Abstract
The aim of this study was to investigate increase of QTc dispersion and P-wave dispersion during migraine attacks. Fifty-five patients (16–65 years of age, 49 women, six men) with migraine were included in our study. Heart rate, QTc interval, maximum and minimum QTc interval, QTc dispersion, maximum and minimum P-wave duration and P-wave dispersion were measured from 12-lead ECG recording during migraine attacks and pain-free periods. ECGs were transferred to a personal computer via a scanner and then used for magnification of x400 by Adobe Photoshop software. Maximum QTc interval (454 ± 24 ms vs. 429 ± 23 ms, P < 0.001), QTc interval (443 ± 26 ms vs. 408 ± 22 ms, P <0.001) and QTc dispersion (63 ± 18 ms vs. 43 ± 14 ms, P <0.001) were found significantly higher during migraine attacks compared with pain-free periods. Maximum P-wave duration (107 ± 11 ms vs. 100 ± 11 ms, P <0.001) and P-wave dispersion (45 ± 13 ms vs. 35 ± 13 ms, P <0.001) were found higher during migraine attacks than pain-free periods. We concluded that migraine attacks are associated with increased QTc and P-wave dispersion compared with pain-free periods.
Introduction
Migraine is a very common illness characterized by nausea, vomiting, headache, photophobia, phonophobia and dizziness (1). The autonomic nervous system (ANS) is involved in the pathophysiological mechanism of migraine because autonomic symptoms are common during acute migraine headaches (2).
Dysfunction of the ANS may affect atrial and ventricular repolarization, as prolonged QT intervals appear to correlate with the severity of autonomic dysfunction in various diseases (3, 4). Therefore, disrupted autonomic innervation of the heart and coronary arteries in patients with migraine may cause possible electrocardiographic (ECG) abnormalities during headache (5). It has been reported that this disrupted innervation may be associated with an imbalance between the sympathetic and parasympathetic systems (6, 7). Migraine case–control studies of cardiovascular function have reported both sympathetic hypofunction (8–10) and sympathetic hyperfunction (11, 12).
The QTc dispersion has been suggested to be an electrocardiographic index reflecting the physiological variability of regional ventricular repolarization. Increased QTc dispersion and prolonged QT interval have been reported in a number of cardiovascular diseases linked to increased risk of ventricular arrhythmias and sudden cardiac death (13–15). In addition to QTc dispersion, P-wave dispersion has been recently reported to be a predictor for paroxysmal atrial fibrillation (16). P-wave dispersion has been associated with inhomogeneous and discontinuous propagation of sinus impulses (16, 17). Prolongation of intra-atrial and interatrial conduction time and inhomogeneous propagation of sinus impulses are known electrocardiological characteristics of the atrium prone to fibrillation (16, 17).
Cardiac arrhythmias in patients with migraine have been reported (7, 8, 18). However, data concerning QT interval changes during migraine attacks are limited. There is also little information about the increase of P-wave dispersion during migraine attacks compared with pain-free periods.
We hypothesized that QTc and P-wave dispersion may increase in patients with migraine because of disrupted ANS. The aim of this study was to investigate the increase of QTc dispersion and P-wave dispersion during migraine attacks.
