Abstract
The purpose of this study was to examine basic ocular motor function in individuals with migraine. We used an infrared eye-tracking system to measure horizontal smooth pursuit to a sinusoidal target, saccades to horizontal target displacements of 5–20°, and the stability of fixation in 19 migraine without aura (MoA), 19 migraine with aura (MA) and 19 headache-free control (C) subjects. Eye movement measurements were made at two target displacement rates and against both homogeneous grey and patterned backgrounds. We found no statistically significant differences between migraine and control subjects in any of the eye movement parameters measured, but did find highly significant effects of both target speed and background pattern in all groups. Our results do not provide support for subclinical cerebellar impairment in migraineurs, and do provide evidence that previously described visual abnormalities in migraine are not artefacts of abnormal fixation or eye movements.
Introduction
The involvement of the visual system in migraine has received a great deal of attention in recent years (see (1) for review); however, the ocular motor side of visual function has been largely ignored. This is surprising in two regards. Normal visual function depends in part on good ocular motor control: for example, unstable fixation will degrade performance on many visual tasks. Conversely, visual processing deficits may produce apparent abnormalities in eye movements: if visual motion analysis were abnormal, as it has been reported to be in migraine (2–4), pursuit tracking would be likely to reflect this.
The lack of eye movement studies in migraine is also surprising because the cerebellum, which plays an integral role in ocular motor control, has been implicated in migraine in recent genetic and imaging studies. The genetic studies (5, 6) focus on a rare but striking variant of migraine, familial hemiplegic migraine (FHM). There is convincing evidence that FHM involves a calcium channelopathy, specifically affecting the voltage-gated P/Q calcium channel (see (7) for review). The distribution of this channel type in the central nervous system is most pronounced in the cerebellum, cerebral cortex, thalamus and brainstem regions, including the hypothalamus (8–12). There is also functional evidence of cerebellar involvement in FHM, including ocular motor abnormalities such as nystagmus and reduced pursuit gain (13). While interesting in their own right, these findings would take on enormous significance if they were demonstrated to have direct relevance to the more common forms of migraine, migraine with and without aura, the genetic component of which is far more complex and heterogeneous than that of FHM (7, 14).
The most direct evidence of a link between the cerebellum and more common forms of migraine is the recent report of a greater than expected prevalence of cerebellar infarct in migraine patients, which was particularly notable in individuals with frequent episodes (>1/month) of migraine with aura (15). A number of groups have begun looking for functional indicators of cerebellar dysfunction. Sándor et al. have reported evidence of hypermetric arm movements in migraine with and without aura which was exacerbated with increasing inertial load, and which the authors suggest may be indicative of lateral cerebellar abnormality (16). Harno et al. reported subclinical dysfunction on a variety of vestibulocerebellar tasks, which was more pronounced in migraineurs with aura (17). However, only two studies have examined eye movements, one reporting poorer saccadic accuracy (both hypermetria and hypometria) in migraineurs than in controls (17), while the other reported normal saccades, but abnormalities in smooth pursuit (18). To our knowledge there has been no examination of fixation stability.
Smooth pursuit is a particularly sensitive ocular motor marker of cerebellar dysfunction (19) and, thus, is a valuable test of subclinical abnormalities. However, it is also prone to contamination by other variables because the pursuit system is affected by ageing (20–22), by numerous medications (19) and by a variety of neuropathologies including schizophrenia (23). Saccadic eye movements are also affected by cerebellar pathology, the most prominent consequence of which is hypermetria or overshooting (19). Saccadic velocity and latency are also sensitive to a variety of neurological disorders.
The present study was designed to examine the integrity of the smooth pursuit and saccade systems and the ability to maintain steady fixation in migraine with and without aura. In order to be able to examine ocular motor function under a variety of visual demands, we limited our study to horizontal eye movements. The literature on humans with normal visual and ocular motor function reveals that the visual background upon which pursuit (24, 25) is carried out exerts a significant influence on the spatial and temporal parameters of the movements. Since there is also a literature indicating particular sensitivity of migraineurs to some forms of high-contrast repetitive visual patterns (26, 27), we made our eye movement and fixation measurements against three visual backgrounds—a solid grey field, high-contrast stripes (sinusoidal grating) and a pattern of random noise. We anticipated that the reported effects of background pattern on pursuit gain might be exacerbated in migraineurs. Poor fixation ability or abnormal eye movement patterns could also contribute to the illusions and visual discomfort reported in migraine (26, 27) in the presence of high-contrast stripes.
Methods
Participants
The study was approved by the ethics review boards of York University and of the University Health Network and was conducted in the Ocular Motor Laboratory at the Toronto Western Hospital. All participants gave written informed consent. Participants were recruited by advertisements posted in local hospitals, universities and one local newspaper. Each potential participant was interviewed by phone, using structured health and headache questionnaires. This was followed up with a more detailed structured interview in the laboratory for those who passed the initial screening. International Headache Society (IHS) criteria (28) were followed to identify individuals with migraine with (MA) and without (MoA) aura. Control participants (C) had no history of migraine or of frequent headaches (<12 tension headaches per year) and were matched as closely as possible to the migraine groups in age and gender ratio.
