Abstract
Background
Cluster headache (CH) is a disabling headache disorder with chronobiological features. The posterior hypothalamus is involved in CH pathophysiology and is a hub for autonomic control. We studied autonomic response to the head-up tilt table test (HUT) including heart rate variability (HRV) in CH patients and compared results to healthy controls.
Methods and materials
Twenty-seven episodic and chronic CH patients and an equal number of age-, sex- and BMI-matched controls were included. We analyzed responses to HUT in the time and frequency domain and by non-linear analysis.
Results
CH patients have normal cardiovascular responses compared to controls but increased blood pressure. In the frequency analysis CH patients had a smaller change in the normalized low- (LF) (2.89 vs. 13.38, p < 0.05) and high-frequency (HF) (–2.86 vs. –13.38, p < 0.05) components as well as the LF/HF ratio (0.81 vs. 2.62, p < 0.05) in response to tilt. In the Poincaré plot, the change in ratio between long- and short-term variation was lower in patients (SD1/SD2, –0.05 vs. –0.17, p < 0.05).
Conclusions
CH patients show decreased autonomic response to HUT compared to healthy controls. This can be interpreted as dysregulation in the posterior hypothalamus and supports a theory of central autonomic mechanisms involvement in CH.
Introduction
Cluster headache (CH) has a significant impact on patients’ lives yet little is understood of the cause of this enigmatic and extremely painful disorder. Different investigative modalities have been employed to study what triggers the characteristic, unilateral, severe pain attacks and accompanying symptoms. Chronobiological features are prominent, which together with endocrinological and radiological findings have focused attention on the hypothalamus (1,2). The hypothalamus is involved in the systemic regulation and integration of a multitude of functions including autonomic control (3). Locally, the cranial autonomic nervous system (ANS) is involved ictally, observable to the surroundings in the form of accompanying symptoms of ipsilateral lacrimation, rhinorrhea and conjunctival injection, among others. It is the manifestation of hyperfunction of the parasympathetic division and hypofunction of the sympathetic division, a result of activation of the trigeminal autonomic reflex (4). The question whether systemic autonomic control is also affected in CH in the attack-free state has been raised before but never fully answered as results from previous clinical studies are somewhat diverging (5–16). However, most support a theory of central autonomic dysregulation in CH. Various alterations in the electrocardiogram (ECG) and (relative) bradycardia in relation to provoked and spontaneous attacks have been reported previously (7,14–17). On the contrary, a recent study found normal responses and no difference in autonomic function when comparing the same 18 patients in- and outside of bout at rest, during deep breathing, after standing up and during a Valsalva maneuver (6).
Heart rate variability (HRV) measures are derived from analysis of variation in time between heartbeats (RR intervals). The analysis can be performed in the time and frequency domain as well as by non-linear methods. It is a relatively novel way to characterize autonomic function and only a few studies of spectral analysis in CH patients exist (5,8). Our aim was to characterize overall autonomic function, as measured in a head-up tilt table test (HUT) by means of spectral analysis, in a population of CH patients during the active bout of their headache disorder and compare this to matched, healthy controls. Our working hypothesis was that autonomic dysregulation is present in CH patients and is either directly involved in the pathological mechanisms leading to attacks or is a consequence of central pathology—an epiphenomenon.
Methods
The study was approved by the ethics committee of the Capital Region of Denmark (H-2-2012-016) and all patients and controls gave their written consent. All investigations were conducted at Glostrup Hospital, Dept. of Diagnostics and at the Coordinating Research Centre, Frederiksberg Hospital.
RR-intervals and blood pressure (BP) were measured continuously from a bipolar two-channel ECG and by Finometer equipment (Finapres Medical Systems BV, Amsterdam, the Netherlands), respectively. The Finometer measures continuous BP non-invasively using infrared photoplethysmography through a finger-cuff. The ECG was band-pass filtered with a cut-off frequency of 0.6 and 40.0 Hz, and the BP data were low-pass filtered with a cut-off frequency of 40.0 Hz. RR intervals were converted to instantaneous heart rate (HR) and systolic blood pressure (SBP) was derived from the maximum value on the continuous BP recording on a beat-by-beat basis. Data were sampled at 1.0 kHz and analyzed using commercial software (LabChart, AD Instruments Inc, Colorado Springs, CO, USA, and Taskforce, CNSystems Medizintechnik AG, Graz, Austria).
Patients
Clinical characteristics of patients and controls.

Data are expressed as means (range). BMI: body mass index; CH: cluster headache; ECH: episodic CH; CCH: chronic CH; M: male; F: female. ap < 0.05.
Controls
Twenty-seven controls matched according to age, sex, body mass index (BMI) were included (Table 1). All controls were interviewed to ensure they were healthy and did not suffer from disorders including primary headaches. Controls were recruited via mail from the Copenhagen City Heart Study between May and December 2011 (20).
