Abstract
Introduction
Knowledge of the clinical features of cluster headache is mainly based on retrospective and cross-sectional studies. Here, we present a case of a chronic cluster headache patient who prospectively recorded timing and clinical features of all attacks for 6 years, aiming to describe the clinical spectrum and timing of cluster headache symptoms experienced and to identify daily and/or seasonal rhythmicity.
Methods
Registration of attack timing, duration, associated symptoms and severity was done prospectively on a smartphone application. Pain severity was recorded on a 0–10 scale. Attacks were divided into mild, moderate, severe, and very severe. We analysed diurnal rhythmicity by multimodal Gaussian analysis and spectral analysis.
Results
In total, 4600 attacks were registered (mean duration 39.3 (SD 18.5) min. Mean severity 3.6 (SD 1.28)). Mild attacks accounted for 14.2%, moderate 65.7%, severe 16.9% and very severe 3.2% of all attacks. Nocturnal attacks were more severe than daytime attacks. The number of autonomic symptoms and duration of attacks increased with pain severity. Peak chronorisk (risk of attacks occurring according to hour of day) was at 12.48 in the registration period. Over time, circadian rhythmicity and attack frequency varied.
Conclusion
Clinical characteristics of cluster headache attacks can vary greatly within the individual patient. Clinicians attempting to personalise the administration of preventive treatment should pay notice to the variation over time in diurnal rhythmicity. The recorded self-limiting mild attacks that do not fulfill the ICHD-3 criteria for a cluster headache attack warrant further investigation, as they could hold important information about disease activity.
Introduction
Cluster headache (CH) is characterised by one-sided, short-lasting attacks of unbearable, periorbital pain accompanied by cranial autonomic symptoms (CAS) and/or a sense of restlessness (1). Given the severity, CH can lead to profound personal, social and economic consequences (2,3). CH is divided into a chronic (cCH) and an episodic (eCH) phenotype, the first being characterised by periods of remission lasting less than 3 months over one year and the latter by attack-free periods lasting at least 3 months (1). The pathophysiology of CH is not fully elucidated, but imaging findings, the trigeminal distribution of pain and the fact that CH shows striking chronobiological features suggest trigeminal-hypothalamic interplay as an important component (4–6). Daily and seasonal rhythmicity in CH, possibly influenced by both daylight and daily life rhythms, is a hallmark of eCH, but patients suffering from cCH also report both diurnal and to a lesser extent circannual attack dependency (7–14). Results have been contradictory, and further description of rhythmicity in the literature is still warranted as preventive treatment administered on the basis of diurnal rhythms (chronotherapy) might be the next step towards personalising CH treatment (14).
Evidence of the clinical features and chronobiological rhythms of CH has predominantly been based on retrospective data and mainly focused on describing a “typical” attack, thereby not accounting for the intra-patient variability and temporal development of attacks (3,8,9–17). Furthermore, a growing body of evidence pinpoints interesting aspects of CH not pertaining to the headache phase itself. Firstly, there is increasing evidence of post- and pre-ictal CH activity (15,18–20). Secondly, it is our clinical experience that patients can have episodes of minor painful sensations, with or without autonomic symptoms in the area of their usual attacks, a scarcely documented aspect of CH (16,21,22). Documenting CH activity that does not strictly fall within the ICHD classification may ultimately hold the potential of improving diagnostics and treatment (1). We present a cCH patient who prospectively and systematically registered the clinical features of 4600 attacks between 2011 and 2017. We aim to describe the intra-patient variability in the clinical characteristics of attacks together with 24-hour and seasonal CH rhythmicity over time.
Methods
Data was collected using a dedicated smartphone application (My Headache Log Pro, by Solar Embedded). The registered data was: Time, duration, maximum pain severity, location when having an attack, CAS, restlessness/agitation, mild pain in the subsequent attack area and treatment.
Recordings of duration were initiated at the time of noticing the first CH symptom and terminated when symptom free. Severity was registered on a numeric rating scale (NRS) scoring pain from 0–10. An extensive medical interview and a timeline of major life events (not shown) was conducted and medical records were accessed with permission, to document the medical history. A written consent was obtained.
For descriptive purposes, we divided the attacks into four categories of severity: Mild (NRS 1–2), moderate (NRS 3–4), severe (NRS 5–6), and very severe attacks (NRS 7 + ).