All participants were screened for medical and ocular conditions relevant to visual and ocular motor performance, including a family history of schizophrenia which is known to affect smooth pursuit in affected individuals and first-degree relatives (29). Other exclusionary criteria included use of prophylactic migraine medication, antidepressants, hypnotics, anxiolytics or herbal remedies. Individuals with known refractive errors of more than 3.5 diopters (D) of myopia were excluded because spectacle or contact lens correction could not be used with our eye tracker. Distance visual acuity (Sloan letter chart) and stereopsis (Titmus test; Titmus Optical Co. Inc., Petersburg, VA, USA) were measured in all participants. Participants had all been headache free for at least 5 days at the time of testing.
Apparatus and stimuli
Visual stimuli were generated on a Macintosh G4 computer using VPixx™ software and were displayed on a 19-in monitor (Samsung, SyncMaster 900NF™ flat screen CRT, 1024 × 768 pixel resolution and 120-Hz frame rate) positioned 57 cm from the participant’s eyes. Head position was maintained with a chin rest and eye movements were recorded using an El-Mar 2020™ head-mounted binocular infrared eye tracking system (El-Mar Inc., Toronto, Canada) with a sampling rate of 120 Hz, resolution of 0.1° and accuracy of 0.5°. Corneal reflections and the centre of the pupil are used to calculate the angular position of the eye. Head movements were recorded using a magnetic head tracker (Flock of Birds™; Ascension Technology Corp., Burlington, VT, USA).
The visual target was a 0.5° yellow dot. In different test intervals the dot moved against either a homogeneous grey background (luminance = 40 candelas/m2) or against structured backgrounds of stripes (Fig. 1a) or visual noise (Fig. 1b) of equal space-averaged luminance.
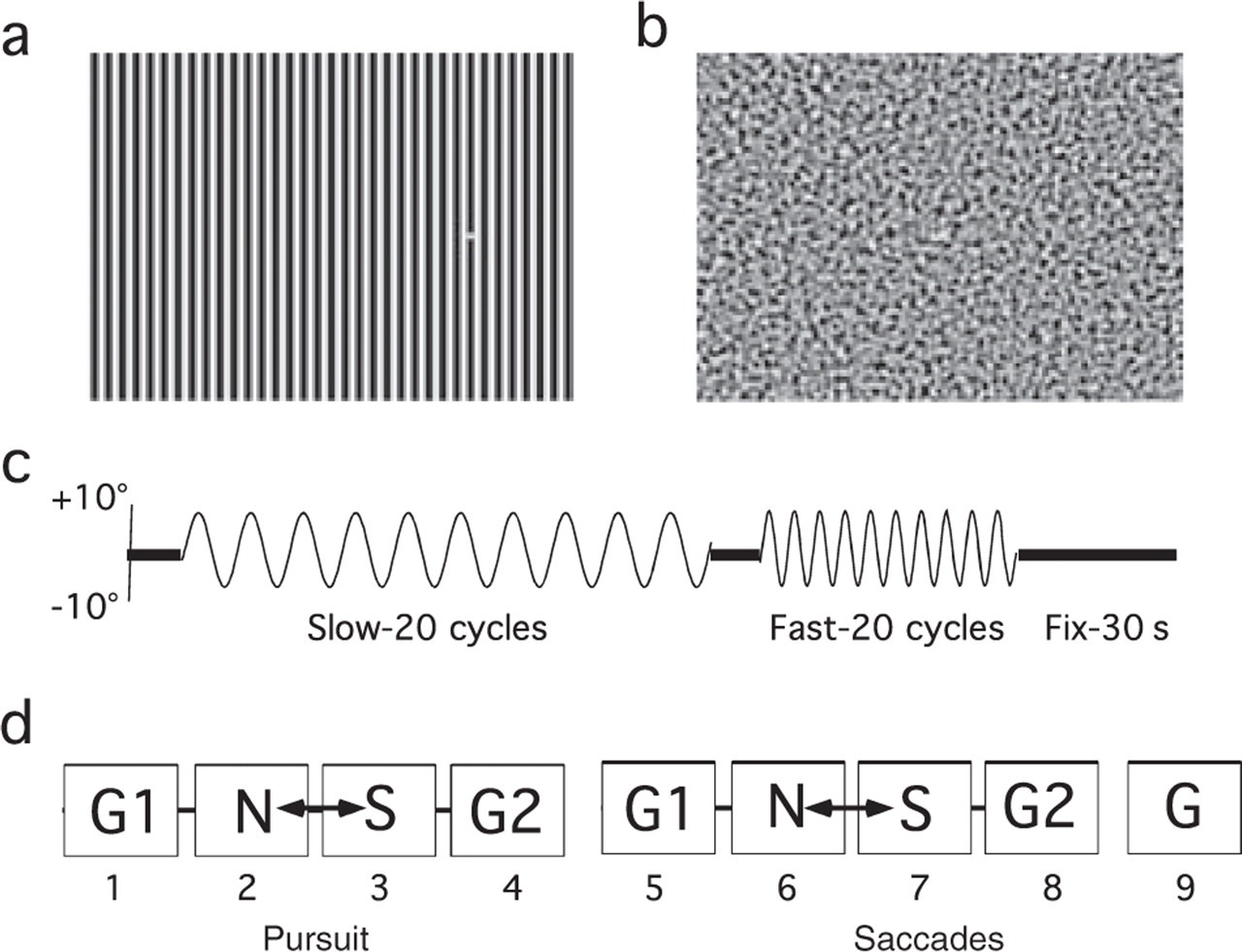
(a) High-contrast 1 c/deg sinusoidal grating, which is in the range previously shown to cause visual discomfort in some migraineurs, but is below the peak frequency for such effects (26, 27). The diameter of the target dot was equal to the width of one bar of the grating (0.5°). (b) Bandpass filtered visual noise, peak frequency 1 c/deg. (c) Test sequence for smooth pursuit test blocks, showing target position over time. Fixation periods of 5 s preceded the slow and fast pursuit phases, and a 30-s fixation period followed the fast pursuit phase. Saccade sequences were similar but with square-wave target displacement. (d) Sequence of test blocks. Smooth pursuit, blocks 1–4; predictable saccades, blocks 5–8; non-predictable saccades, block 9. Noise (N) and Stripe (S) blocks were counterbalanced across subjects. All grey (G) backgrounds were identical. Practice/fatigue effects were assessed by comparing performance in G1 and G2 blocks for smooth pursuit and for predictable saccades.
