresholds for static and moving stimuli (40) have implicated both cortical and precortical visual sites. Although retinal migraine has usually been viewed as a benign condition, it appears that subclinical precortical visual dysfunction and permanent attacks of partial or complete monocular visual loss may occur in patients with migraine more often than generally appreciated.
Comorbid risk factors for vascular disease were noted in only a few patients with transient and permanent monocular visual loss. They included hypertension, hyperthyroidism, diabetes, oral contraceptive use and smoking. These conditions were not severe and were not thought to be the main cause of the visual loss.
Patients often have difficulty distinguishing between the loss of vision in one homonymous hemifield and the loss of vision in one eye. To make this distinction accurately the patient must alternately cover each eye. The description of hemifield loss with both eyes open is characteristic of a homonymous hemianopia rather than monocular visual loss. If uniocular visual loss is confirmed, one must attempt to exclude all causes of transient or permanent monocular visual loss; retinal migraine is a diagnosis of exclusion. The differential diagnosis of retinal migraine includes amaurosis fugax. In retinal migraine, visual loss typically evolves slowly and often lasts longer than amaurosis fugax of carotid artery origin. The ‘shade dropping over a visual field’, typical of microembolization, was not reported by patients with retinal migraine. Other causes of transient monocular visual loss include atherosclerosis, thrombus orginating from the carotid artery, heart or great vessels, giant cell arteritis, other arteritides with or without autoimmune diseases, primary vascular disease of the central retinal artery or vein, illicit drug use, and hypercoagulable states such as macroglobulinaemia, polycythaemia, anticardiolipin antibody syndrome and sickle cell disease. Less common causes are orbital diseases including mass lesions, retinal detachment and intermittent angle closure glaucoma (41).
The mechanisms of prolonged and permanent monocular visual loss in association with migraine are uncertain. Bouts of transient monocular visual loss lasting <1 h, transient monocular visual loss of prolonged duration and transient monocular visual loss that later becomes permanent correspond clinically to the conventional auras of migraine, prolonged auras, and migrainous infarction, respectively. Perhaps these three phenomena affecting the cerebrum (especially the cortex) or the eye (especially the retina) share common pathophysiological mechanisms. Spreading depression of neurons is the accepted concept of the typical aura of migraine and there may be a similar mechanism affecting the retina. This phenomenon has been noted in the retina of the chicken (42). One patient who described her transient monocular visual loss as black paint slowly dripping down from the corner of her monocular visual field may well be describing spreading depression of the retina (11).
Ischaemia is the other mechanism commonly invoked to explain permanent monocular visual loss in the setting of migraine. As noted previously, vasospasm of retinal arterioles and veins has been demonstrated in cases of transient monocular visual loss where no other predisposition to vascular disease was discovered (43). Vasospasm during migraine headaches has also been angiographically documented (44). While vasospasm is no longer considered to be the primary cause of the focal neurological deficits of migraine, this older concept of migraine may account for a shared pathophysiology in some cases.
Prolonged and permanent monocular visual loss appear to occur much more commonly in patients with retinal migraine than in cases of prolonged typical aura or migrainous infarction in those with conventional migraine. The high number of patients with transient monocular visual loss, who eventually develop permanent monocular visual loss, makes retinal migraine a less benign condition than migraine with conventional visual aura. Therefore, although there are no data to determine the efficacy of preventative treatment for this entity, prophylactic drug therapy seems prudent, even if attacks are infrequent. There are no studies addressing the efficacy of any one preventative agent for retinal migraine. Aspirin is a logical agent because of its antiplatelet activity. In a few cases reported above simple monotherapy reduced the frequency of migraine with and without monocular visual defects. We favour antiepileptic drugs (i.e. topirimate or divalproex sodium) and tricyclic antidepressants (i.e. amitriptyline or nortriptyline). Although some patients respond to β-blockers, we do not usually recommend them because of their theoretical potential for arteriolar constriction. Although episodes of vasospastic amaurosis fugax appear to have been successfully treated with calcium-channel blockers (45), they were not effective in the few patients we treated. Several of our patients were refractory to many migraine preventives, given as monotherapy or in combination. Therapy of the acute attack of retinal migraine should probably not include triptans or ergots because of their vasoconstrictive properties. In a few cases, acute vasodilators appeared to shorten the period of monocular visual defects (but little if anything was said about the potential for aggravating migraine).
Based on our clinical series and review of the literature, we propose a revision of the ICHD-2 criteria for retinal migraine (Table 5). The current ICHD-2 criteria require transient visual loss exclusively, yet we found that nearly half of the patients later developed permanent monocular visual loss. The pathophysiology of visual loss in retinal migraine is uncertain, making the nomenclature of ‘retinal migraine’ problematic. Patients and physicians tend to use the terms ‘retinal migraine’ and ‘ocular migraine’ to describe various syndromes of headaches and visual loss. An on-line survey by the American Headache Society has shown that even headache specialists are confused about the terminology. We propose abandoning the term ‘retinal migraine’ in favour of more accurate terminology, migraine associated with monocular visual symptoms. Our criteria divide this type of migraine into those with only transient monocular visual loss and those who develop permanent monocular visual loss. The types of associated migraine include migraine without aura and migraine with aura, given that there are no concurrent cerebral visual symptoms. Furthermore, the migraine headache may begin during the transient monocular visual loss or within 60 min after or (rarely) before the transient monocular visual loss.
