Abstract

In accordance with the diagnostic criteria of the International Headache Society (IHS) (1), basilar-type migraine (BTM) is a cephalalgic syndrome with aura symptoms clearly originating from the brainstem and/or from both hemispheres simultaneously affected, including: dysarthria, vertigo, tinnitus, hypacusia, diplopia, visual symptoms, ataxia, bilateral paraesthesias and alterations of consciousness, but no motor weakness. These symptoms are quite frequently associated with seizures and/or severe EEG abnormalities (2–4). The alterations of consciousness can include stupor or, less frequently, a transient state of coma (5).
We describe a clinical case of headache in which the clinical and instrumental characteristics initially appeared to meet the IHS criteria for a diagnosis of BTM. However, the unexpected clinical evolution led to a diagnosis of a form of basilar migraine-like symptomatic headache.
Case report
A 36-year-old overweight male smoker started to complain of cephalalgic episodes with occipital pulsating pain. These episodes occurred at least once a day and were associated with one or more of the following symptoms: vertigo, nausea, vomiting, double vision, photophobia, cheiro-oral paraesthesias, mono or bilateral visual disorders and wandering tingling paraesthesias, frequently on one side of the body. On admission to hospital the patient’s neurological examination, including fundus oculi, were normal. Magnetic resonance imaging (MRI), performed on admission, revealed two focal lesions, one in the right cerebellar hemisphere (approximately 2 cm in diameter) (Fig. 1) and the other in the left paramedian occipital cerebral cortex, characterized by hypointensity in T1-weighted and hyperintensity in T2-weighted images with modest contrast enhancement.

Axial view magnetic resonance image, performed at admission, shows a focal lesion in the right cerebellar hemisphere (black arrow), approximately 2 cm in diameter, characterized by hyperintensity on T2-weighted images.
Transcranial Doppler (TCD) and echocolour Doppler showed a reduction in the mean flow velocity in the vertebral and posterior cerebral arteries. Single-photon emission computed tomography (SPECT) showed hypoperfusion throughout the left cerebral hemisphere in the interictal phases and during migraine attacks a considerable degree of hypoperfusion in the left occipital cortex and in the right cerebellar hemisphere.
During migraine attacks the EEG recordings showed a slow activity in temporo-occipital lobes. Treatment with amitriptyline 20 mg daily and flunarizine 10 mg daily was started with reduction in frequency and intensity of cephalalgic attacks.
After 15 days the patient was discharged with a diagnosis of ‘basilar-type migraine complicated by occipital and cerebellar ischaemic lesions’.
At the time of discharge, as a consequence of the finding of a high haematocrit (53.4), with 6 × 106 red cells and 18.4 g/dl haemoglobin, the patient was referred to the Department of Haematology, where he was bled.
After temporary improvement, the patient started to complain again of frequent severe attacks of occipital pulsating headache associated with vertebrobasilar focal transient neurological deficits and was re-admitted to hospital.
Among the most important clinical symptoms that occurred during headache episodes, we observed transient paresis of the third and sixth cranial nerves, sometimes alternating, sudden loss of extensor tone of the lower limbs, episodes of loss of consciousness with clonic movements of the upper limbs lasting several minutes accompanied by anisocoria and paralytic mydriasis, ictal episodes of confusion and epileptiform episodes like ‘dejà vu’ and ‘jamais vu’.
A new MRI, performed 45 days after the first, showed further small focal paramedian lesions of the left pons and a new lesion in the right occipital lobe (Fig. 2). Magnetic resonance angiography was normal.