Descriptive data is presented by means and standard deviation (SD) when suitable. Time of day is reported in 24-hour military time. Twenty four-hour variation of attack frequency was analysed by multimodal Gaussian modeling and spectral analysis as described by Barloese et al. (14). The 24-hour chronorisk probability distribution of attacks represents the probability for an attack by hour of day. Gaussian modeling quantifies chronorisk peak times, amplitude and spread, thereby identifying hours of elevated risk of attacks. Goodness of fit was evaluated with R2 statistics. The magnitude of chronorisk attributable to one daily oscillation (circadian oscillation) and to several daily oscillations (ultradian oscillations) were identified by spectral analysis, applying a discrete Fourier transformation (14). For comparative purposes, periods of high attack frequency were defined as being coherent months with attack frequency above the average (1.9 attacks a day) terminated by at least 3 months with attack frequency equal to or below average. The effect of preventive treatments attempted during the registration period was evaluated by comparing attack frequency, average duration and average severity 30 days prior to initiation with the periods after initiation (up to 30 days after).
Systematic reductions (two or more) exceeding 20% would be regarded as an indicator of possible effect of treatment. Correlation between sunlight exposure and attack frequency was calculated using Spearman rank correlation. Statistical analyses were performed using GraphPad Prism 7.02 and MATLAB version 8.5.
Case story
The patient is a 34-year-old (2018) Danish man of mixed Caucasian and Inuit origin with no history of serious head trauma, neurological disease or other primary headache disorders. He first experienced a cluster period at age 7. Being otherwise headache free through childhood and adolescence, the CH attacks returned at age 20 and he was diagnosed 3 years later. Magnetic resonance imaging of his brain, performed at the time of diagnosis, and neurological examination were normal. From 2003–2010, the patient displayed an episodic pattern (ICHD-2), with a maximum of two cluster bouts a year, each lasting approximately 5 months, separated by remission periods lasting between 2 months and 1 year. In 2010, the patient progressed to fulfill the ICHD-3 (and ICHD-3 beta) criteria for cCH (1,23). The diagnosis of cCH was confirmed by a headache specialist from the Danish Headache Center and the patient fulfilled the cCH criteria until the end of registration.
Prior to the collection of the presented data, prophylactic treatments with verapamil were initiated (maximum dose 400 mg), which had a substantial effect on pain intensity but not attack frequency. Due to nightmares and disturbed sleep that disappeared with termination of treatment and reoccurred when reinitiating treatment, verapamil was abolished. Furthermore, treatment with lithium, gabapentin, indomethacin, propranolol, melatonin, topiramate, and steroids were tried and subsequently discontinued due to either side effects or lack of efficacy. Prior to, and in the early months of registration, triptans were tested in oral and injectable parental formulation. Although effective, triptans were discontinued due to side effects and a superior effect of oxygen treatment (Figure 1). In February 2013, the patient underwent implantation of a sphenopalatine ganglion (SPG) neurostimulation device. During five periods between 2013–2016, SPG stimulation for 15–25 min a day was applied as prophylactic treatment and 15 minutes' stimulation as acute treatment; it was, however, only systematically applied during the period lasting until January 2014 due to the lack of treatment response. During periods of 2015–2017, prophylactic treatment with mushrooms containing psilocybin was tested 1–4 times a month on his own initiative, though with no effect. The preferred, and only effective/tolerable abortive treatment was oxygen 10–25L/min delivered by non-rebreather and Demand Valve Oxygen mask. With oxygen constantly at hand, allowing early initiation of attack treatment, the patient obtained good control of pain intensity.