Horizontal smooth pursuit was assessed for a target moving with a sinusoidal velocity profile along a trajectory of ±10° from the centre of the visual field (Fig. 1c). Two frequencies, 0.25 Hz (Slow) and 0.5 Hz (Fast), were tested yielding maximum velocities of 15.7°/s and 31.4°/s, respectively. The predictable saccade sequences, with square-wave target alternation, were designed to mimic the pursuit stimuli. A final unpredictable saccade condition was included in which the target made 30 horizontal jumps at 2-s intervals against a grey background with distance (5°, 10°, 15° or 20°) and direction (leftward, rightward) determined randomly with the constraint that target position never exceeded ±10° of the centre of the field.
The recording session was broken into nine blocks, four of smooth pursuit (nos 1–4; 160 s each) and five of saccades (nos 5–9; 100 s each). Blocks were separated by short breaks to allow the dissipation of any afterimages induced by the backgrounds. The sequence of backgrounds across the nine blocks is shown in Fig. 1d; the order of stripes and visual noise blocks was counterbalanced across subjects. Subjects were asked to report any visual illusions that had occurred when viewing structured backgrounds.
Analysis
Smooth pursuit
Two dependent measures were used to assess the quality of smooth pursuit movements. Pursuit gain compares the velocity of pursuit movements with the velocity of the target. A gain of 1 would indicate that the subject’s eyes tracked the target perfectly. Pursuit phase (measured in angular terms) assesses the degree to which the eye movements are locked in time to the stimulus movement. Positive phase angles indicate that the eyes actually anticipate and lead the movement of the target, whereas negative phase angles indicate the degree to which the eyes lag the target movement. Pursuit gain is rarely perfect. Compensation normally takes the form of small catch-up saccades that bring the eyes back onto the target when slip has occurred. We assessed two measures of this compensation: number of catch-up saccades and catch-up saccade amplitude.
Blinks and saccades (peak velocity >60°/s) were marked manually by a trained rater who was blind to the group membership of individual records. Custom-written software replaced these segments with extrapolated best-fitting linear segments. This provided an estimate of eye position over time had corrective saccades not occurred (i.e. a pure pursuit record). Target and eye position traces were then differentiated to produce velocity data. The best-fitting sinusoid for each cycle of the pursuit record was determined and was compared with the target velocity sinusoid to determine phase and gain. Numbers and amplitudes of catch-up saccades were measured and included in separate analyses. Data for the two eyes were averaged for all measures.
Saccades
Following visual inspection of the records to remove blinks and eye tracker artefacts, custom-written MatLab™ software identified saccades made in response to the target displacements on the basis of velocity (>40°/s) and/or acceleration (>3500°/s2) criteria. A minimum latency criterion of 100 ms was employed to separate saccades made in response to the target displacement from anticipatory saccades, which were identified and counted separately. For each subject, mean amplitude, peak velocity and latency (and standard deviations) were calculated for each background and target alternation rate. Data for the two eyes were averaged, as were rightward and leftward movements.