Methods
The study consisted of 55 patients (16–65 years of age) with migraine who were consecutively admitted with headache to the emergency unit in our university hospital. All the patients were eligible for criteria migraine with or without aura by the criteria of the International Headache Society (IHS) (19). Pain severity was defined as follows: scale 0 = no pain, scale 1 = mild pain, scale 2 = moderate pain and scale 3 = severe pain. Patients were screened by medical history, physical examination and clinical laboratory tests (complete blood count, blood biochemistry, urinary analysis). Patients with thyroid dysfunction, anaemia, electrolyte imbalance, uncontrolled hypertension, diabetes mellitus, heart failure, coronary artery disease, taking migraine-specific medication during migraine attacks and left bundle branch block, atrioventricular conduction abnormalities on ECG were excluded. All patients were in sinus rhythm and none of them was taking medications such as antiarrhythmics, tricyclic antidepresants, antihistaminics and antipsychotics. Blood pressure was measured every 30 min and averages of these were taken. Twelve-lead ECG was recorded for each patient within the first 60 min of arrival and monitored in the emergency unit until a pain-free period. ECG abnormalities such as atrial and ventricular arrhythmias, sinus bradycardia and tachycardia, and adverse events were recorded during monitoring. The ECG during the pain-free period was recorded after at least 24 h from a headache. No patient was kept in the emergency department for a pain-free period ECG record. The 12-lead ECG was recorded at a paper speed of 25 mm/s and gain of 10 mm/mV (Cardiofax V; Nihon Kohden Corp., Tokyo, Japan) with the papient in the supine position and breathing freely but not allowed to speak during the electrocardiographic recording. All of the ECGs were transferred to a PC via a scanner and then used for ×400 magnification by Adobe Photoshop software. The QT interval, which is the duration between the beginning of QRS complex to the end of the T-wave, was measured in all derivations in which T-wave was clearly seen and not mixed with a U-wave. In all patients, derivations in which the beginning and end-point of QT could not be distinguished were excluded. The starting point of P-wave was referred as the positive deflection crossing the iso-electric line and the end-point was referred as the end of the deflection crossing the iso-electric line. P-wave duration was measured from the onset to the offset of the P-wave. Patients were excluded if these points were not clear.
QT dispersion was defined as the difference between the longest QT interval (QTmax) and shortest QT interval (QTmin). Measured QT intervals were corrected by Bazett’s formula (20) (QTc = QT/√RR) and defined corrected QT interval (QTc). Prolongation of the QTc interval was defined as ≥440 ms. The difference between the longest QTc (QTcmax) and shortest QTc (QTcmin) interval was defined as QTc dispersion. The maximum and minimum P-wave durations were measured. P-wave dispersion, defined as the difference between the maximum P-wave duration and the minimum P-wave duration, was also calculated. All measurements were repeated three times and average values were accepted for each of the electrocardiographic parameters. All measurements were performed by two experienced investigators blind to the patients’ clinic status. The study was approved by the local ethics committe. All patients gave informed consent.
Statistical analysis
SPSS 11.0 software was used (SPSS Inc., Chicago, IL, USA). Continuous variables were expressed as mean values ± SD. Correlations were performed using Pearson’s correlation coefficient. The paired-sample t-test was used to evaluate the relative differences between attacks and pain-free periods for each ECG parameter. Relationships between age, aura and duration of disease during attacks and pain-free periods were assessed by simple linear regression analysis. P-values < 0.05 was considered statistically significant.
Results
Fifty-five patients (49 women, six men; mean age 32 ± 10 years) were included in the study. Thirty-one (56%) patients had moderate headache and 24 (44%) had severe headache. Baseline clinical characteristics of patients are shown in Table 1. During the recording of the ECGs, only non-narcotic analgesics were given to the patients for treatment of pain. Forty-four (80%) patients were taking medication for migraine prophylaxis, but none of them was on any medication during migraine attacks. The drugs that patients used for migraine treatment are shown in Table 1. Blood pressure was not different between migraine attacks and pain-free periods (Table 1). However, the heart rate was higher during migraine attacks compared with pain-free periods (Table 2).