Monthly attack fruequncy and treatment. *Until January 2014 oxygen was delivered by non-rebreather mask, after that, by demand valve oxygen mask. **Data missing. SPG: sphenopalatine ganglion neuro stimulation. Pc.: psilocybin.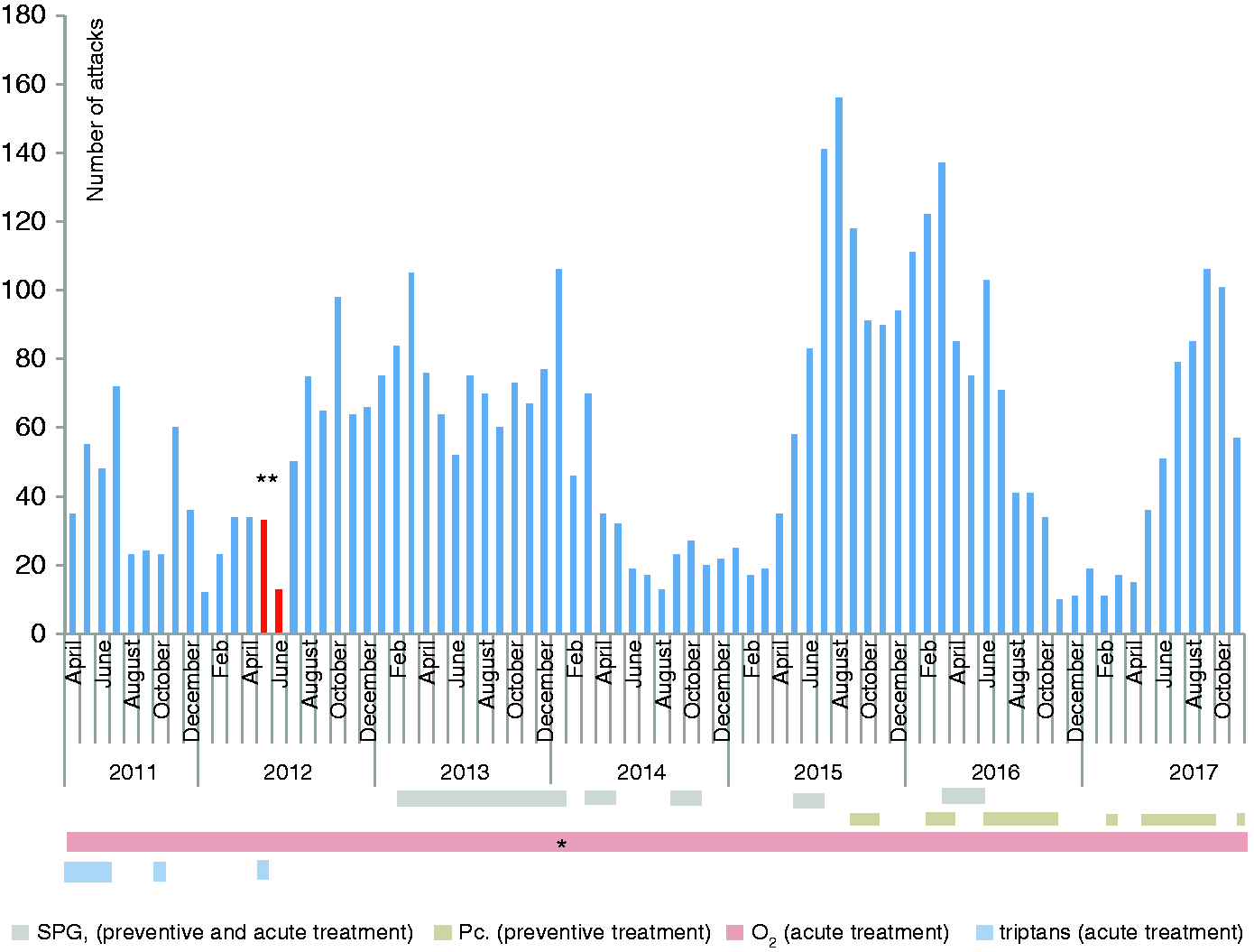
Results
From April 2011 until November 2017, a total of 4600 attacks were recorded. Recordings of some attacks were lost due to technical problems during May and June 2012. Four attacks had missing data on autonomic symptoms and date.
Clinical characteristics of attacks
The mean severity of attacks was 3.6/10 (Range 1–9), duration was 39.3 (SD 18.5) min and daily frequency was 1.9 a day (SD 1.3). Of all attacks, 77.4% were treated with oxygen. Eighty percent of attacks occurred while the patient was at home.
The attacks, by category of severity, duration, number of CAS and associated restlessness and/or agitation, are depicted in Figure 2. The attack duration and likelihood of CAS increased with the severity of the attacks. The most frequent autonomic symptoms were nasal congestion and lacrimation followed by eyelid edema, conjunctival injection and (the least prevalent) facial sweating. Prior to 96.4% of moderate, severe and very severe attacks, the patient registered having mild pain in the subsequent attack pain area. Acute treatment with oxygen was registered in 23.8% of mild CH attacks, 77.2% of moderate CH attacks, 94.7% of severe and 96.9% of very severe CH attacks. Adding the remaining abortive treatments, the percentage of attacks treated were 30.4%, 81.8%, 95.6% and 97.3% for mild-, moderate- severe- and very severe attacks respectively.