Fixation
Fixation analysis was based on a plot of the distribution of instantaneous vertical (Y) and horizontal (X) eye positions (30) over an interval of 30 s following each pursuit and saccade test sequence. A trained rater, blind to the group membership of the subjects, used customized MatLab™ software to mark and remove blinks and artefacts from the eye position records. The remaining data were plotted in XY coordinates representing instantaneous position of the eyes with 0,0 representing the position of the fixation target. We then followed the procedure of Crossland and Rubin (30) to calculate the area of a bivariate contour ellipse (BCEA) which enclosed 86% of eye position samples:
where σh is the standard deviation of point locations over the horizontal axis, σv the standard deviation of point locations over the vertical axis, and ρ is the product-moment correlation of these two position components. The constant k is dependent on the probability area chosen: p = 1 – e − k . In the present analysis, we used a value of k = 1.966, which defines an elliptical area containing 86% of instantaneous eye position samples.
Statistical analysis
For most of our data sets, statistical analysis was performed using SPSS™ split plot
Results
Sample comparison
A total of 422 individuals were screened to arrive at our final group of 57 participants (19 MoA, 19 MA, 19 C). The most common reasons for excluding individuals were failure to meet IHS criteria (n = 94), use of prophylactic medications (n = 82), age (n = 46) and refractive errors >3.5 D (n = 42). The three groups were closely matched in age and in the number of years since their migraine onset (see Table 1). Migraine frequency (episodes/month) was higher for the MoA than the MA group. The proportion of men was higher in the MA than the MoA group; the control group fell between the two migraine groups in this respect. Distance visual acuity (as assessed during the laboratory visit) was converted to minimum angular resolution (min of arc). Although the range of acuity scores was similar across groups, median acuity was poorer in the migraine groups than in the control group, despite the fact that our preliminary screening eliminated anyone with known refractive errors of greater than −3.5 D. However, when compared using the Kruskal–Wallis test (one-way
Group characteristics

Smooth pursuit and predictable saccades
Group effects
The results of the
Results of split plot
Migraine pathophysiology could be reflected in within-subject variability, rather than affecting the average level of our measures. For example, control and migraine subjects might have equal average gains, but this could arise if migraineurs exhibited larger gains on some trials and smaller gains on other trials, rather than the consistently smaller gains we had predicted. We assessed this for pursuit gain by carrying out split-plot
Effect of target speed
Target speed exerted a significant effect on most of our measures (Table 2, speed column). Increasing the speed of the target led to a significant reduction in the gain of smooth pursuit (Fig. 2a). However, this reduction was compensated for by significant increases in both the number and the average size of catch-up saccades (Fig. 2b,c). Target speed also affected pursuit phase (Fig. 2d). Whereas all subject groups showed a small phase advance (anticipated the target position) for the slower target, all showed a small phase lag for the faster target condition.

Results for the four smooth pursuit measures collapsed across groups: (a) gain, (b) number of catch-up saccades, (c) amplitude of catch-up saccades, and (d) phase, with positive values representing phase advances and negative values, phase lags. In all cases, results are plotted against background visual pattern. Solid symbols represent responses to the 0.25-Hz sinusoidal stimulus; open symbols, the 0.5-Hz stimulus. Error bars = ±1 SE.
Target displacement rate also affected some measures of saccade performance (Fig. 3). Peak velocity was significantly higher and onset latency was significantly shorter for the rapidly alternating target than for the slower target jumps. The number of anticipatory saccades was also affected by displacement rate, with significantly more anticipatory saccades occurring in the fast condition. However, there is no evidence that saccade amplitude is affected by this variable (Fig. 3a).

Upper panels: saccade amplitude (a), peak velocity (b) and latency (c) plotted against stimulus background for predictable saccades. Solid symbols represent responses to the 0.25-Hz stimulus alternation rate; open symbols, the 0.5-Hz stimulus. Data are collapsed across groups. Lower panels: data for the three subject groups plotted against target displacement for the non-predictable saccade condition. Data from the 0.25-Hz Grey1 predictable saccade condition (20 pred) are plotted for comparison. (d) Saccade amplitude, (e) peak velocity, (f) latency. Error bars = ±1 SE.
Effect of background pattern
Background exerted a significant main effect on all measures of smooth pursuit performance (Table 2, background column). Pursuit gain was reduced against patterned backgrounds (P < 0.05) compared with the homogeneous grey conditions (Fig. 2a); however, performance on noise did not differ from performance on stripes (P = 0.61). The number of catch-up saccades mirrors the gain results in that more catch-up saccades occurred against patterned than against grey backgrounds (P < 0.05 in all cases) (Fig. 2b). However, contrary to the results for speed described above, the amplitude of catch-up saccades did not show a compensatory increase for patterned backgrounds (Fig. 2c).