Baseline clinical characteristics of patients
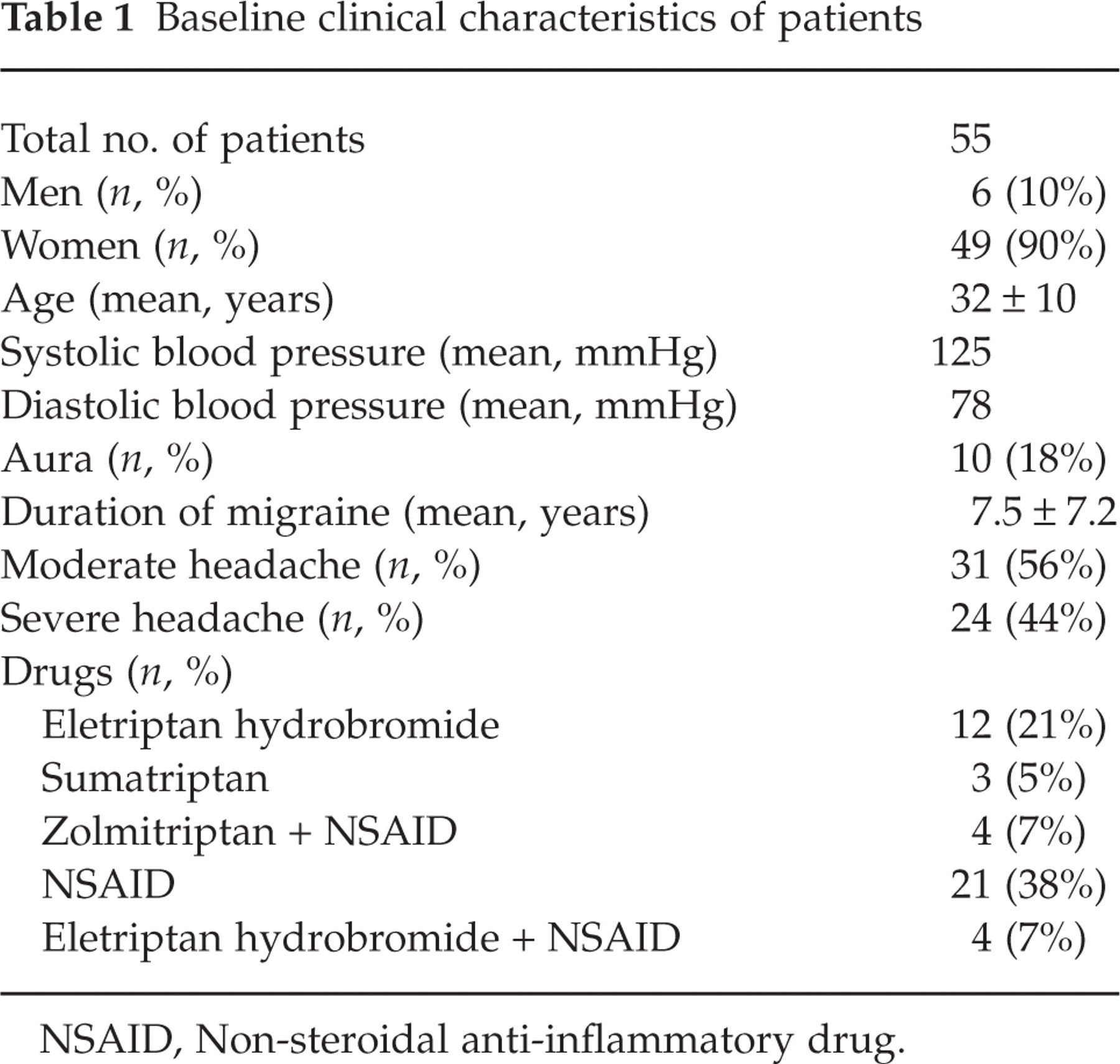
NSAID, Non-steroidal anti-inflammatory drug.
Electrocardiographic parameters during migraine attacks and pain-free periods

CI, Confidence interval; SD, standard deviation.
QT dispersion and P-wave dispersion
Maximum QTc interval (454 ± 24 ms vs. 429 ± 23 ms, P < 0.001), QTc interval (433 ± 26 ms vs. 408 ± 22 ms, P < 0.001) and QTc dispersion (63 ± 18 ms vs. 43 ± 14 ms, P < 0.001) were found significantly higher during migraine attacks compared with pain-free periods. However, there was no significant difference between the minimum QTc interval (390 ± 26 ms vs. 386 ± 21 ms, P > 0.05) during attacks and during pain-free periods (Table 2). The maximun P-wave duration (107 ± 11 ms vs. 100 ± 11 ms, P < 0.001) and P-wave dispersion (45 ± 13 ms vs. 35 ± 13 ms, P < 0.001) were found to be higher during migraine attacks than during pain-free periods. The minimum P-wave duration (62 ± 11 ms vs. 64 ± 12 ms, P > 0.05) was not different between migraine attacks and pain-free periods (Table 2). Prolonged QTc interval was detected in 24 of 55 patients during migraine attacks, while it was detected in five of 55 subjects during pain-free periods. This finding was statistically significant (P < 0.001) (Fig. 1). In addition, there was not correlation between electrocardiographic parameters (QTc and P-wave dispersion) and age (r =−0.159, P = 0.096 and r = 0.005, P = 0.955), and duration of migraine (r =−0.134, P = 0.164 and r = 0.018, P = 0.852, respectively).