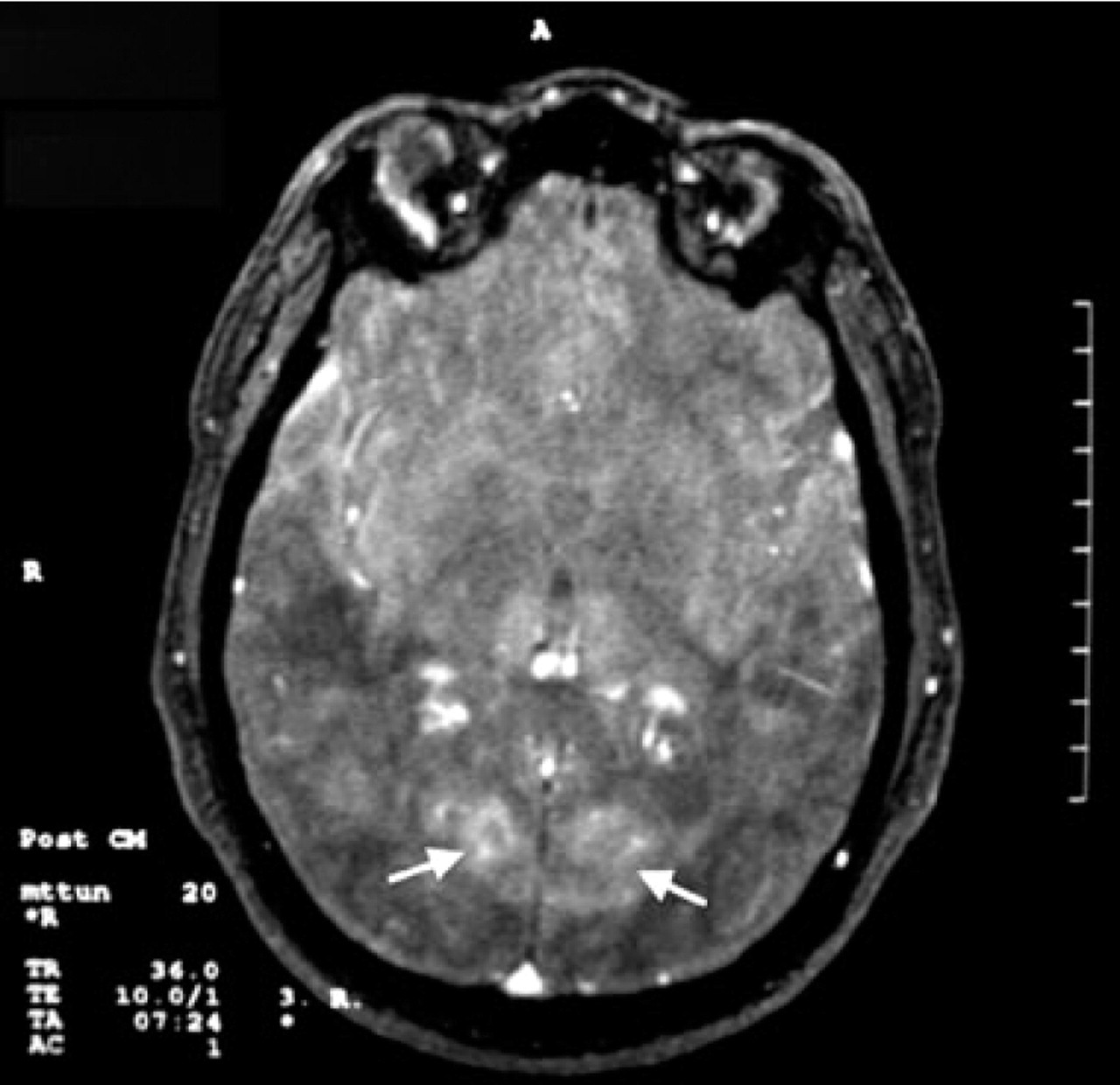
Axial view by magnetic resonance imaging (MRI), performed 45 days after the first MRI, shows bilateral lesions (white arrows) in the paramedian occipital cerebral cortex characterized by hypointensity on T1-weighted images with modest contrast enhancement.
The fundus oculi examination showed bilateral papilloedema with peripapillary haemorrhage; spinal fluid lumbar pressure was high at 75 cmH2O and a ventriculoperitoneal shunt was implanted. The patient’s neurological and general condition continued to deteriorate and after some weeks he died. Neuropathological examination revealed that the cerebellar lesion observed at MRI, which appeared as a large migraine infarction, and the other encephalic lesions were actually a disseminated medulloblastoma (Fig. 3).

Histological section of cerebellum shows tumour with roundish and slightly spindled ‘small blue cells’ that constitute a ‘cuff’ on the organ surface (haematoxylin and eosin × 250).