Recordings
All tests were performed in the fasting state between 8 a.m. and 2 p.m. at standard room temperature. After 10 minutes of supine rest, baseline data were acquired during a further 10 minutes of rest, during which the participants refrained from speaking and from unnecessary movements. The participants were then loosely strapped to an electrically driven tilt table and tilted to a 60-degree head-up position (HUT) within 20 seconds and stayed in this position for at least 10 minutes if intolerable symptoms did not appear.
Data analysis
Baseline values of HR, SBP and diastolic blood pressure (DBP) were calculated as mean values from the 30 seconds preceding tilt in the supine position and during 30 seconds obtained in the fifth and 10th minute of the HUT.
Analysis of HRV was performed according to current guidelines (21) using shareware (Kubios, version 2.0, http://kubios.uku.fi). Artifact- and ectopic-free five-minute segments of RR intervals from supine and tilted rest were analyzed (Figure 1). In the time domain: mean value of successive normal beats (meanNN), standard deviation of successive normal beats (SDNN) and the numerical difference between successive normal beats (root mean successive squared difference (RMSSD)) were calculated. The power spectral densities were calculated using the autoregressive method (model order 16) and expressed as total power as well as the power in the low-frequency (LF: 0.04–0.15 Hz) and high-frequency (HF: 0.15–0.40 Hz) bands and expressed both as absolute power and in normalized units (LF n.u., HF n.u.) as well as by the LF/HF ratio (LF/HF). Pack-years were calculated as the number of packs of cigarettes per day multiplied by the number of smoking years.
Example of series of R R intervals ((a) and (d)) and the corresponding spectral ((b) and (e)) and nonlinear analysis ((c) and (f)—Poincaré plot) in the supine ((a)–(c)) and tilted ((d)–(f)) positions. In response to tilt, power in the 0.15–0.4 Hz range is withdrawn, indicating a shift away from parasympathetic dominance. Likewise, in the Poincaré plots, in response to tilt, activity narrows around the SD2 axis. RR: heartbeat intervals.