Increased QTc interval during migraine attacks compared with pain-free periods. ▪, >440 ms; □, ≤440 ms.
Discussion
Migraine is a disorder with multiple manifestations affecting the circulation, gastrointestinal tract and the central nervous system (21). Autonomic symptoms including nausea, vomiting, diarrhoea, cutaneous vasoconstriction (pallor), vasodilation (flushing), pilo-erection and diaphoresis are very common in patients (22). It has been suggested that these inconsistent findings may be explained by varied autonomic dysregulation, perhaps the results of an imbalance between the sympathetic and parasympathetic systems (6). Appel et al. (7) postulated both sympathetic hyperfunction and a concominant parasympathetic hypofunction in patients with migraine. The rich autonomic innervation of the heart plays an important role in modifying cardiovascular function. For example, increased sympathetic activity causes sinus tachycardia, but increased parasympathetic activity causes sinus bradycardia, atrioventricular block and ST-segment and T-wave abnormalities. Previous studies have reported ECG abnormalities such as sinus bradycardia, premature ventricular contraction (PVC), non-specific ST-T changes and right bundle branch block using lead-II ECG monitoring during both migraine attacks and pain-free periods (18). Pogacnik et al. (8) have reported an increased frequency of cardiac arrhythmias using single-lead ECG monitoring during attacks compared with pain-free periods. Therefore, dysfunction of the ANS may affect atrial and ventricular repolarization in patients with migraine. In various studies, prolonged QT interval has been reported in other disorders of the central nervous system (23–25). However, ventricular repolarization abnormalities during migraine attacks have been less studied. Recently, Aygun et al. (26) reported increased QTc and PR intervals during migraine attacks from pain-free periods. In contrast to the QTc interval, P-wave dispersion has not yet been studied during migraine attacks. In our study, we found higher QT interval parameters (QTc interval, maximum and minimum QT duration and QTc dispersion) and P-wave dispersion during migraine attacks than during pain-free periods. However, the increase of PR interval was not found different between migraine attacks and pain-free periods in our study. Increased heart rate during attack and the different measurement techniques (manual or PC-based measurement) may effect the PR interval. In our study, we did not use manual measurement. Compared with PC-based measurement, manual measurement of QT and P-wave parameters performed on standard paper-printed ECGs is of limited accuracy. It has been reported that scanning and digitizing ECG signals from paper records using an optical scanner is a feasible and accurate alternative method (27).
We believe that migraine attacks may be due to a disrupted ANS (sympathetic and/or parasympathetic nervous system dysfunction). It has been reported that dysfunction of the ANS influences ventricular repolarization and therefore increases the QT interval on the ECG (24, 28). Tukek et al. (29) have reported that increased sympathetic activity causes a significant elevation in P-wave dispersion. In addition to dysfunction of the ANS, drugs that patients use can affect ECG parameters, as angina pectoris due to coronary vasospasm following migraine treatment is more commonly reported (30, 31). However, none of the patients in our study suffered from chest pain or was on any medication during migraine atttacks.
Previous studies have reported the association between the ANS and atrial fibrillation (32, 33). Information on the association between atrial fibrillation and migraine is also limited in case reports (34, 35). The sympathetic nervous system is known to play an important role in the genesis of ventricular arrhythmias (36). However, there is no evidence of increased incidence of ventricular arrhythmias and sudden death in patients with migraine. This could be explained by the younger age of subjects and by patients with migraine being free of cardiovascular risk factors.
Our study has several limitations. There was no healthy control group in our study. We could not evaluate the difference between a healthy control group and the patients group (during attacks and pain-free periods). Twenty-four hour rhythm Holter that is needed to detect rare arrhythmias is not used. Further studies are needed to investigate the prevelance of atrial and ventricular arrhythmias during attacks in patients with migraine who have increased QTc and P-wave dispersion by using a 24-h rhythm Holter or event recorder.
We conclude that patients with migraine during attacks are associated with increased QTc and P-wave dispersion. These findings may be related to a disrupted autonomic nervous system in migraine patients. Therefore, we can estimate that patients with migraine during attacks may be under risk for atrial and ventricular arrhythmias.