24-hour rhythmicity
Attacks at night were more severe than attacks experienced in daytime (Figure 3(a)). The most severe attacks (NRS 4.7, SD 1.4) were experienced at 04.00 and the mildest attacks (NRS 3.3, SD 1.4) at 09.00. Autonomic symptoms had a corresponding nightly peak with a maximum of symptoms at 05.00, while there was no obvious difference in duration of attacks occurring at night compared to those in the daytime (Figure 3(a)). Of mild attacks, 90.0% were registered in daytime (06.30–24.00)
Clinical characteristics of attacks. Mild attacks: NRS 1–2; Moderate attacks: NRS 3–4; Severe attacks: NRS 5–6; Very severe attacks: NRS 7–10. The mean number of autonomic symptoms registered/attack is divided in to accountable symptoms, marked by colour blocks. 24-hour rhythmicity of attacks.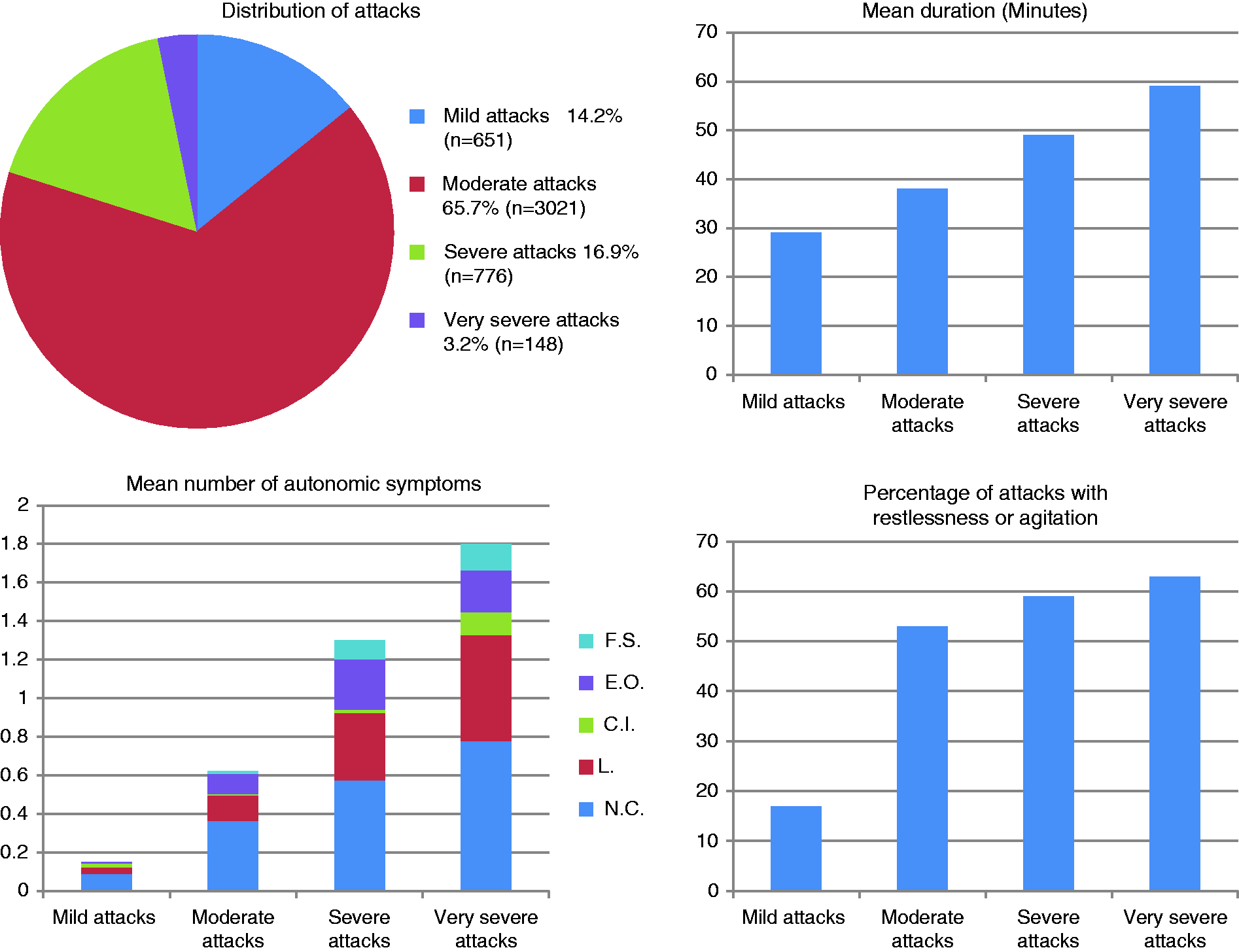
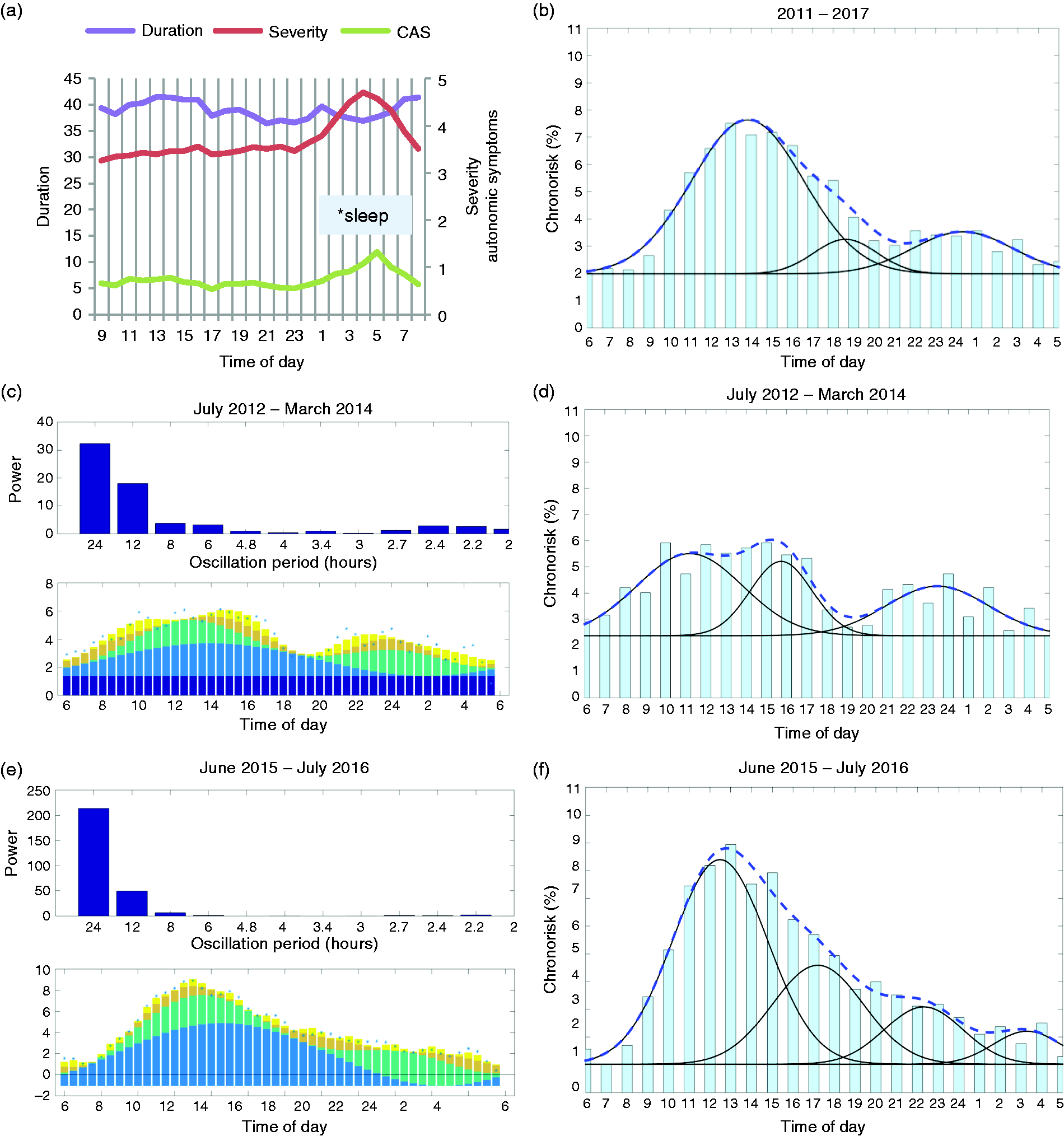
24-hour chronorisk analysis.