Performance did not differ in the two grey background conditions for pursuit gain; however, both the size and number of catch-up saccades were significantly lower for G2 than for G1, indicating less effective compensation for image slip over the course of the test session. Pursuit phase was slightly but significantly more negative for G1 than for the other three background conditions, suggesting that practice may have improved subjects’ ability to track the target.
Predictable saccades were also affected by target background. In contrast to pursuit gain, saccade amplitude was larger against the patterned backgrounds than against either grey (P < 0.05; see Fig. 3a). Again, the effects of noise and stripes did not differ (P = 0.92). All other background effects for saccades failed to differentiate patterned backgrounds from grey, but pointed instead to order effects. Saccade amplitude was significantly higher and saccade latency was significantly shorter against G2 than G1 (P < 0.05), suggesting improved speed and accuracy over the session. However, peak velocity was significantly reduced in the G2 interval compared with G1 (P < 0.05).
Non-predictable saccades
Results for non-predictable horizontal saccades of 5–20° are shown in Fig. 3d–f. For statistical analysis (Table 2c), amplitudes were converted to gains (ratio of saccade amplitude/target displacement) to permit direct comparison across displacement sizes. Once again, there were no significant between-group effects on gain, peak velocity or latency of saccades and no significant interactions involving the group factor. However, the size of target displacement did elicit differential effects. Gains were slightly larger for the smallest (5°) and largest (20°) displacements than for the intermediate jump sizes. Peak velocity also varied with displacement size, which is not surprising, since peak velocity is normally highly correlated with saccade amplitude. Latencies to initiate 15° and 20° saccades were significantly greater than for either 10° or 5° displacements (P < 0.05).
Fixation
Each subject’s BCEA scores were averaged over the two fixation trials on striped backgrounds, one following the pursuit task and one following the saccade task. The data for each group were skewed with most scores falling below 6 sq deg, but with a long tail to larger values (Fig. 4a: cumulative distributions by groups; Fig. 4b,c: sample records). Because the data were highly skewed, the groups were compared using the non-parametric Kruskal–Wallis one-way
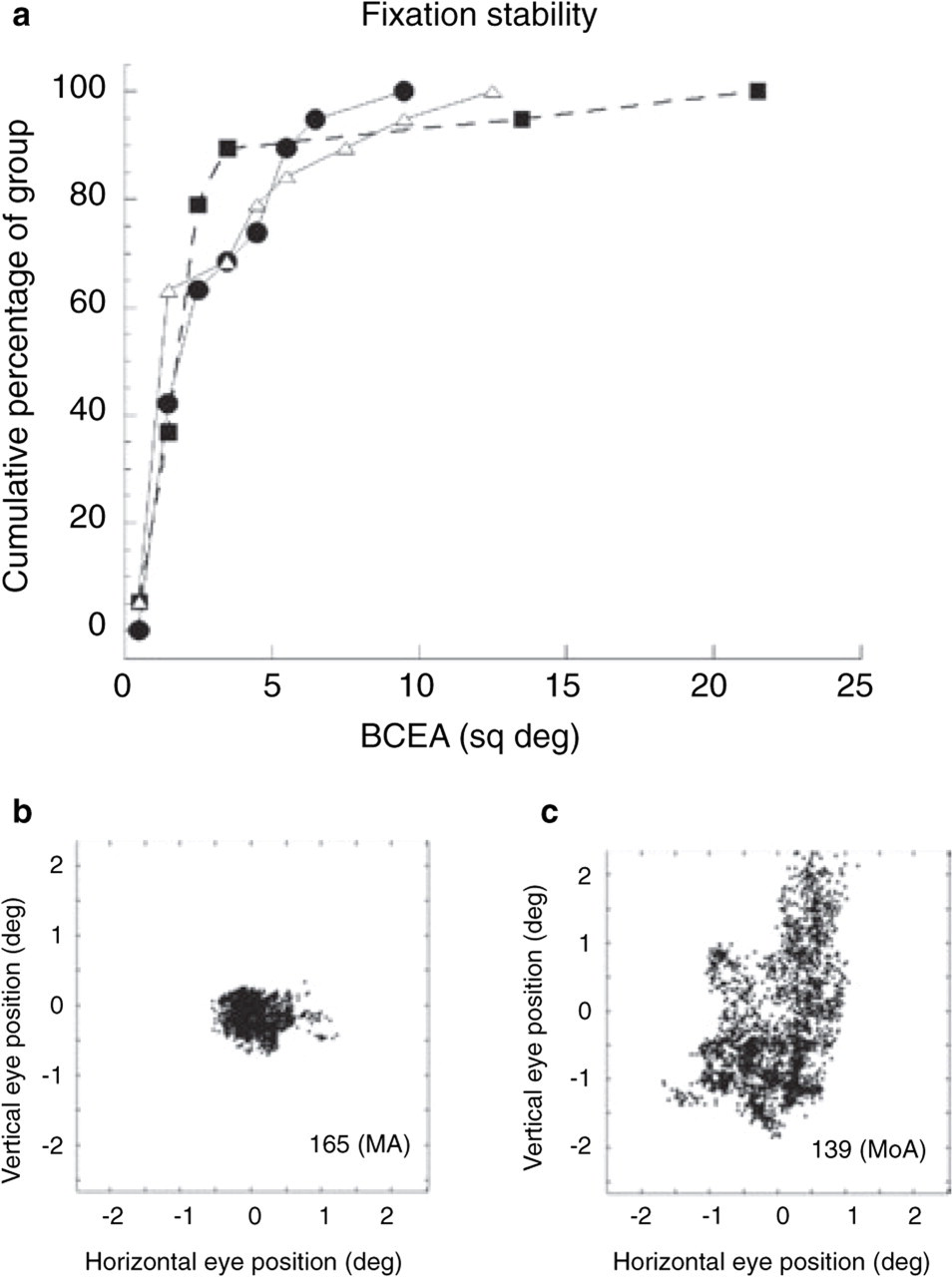
(a) The cumulative percentage of each of the three subject groups plotted separately against bivariate contour ellipse area. Over 90% of each group had area of a bivariate contour ellipse (BCEA) scores of <6 sq deg. •, MoA; j, MA; ▵, C. (b, c) Records of eye position distributions illustrating good and poor fixation stability in two migraine subjects. Each dot represents one eye position measure. Sampling rate = 120 Hz.
Outlier analysis
We next asked whether abnormalities on any of our measures might be manifested by only a subset of migraineurs. To do this, we identified scores that fell more than ±2 SD from the mean (all groups combined) for each measure. An individual was considered an outlier on a particular measure (e.g. pursuit gain) if their score met this criterion on at least four of the eight relevant test blocks.