Proposed criteria for migraine with monocular visual symptoms
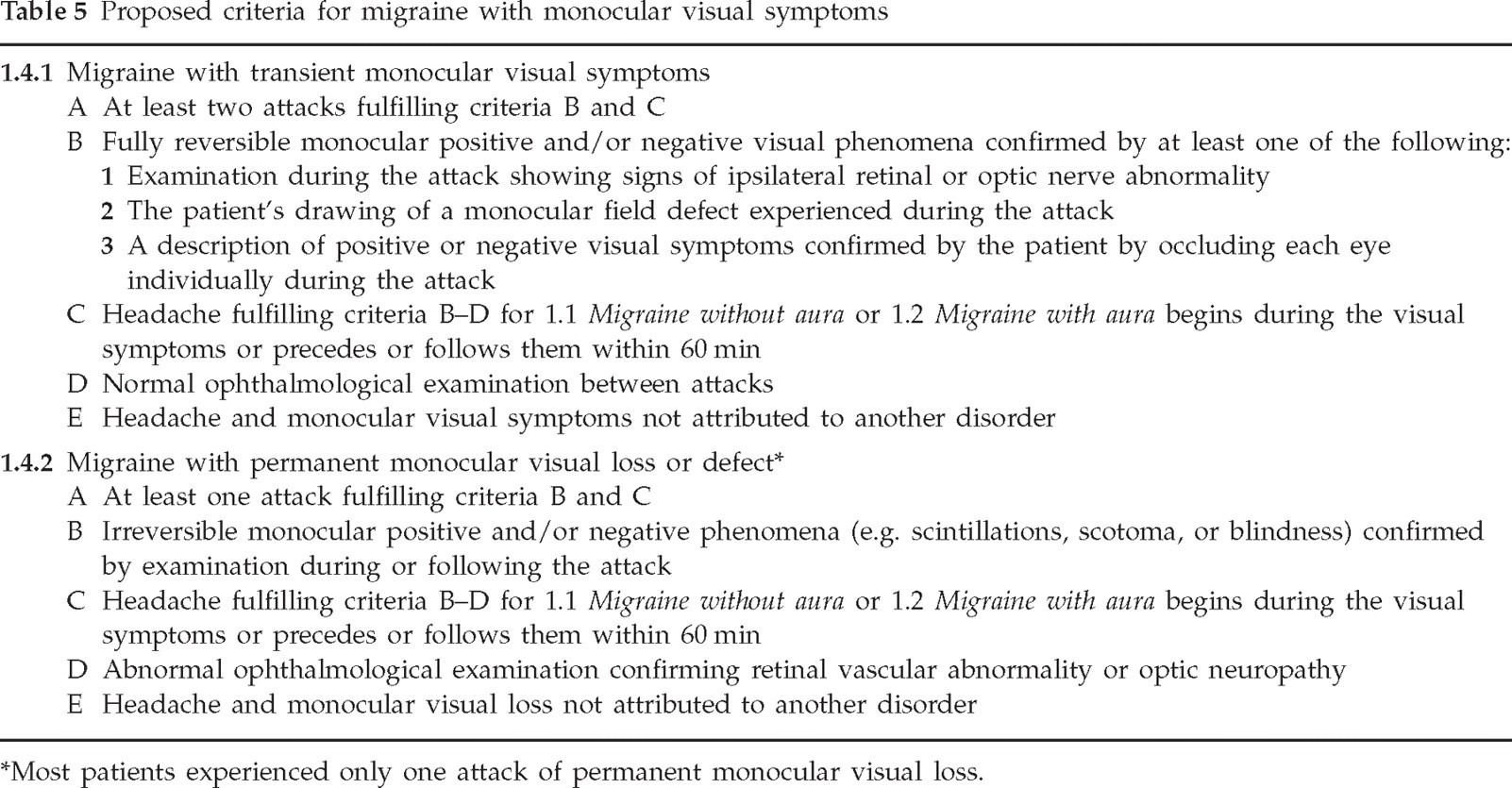
Most patients experienced only one attack of permanent monocular visual loss.
An issue for future research is the classification of acephalgic transient monocular visual loss, commonly referred to as ‘migrainous’, ‘migraine variant’, ‘acephalgic migraine’ or ‘complicated migraine’. These episodes usually occur in adults with a history of migraine, last minutes, and may occur in a ‘flurry’ over days to weeks (46, 47). As with other migraine disorders, this must be a diagnosis of exclusion, particularly among patients aged >50 years.
Footnotes
Acknowledgements
Supported by the National Eye Institute 1K23EY015525 and a Research to Prevent Blindness Challenge grant. These findings were presented in part in abstracts and a review article.