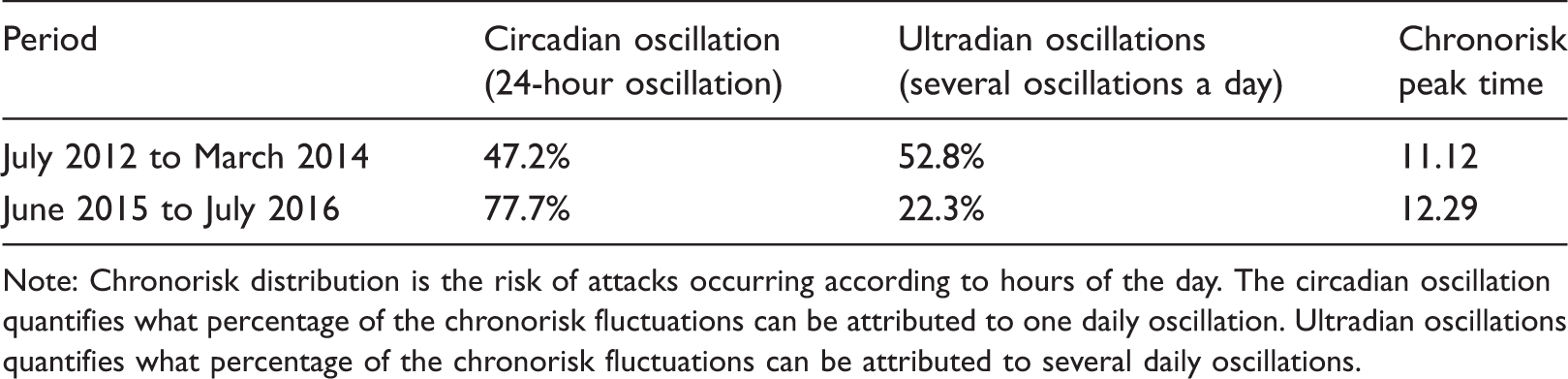
Note: Chronorisk distribution is the risk of attacks occurring according to hours of the day. The circadian oscillation quantifies what percentage of the chronorisk fluctuations can be attributed to one daily oscillation. Ultradian oscillations quantifies what percentage of the chronorisk fluctuations can be attributed to several daily oscillations.
Seasonal rhythmicity
Preventive treatments.
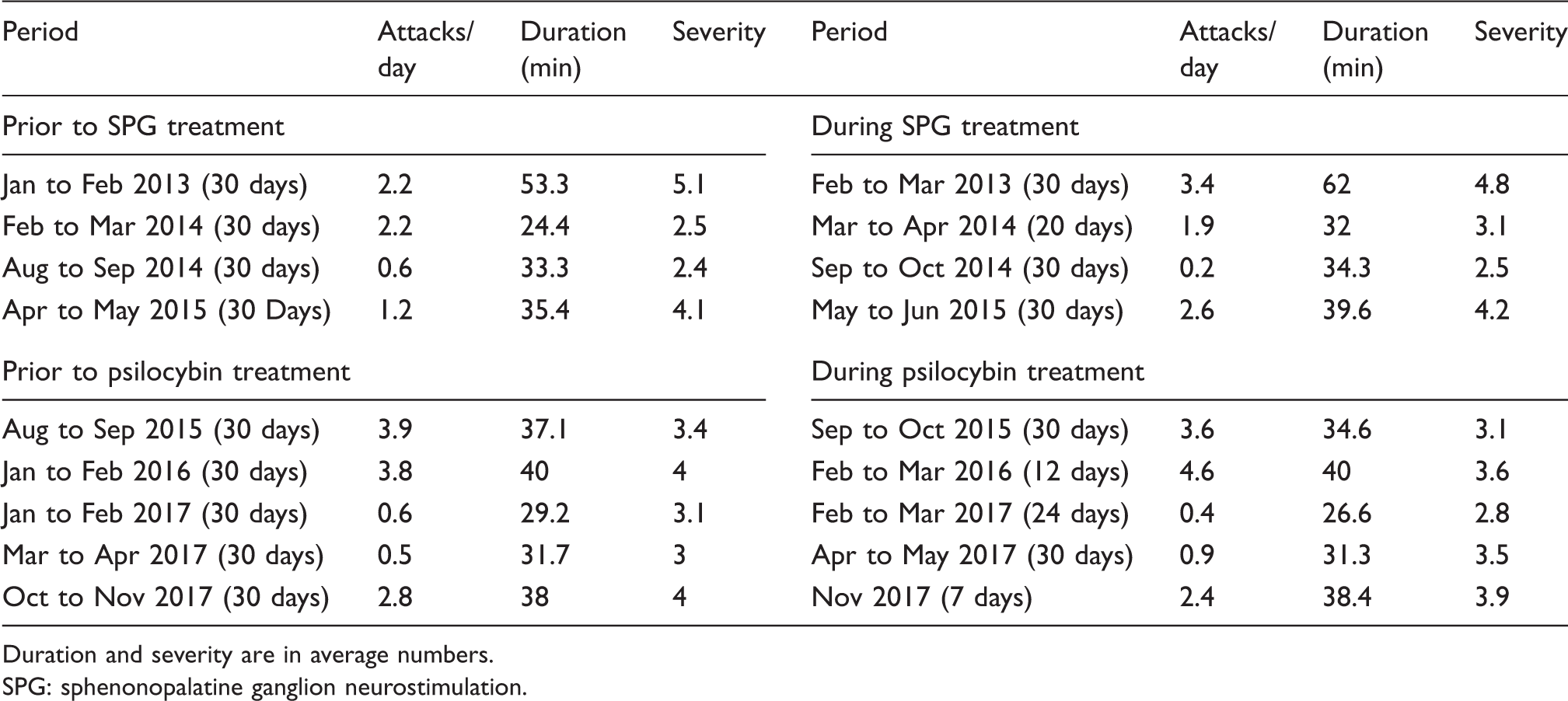
Duration and severity are in average numbers. SPG: sphenonopalatine ganglion neurostimulation.