Twelve individuals were identified as outliers (three MoA, six MA, three C), each on a single measure. This differential distribution across groups was not statistically significant (χ2 = 1.9, d.f. = 2, P = 0.39). There was no clustering of outliers from any group on a particular task. Two MA and one C subject showed markedly abnormal ability to maintain fixation. Two MA subjects had a strong tendency to anticipate the trajectory of the pursuit target. Two individuals (one MoA, one C) showed abnormally low pursuit gain; both scored at the high end of their groups on the number and/or amplitude of corrective saccades, indicating that they were able to compensate for their impairment. One individual (MA) showed consistently hypermetric saccades, whereas two (one MoA, one C) had consistently hypometric saccades. The remaining two outliers also had abnormal saccades: one (MA) had long saccade latencies and the other (MoA), high peak velocities.
The outliers in the migraine groups did not have significantly longer duration of migraine (14.64 vs. 13.99/year) or more frequent episodes (1.96/month vs. 2.06/month) than the rest of their group members. Nine of the 12 outliers were either older than 40 years (n = 2), had visual acuity of ≥5 min of arc (n = 5), or both (n = 2), suggesting that these two factors may have played an important role in their performance. If the outlier scores were due to causes other than migraine condition, their inclusion in the analyses might have masked true group differences by increasing group variability. To check this possibility we reran all analyses described above with the outliers’ data omitted. Again, we found no significant main effects of group or interactions of group with other variables.
Visual acuity and family history
Two issues about our group composition were of some concern to us. As described above, there was a non-significant trend toward poorer distance acuity in the MoA and MA groups than in the controls, and the majority of outliers independent of group fell near the upper end of our acuity range. We also noted that six members of our control group who had never experienced migraine themselves nevertheless reported the presence or possible presence of migraine in their immediate family (mother or sibling). To address these issues, we took the remaining 13 members of the control group (those with no family history of migraine) and matched each of them to one member of the MA group and one member of the MoA group based on acuity score, using the criterion that the first individual recruited to the study who met the criterion was selected (matching was done without knowledge of the eye movement results). This yielded three groups of 13 individuals well matched in terms of acuity scores (MoA 2.5 ± 1.9; MA 2.5 ± 2.1; C 2.3 ± 2.0) and age (MoA 32.8 ± 9.0; MA 34.2 ± 10.2; C 33.3 ± 8.5 years). We reran all the split plot
Discussion
Our findings have failed to provide any evidence of ocular motor abnormality in migraine, at least under the conditions of our study (horizontal pursuit of sinusoidal target motion and comparable horizontal saccades). Not only did we find no evidence of differences in the mean performance of migraine groups relative to controls, we also found no greater incidence of extreme scores (outliers) in the migraine groups, and no evidence of greater within-subject variability on the measures for which this could be meaningfully tested. Thus, the migraineurs in our study did not show impaired, enhanced or more variable performance on our measures relative to the headache-free controls. It is, of course, not possible to ‘prove’ that no difference, however small, exists between our groups. In the following, we consider the ways in which such a difference might have gone undetected and the likelihood that this occurred by assessing the validity and sensitivity of our measures and the representativeness of our subject sample.