Discussion
In the correct diagnostic screening of headaches, it should first be determined whether the patient has symptomatic or idiopathic headache, even when the headache appears to meet the IHS criteria (1) for migraine with or without aura, and especially in the case of BTM. It is common knowledge that Bickerstaff migraine (6) or BTM may manifest with a variety of symptoms resembling those of other neurological, psychiatric, vascular and metabolic disorders (7).
The clinical case described here is an example of such a diagnostic pitfall in BTM. The patient was referred to our centre due to his clinical symptoms, pointing to a likely case of transient ischaemia in the distribution of the basilar artery and its branches (8), apparently meeting the IHS criteria for BTM. This diagnosis was further supported by Holter-EEG findings (4), TCD and echocolour Doppler studies, SPECT examination (2) and neuroimaging studies (8–10). Moreover, polyglobulia appeared to represent a clinically relevant factor in explaining the extension and high number of migrainous infarcts and frequency of the attacks.
The temporary remission of migraine attacks subsequent to treatment with flunarizine and amitriptyline substantiated our impression of dealing with a basilar artery migraine; paradoxically, the case described by McDonald (9) added additional support to this hypothesis, in spite of the presence of an obstructive hydrocephalus. Unfortunately, medulloblastoma was identified as the primary cause of this complex symptomatology only by means of autoptic findings and neuropathological examination.
Medulloblastoma is a highly malignant primitive neuroectodermal tumour of the cerebellum more frequently diagnosed in males aged <10 years and rarely in adults. The tumour is most often localized in the midline cerebellar region, with a propensity to spread to the meninges and cerebrospinal fluid spaces. In a previous report describing 15 adult patients with medulloblastoma, headache was reported in 93% of patients and represented the most frequent symptom (11). In a rectrospective study of 111 patients with various cerebral neoplasms, 48% of patients were affected by headaches: 77% had tension headaches, 9% migraine and 14% other types (12). Indeed, headache associated with brain tumours does not necessarily meet the IHS criteria for tumour-correlated headache (12, 13) with increased intracranial pressure or hydrocephalus (1), and is at times present as a migraine-type headache (12, 13).
In this regard, it would be intriguing to identify the physiopathological mechanism involved in the inducing of migraine-type headache in medulloblastoma.
Although the pathophysiological mechanism of migraine is still the subject of debate, several facts are commonly acknowledged: namely, that migraine is a complex neurovascular disorder in which both interrelated neuronal and vascular elements play an important pathophysiological role (13, 14).
Through activation of a mechanism inducing compression, malignant infiltration, traction and aseptic inflammation, among others, medulloblastoma may have triggered pathophysiological mechanisms inducing migraine. First, by means of the release of substance P and other vasoactive peptides into the wall of cranial blood vessel and the meninges, the trigeminovascular system (15) may have induced an aseptic inflammatory response. Vasoactive peptides and other biochemical and metabolic alterations may have supported a state of neuronal central hyperexcitability (16) and increased excitability of the cerebral cortex similar to that observed in migrainous patients (17), thus eliciting a painful response to otherwise harmless stimuli. Moreover, even the spreading depression (18) and spreading oligaemia (19), adding further support to the hypothesis that the aura of migraine is primarily a neuronal event coinciding with the onset of a wave of cerebral cortical electrical depression followed by reduction of regional cerebral blood flow, are inducible by means of different stimuli such as hypoxaemia and mechanical and chemical injury (18, 20).
Paramedian lesion of the pons (21), exerting a direct action of infiltration, and lesion of the right cerebellar hemisphere, with an indirect action of compression, may suggest the involvement of the central antinociceptive neuromodulator systems, including the periaqueductal grey and the rostral ventromedial medulla (22).
In our patient, both paroxysmal epileptiform manifestations (4) and focal symptoms such as transient alternating ophthalmoplegias and paraesthesias, ataxia, cheiro-oral paraesthesias, etc., can be explained by the activation of these physiopathological mechanisms (12, 13, 21). The latter would also provide an explanation for the instrumental findings leading us to hypothesize BTM, thereby constituting a major diagnostic pitfall.
Unfortunately, neuroimaging studies provided no useful information for the formulating of a correct diagnosis. In this regard, however, medulloblastoma has variable appearances at MRI and very few studies have addressed the MRI features of adult medulloblastoma (23).