Mild attack frequency appeared to follow a similar pattern, as did the rest of the attacks. Neither attack severity, duration nor CAS displayed an indication of monthly or seasonal rhythmicity.
Discussion
In this unique, meticulous, and prospective registration of clinical CH data over several years, we found considerable intra-patient variability in attack manifestation. Diurnal rhythmicity changed from predominantly ultradian to circadian between periods of high attack frequency. The majority of attacks occurred during the day and severity increased during night-time.
Clinical characteristics of attacks
A relatively low average pain intensity was recorded, which presumably is because the patient had effective acute treatment and some attacks were mild and self-limiting.
In this case, most of the mild attacks, sometimes accompanied by restlessness and autonomic symptoms, were not treated. Since treatment cannot explain the course of these attacks, they fit what has previously been described as “abortive attacks”, often referred to by patients as “shadows” (16,21). Russel et al. found 16% of non-treated attacks to be “abortive attacks”, while our patient reported 14.2% (16). Ekbom found that 79.9% of CH patients experience “abortive attacks” and that they were often appearing at the beginning, end, or between clusters, which is also our clinical experience (21). However, monthly distribution of mild attacks in our patient showed no such obvious pattern. Although these mild attacks do not fulfill the ICHD-3 for CH attacks, they are undoubtedly a sign of disease activity and prospectively documenting them on a larger scale seems meaningful. Regarding pre-ictal symptoms, mild pain in the attack area was persistently experienced by this patient. A recent prospective study from our group similarly found that 54.6% of attacks were preceded by local pain symptoms in the pre-ictal phase (20). Pre-ictal pain or sensations in the area of the normal attacks have previously been studied and results demonstrate that a variety of symptoms can precede a CH attack (15,18–20). Investigating the positive predictive value of pre-ictal symptoms in relation to CH attacks could lead to differentiated, early treatment strategies.
One could speculate that the recorded simultaneous increase of maximum severity and duration of attacks was due to treatment response, or the lack of it. Early abortive treatment might be the main factor in terminating the buildup of pain and shortening the CH attack. However, most of the attacks that were mild and short were not treated, thus treatment cannot explain the termination of these attacks. Similar results were found in a previous prospective study using a stopwatch to time attacks, suggesting that a positive correlation between maximum severity and duration could be an inherent feature of CH (16). Furthermore, in accordance with prospective data from our group, the number of CAS increased with pain severity (20). This suggests a common driving mechanism in pain and CAS. However, one retrospective study did not establish such a correlation (8). Our data does not contain the timing of related symptoms but earlier studies found that CAS could exist without pain and pain could exist without autonomic symptoms, prompting further investigations on this subject (20,24–29).
24-hour rhythmicity
Attacks were more severe during the night and in accordance with the description of the clinical characteristics of attacks; the number of CAS also displayed a nocturnal increase. Russel et al. did not find any link between non-treated nocturnal attacks and pain severity, and neither did a recent retrospective study from our group in which the majority of patients were on preventive medication (16,18). This discrepancy is possibly explained by the fact that attacks in this study were mainly treated with oxygen and without the use of efficient preventives. One could speculate that initiation of treatment would be earlier when awake and alert than during sleep, causing more severe nightly attacks. Furthermore, fewer mild attacks were registered during nighttime. Sleeping through mild attacks without recording them would increase average nightly severity. Also, sleeping through the initiation of attacks, and thereby failing to accurately time them, could explain why the recorded duration did not increase with nocturnal worsening of severity.