Validity
Data from our baseline smooth pursuit condition (slow target, grey background) revealed a mean gain of 0.95, averaged over all subjects. Mean phase in this condition was 1.47°, which indicated that subjects rapidly caught up with the target and, in fact, over most cycles their eyes actually anticipated the target movement by a tiny amount (perfect tracking would yield a phase of 0°). These values are consistent with those reported for normal subjects in other studies using similar sinusoidal targets (21, 24, 29, 32, 33).
Classic studies of saccades have demonstrated that normal human subjects very frequently show undershooting of visual targets by about 10% ((34, 35) but see (36)). Thus, our accuracy measures of 91% for both predictable and non-predictable saccades are well within the normal range. The average peak velocity for these saccades was 385.1°/s (SD = 47.6), and their mean latency was 188 ms (SD = 39); again, these values are very close to values reported in the literature for saccades of similar amplitude under similar stimulus conditions (19, 36–39).
Sensitivity: effect of displacement frequency (speed)
In mild disorders, behavioural measures may appear normal under relatively undemanding test conditions and deficits may be revealed only under conditions that push the subjects closer to the limits of the system. In the present study, increasing the displacement rate of the targets provided such a condition. Our results clearly indicate that this manipulation had a marked effect on smooth pursuit, resulting in phase lags and dramatic reductions in pursuit gain (see Fig. 2). These effects were compensated for by more frequent and larger catch-up saccades. Our measures were sufficiently sensitive to detect these differences. Nevertheless, no significant interactions were seen between speed and group membership as would have been expected if this manipulation had uncovered a subtle impairment in migraine.
Representativeness of the sample
We included in our exclusion criteria all prophylactic drugs for migraine (including herbal remedies such as feverfew). Since it is those most severely affected in terms of frequency or severity of episodes who are likely to be on prophylactic migraine treatments, it is possible that our sample reflects the milder end of the spectrum. However, in terms of disease duration and episode frequency, our MoA group is very similar to those studied by Sándor et al. (16) in their analysis of reaching movements. Our MA group is also very similar in terms of disease duration but had less frequent attacks (0.9 vs. 2/month). Because it is well recognized that eye movements, particularly smooth pursuit, are affected by ageing (20–22), we did not test individuals above 52 years of age, and matched our groups carefully on this variable. The mean age of our groups (32–33.5 years) and their range again are quite comparable to those studied by Sándor et al. (16) but considerably less than the groups in two previous ocular motor studies (17, 18). We encountered surprising difficulty in recruiting subjects in all groups who were entirely free of refractive error. Since this was unexpected, we did not make an effort to match subjects precisely on this variable, although we did screen out more than 40 individuals with refractive errors that we considered significant enough to affect performance on our tasks. Despite a small remaining bias toward poorer visual acuity in the migraine groups, we are confident that this factor did not confound our results. None reported difficulty in seeing the target or the background patterns at the test distance of 57 cm, which is about arm’s length. When we compared acuity-matched subgroups, the pattern of findings remained unchanged. Most importantly, refractive errors would have been expected to compound the deficits we had predicted but did not find in the migraine groups.
Comparison with other studies
Previous studies (17, 18) have reported some abnormalities in the eye movements of migraineurs, so it is important to consider the possible sources of the discrepancy with the present findings. Both prior studies used clinical samples and, in both cases, the subjects were older than ours (mean ages differed by approximately 10 years). Wieser et al. (18) were motivated by the observation of an apparent pursuit disturbance in their patients, so it is not clear what degree of selection bias may have affected their migraine sample. The only statistically significant group difference reported in their study was for pursuit phase. The phase shifts were very large (MoA medians 11–51° across all conditions) and appear to be phase advances, although the direction is not specified. By contrast, our subjects in all groups showed much tighter phase-locking to the target (range of phase lags and advances −9.4° to +15.9°; means and medians close to 0° for all groups). The authors also noted a trend toward lower gains in MoA than controls; these differences were not, however, statistically significant. Moreover, the pursuit gain data in both control and MoA groups were surprisingly low for the stimulus parameters used (median gains 67–77%). In the present study, means and medians were in the 85–95% range for comparable stimulus conditions, values consistent with the existing literature (see above). However, our data are in close agreement with Wieser et al. in showing comparable saccade amplitudes in migraineurs and controls, with both groups undershooting the target slightly (approximately 90% gain). We speculate that the differences in pursuit are due to a combination of subject age and the use of electro-oculographic measurements of eye movements, a technique susceptible to artefact (see, for example. (40)).