Even though the majority of CH studies on diurnal rhythmicity, including those with cCH patients, report a preponderance of nocturnal attacks (9–11,13,16), some subgroups report an overweight of daytime attacks, as is the case with this patient (10,11,17). One study, including only cCH patients, reported the bulk of attacks to occur mostly during the daytime (17). Furthermore, Barloese et al. found an increased chronorisk during daytime hours and stronger ultradian oscillations in cCH compared to eCH (14). This might suggest a higher degree of desynchronisation from circadian phasors such as sleep. In this case, overall, circadian rhythmicity was dominant. All together, these findings suggest a profound complexity in what gives rise to rhythmicity and the potential regulating factors. Studies from our group have shown a different chronorisk between men and women and that coffee drinking, sleep rhythm and treatment with verapamil affected attack rhythmicity (14,30). Earlier studies have implicated sleep, relaxation and work patterns as modulating factors (9,11,12,16,21). The oscillational pattern in this patient presented a shift from predominantly ultradian to circadian between the two periods of worsened attack frequency. Change in treatment could be a possible explanation. This could, however, simply be a natural development over time. Predicting timing of attacks on the basis of chronorisk could hold the key to personalised treatment, As proposed elsewhere (14), an obvious approach could be to increase the effective doses at times of increased chronorisk to obtain maximum effect and to decrease doses at times of decreased chronorisk to reduce the risk of side effects. Still, fluctuations in chronorisk over time probably should be taken into consideration if applied. A headache diary can be immensely useful for the clinician when evaluating disease activity and the effect of treatment over time. This case demonstrates that smartphone applications could be a feasible tool with the potential of providing such data.
Seasonal rhythmicity
Predicting seasonal variation in CH holds similar potential regarding personalised treatment, as does predicting daily variation. This patient clearly exhibited periods with high and low attack frequency resembling the cluster/remission pattern characterising eCH. In the study by Barloese et al., 36.4% of cCH patients reported annual rhythmicity (9). Other studies including cCH patients also report rhythmic seasonal variation, indicating that cCH patients do have periods of increased attack frequency or severity (8,10,13–15). Clusters have been proposed to follow both equinoxes and solstices and the number of daytime hours has been correlated to the worsening of CH reported by CH patients. In this case, we found no clear rhythm in changes of attack frequency. There was no correlation between monthly sun hours and attack frequency nor changes in medication or major life events that could explain fluctuations. As with circadian rhythmicity, this might reflect desynchronisation in cCH from circannual phasors such as sunlight supported by the fact that fewer cCH patients, compared to eCH patients, report circannual rhythms (9). It is also possible that long-term fluctuations dissociated from the calendar year, as recorded in this case, perhaps follows an unknown and different biological cycle.
Methodological considerations
This is a case report and as such only serves to generate hypotheses. Recordings of timings of attacks were very accurate due to the applied timing, which is a strength of these data. Associated symptoms registered were not pre-specified. This diminishes the danger of overreporting but could also lead to underreporting symptoms. The applied Gaussian and spectral analysis have been shown to be applicable in analysing 24-hour CH rhythmicity (14). It is a limitation to this method that it is a 24-hour analysis, meaning that only oscillations within 24 hours are detected (14).
Conclusion
CH-attacks, in this case, display a high degree of intrapersonal variation. Mild attacks not fulfilling the ICHD-3 criteria for CH attacks are an indicator of CH activity and were frequent in this case. They may constitute a CH disease phenomenon, self-limiting in nature and warranting further investigations, together with the recorded simultaneous increasing pain severity, number of CAS and duration of attacks. Chronorisk analysis is a new tool in the personalised treatment of CH patients; however, applying this, the evolution of chronorisk patterns over time suggests that continuous, cluster-wise, evaluation of rhythmicity may be necessary. We believe that this case shows that digitising headache diary registration by smartphone applications is feasible and provides a great amount of detailed clinical data.
Key findings
Self-limiting mild CH attacks were frequent. CH diurnal rhythmicity changed over time. Pain severity, number of CAS and attack duration increased simultaneously. A cluster/remission pattern in attack frequency was present.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