Harno et al. (17) did not measure smooth pursuit, but looked instead at saccades and at a variety of vestibulo-cerebellar measures including nystagmus, caloric responses, postural sway and audiology. They found significant numbers of migraineurs with abnormalities on each of these measures. In terms of saccades, they describe significantly larger dysmetrias in migraineurs than in controls, with both hypometria and hypermetria occurring with equal frequency in their migraine groups. In contrast, all of our groups showed a slight hypometria in all conditions (mean 1.4°) that did not differ across groups. Only four of our 57 subjects (two MA, two C) showed consistent hypermetria across saccade conditions, the largest of which averaged only 1.2°. Not only were their groups considerably older than those in the present study (group means 40–47 years vs. 32.1–33.4 years) but all subjects showing abnormally large dysmetrias were over 40 years of age, three of them between 68 and 71 years. The studies differed in several other respects. Their MA group included cases of basilar migraine, hemiplegic migraine and familial hemiplegic migraine as well as simple migraine with aura (who were a minority of the group). Both MoA and MA groups contained more than one member from several families with strong family histories of migraine. Finally, a considerable number of the migraine patients were on β-blockers or prophylactics for other conditions, which suggests that they may have had relevant comorbid conditions.
In contrast, the groups studied by Sándor et al. (16) in their arm movement study are much more comparable to ours in terms of subject age, control of prophylactic medication, IHS subcategories included, and the nature of their analysis of movement dynamics. Whether theirs is a more sensitive measure of subclinical cerebellar impairment than ours or whether the difference arises from dissimilar migraine populations cannot be answered here. While it is also possible that the difference is due to involvement of different cerebellar subsystems in eye and arm movement control, this seems a less likely explanation since familial hemiplegic migraine, the condition which gave rise to the calcium channelopathy model of migraine, is indeed characterized by abnormal eye movements (13). It would therefore be of great interest to test the same individuals using eye and arm movement analyses.
The present study was undertaken to establish a baseline of the most fundamental classes of human eye movements and of fixation stability. While parts of the cerebellum have been implicated in all of these functions (see (19) for review), it may be that these systems are highly resilient or that compensation occurs. Having established this baseline normality, it will now be possible to examine ocular motor tasks that are more demanding of flexibility or learning, such as saccadic adaptation, since the involvement of the cerebellum in such adaptive function is well established (41, 42).
It should be noted in closing that there is another class of visual-motor function for which there is increasing evidence of abnormality in migraine: vestibulo-ocular coordination. Vertigo is a frequent accompaniment to migraine and may exist alone as a migrainous condition (43, 44). Several recent studies have provided evidence of nystagmus, abnormal postural sway and abnormal vestibulo-ocular reflex in MA and MoA interictally (17, 45) and during episodes of migrainous vertigo (44). Moreover, painful trigeminal stimulation has recently been shown to elicit nystagmus in MoA interictally but not in control subjects (46). It is unclear whether these vestibular effects are of central or peripheral origin, or whether this varies across individuals. However, in view of the present findings, brainstem vestibular nuclei, cortical regions of visual-vestibular overlap (47) and/or peripheral vestibular structures seem more likely sites of abnormality than the vestibulo-cerebellum, although the latter cannot be ruled out.
Visual implications
In studies of normal subjects, visual background has been shown to exert a significant influence on the gain of pursuit movements. While bright targets can be equally effectively tracked in the dark and against homogeneous grey backgrounds, introducing structure to the background creates a drag on pursuit gain (24, 25), reducing gain by approximately 10%. The present results are consistent with this pattern (Fig. 2a). Because of the documented sensitivity of migraineurs to repetitive high-contrast visual patterns, we predicted that backgrounds consisting of visual structure, especially stripes, might produce even stronger drag effects in migraine groups. This was not found to be the case: migraineurs and controls showed equivalent gain reduction in the presence of visual structure and equivalent compensation in terms of number of catch-up saccades.
Finally, we found no differential effect of background on saccade measures in migraineurs compared with controls, and no difference among our three groups in terms of the stability of their fixation of a target dot against the striped background. The striped background did elicit reports of illusions from more than half of each of our subject groups (MoA 53%, MA 68%, C 59%). In this study we did not explicitly evaluate visual discomfort (the degree to which individuals find such patterns to be aversive or even painful), although individual subjects did indicate that it was unpleasant. However, our results suggest that the discomfort often reported by migraineurs in the presence of such high-contrast periodic stimuli is not a product of apparent background motion arising from poor fixation stability. Many measures considered to be tests of visual function can be adversely affected by abnormal eye movements or abnormal fixation. Thus, our findings provide reassurance that previously reported visual abnormalities in migraine really do have their origin in the visual pathways and not in the ocular motor system.
Footnotes
Acknowledgements
This research was supported by the Canadian Institutes of Health Research (grant no. 49596), the Krembil Foundation, Toronto, Canada, the Natural Sciences and Engineering Research Council of Canada (no. A7664) and the Sir Jules Thorn Foundation. We are grateful to Alan Blakeman for making his pursuit analysis software available to us.