Statistical analysis
SAS 9.4 was used for all statistical analyses. Wilcoxon rank sums test was used for non-parametric data and standard two-sample t-test for parametric. P values below 0.05 were considered significant. HR and BP and changes in the time domain were analyzed as changes in percentage to account for baseline values. Chi-squared test was used to compare smoker and sex-composition in groups.
Results
Twenty-seven patients and 27 controls were included in the final analysis. Data after tilt were not available for one control and BP measurements were not available for another because of technical malfunctions. There were no differences between patients and controls in age (p = 0.27), sex (p = 1) or BMI (p = 0.14). Patients had a higher percentage of smokers (p = 0.0293) but number of pack-years did not differ significantly although mean was higher in patients (19.5 vs. 12.3, p = 0.12).
HR and BP
Changes in percent in HR, SBP and DBP from supine to 4.5 and 9.5 minutes after tilt.

Data are expressed as means (SD). ECH: episodic cluster headache; CCH: chronic cluster headache; HR: heart rate; SBP: systolic blood pressure; DBP: diastolic blood pressure; n.s.: not significant.
Time domain
Changes in percent in mean normalized RR-intervals, standard deviation and numerical difference between normal beats after HUT compared to supine.
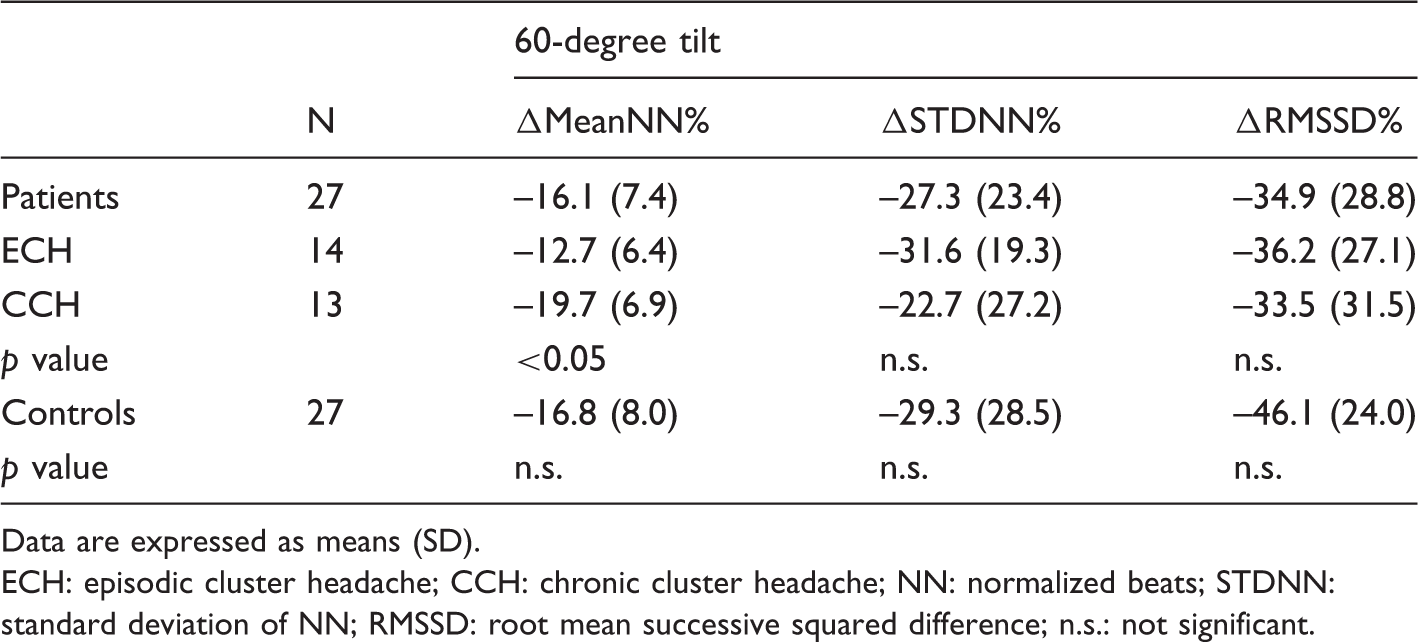
Data are expressed as means (SD). ECH: episodic cluster headache; CCH: chronic cluster headache; NN: normalized beats; STDNN: standard deviation of NN; RMSSD: root mean successive squared difference; n.s.: not significant.
Frequency domain and non-linear analysis
Non-linear analysis of the response to tilt.
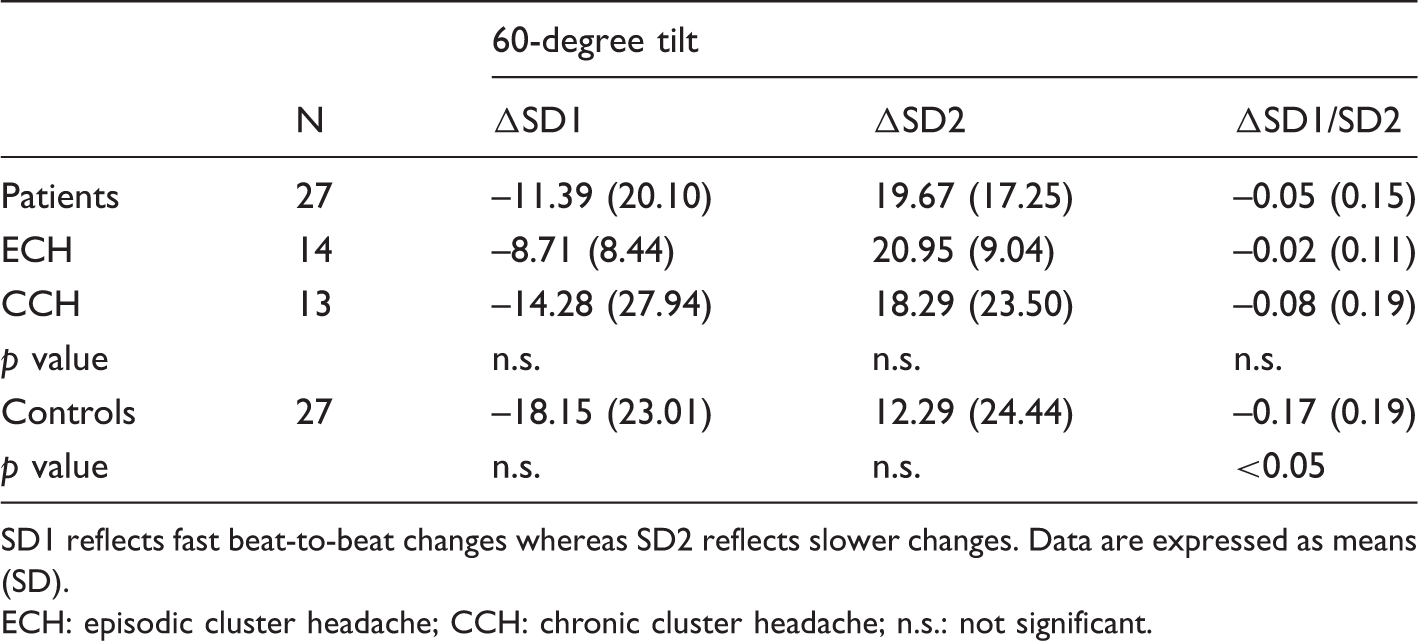
SD1 reflects fast beat-to-beat changes whereas SD2 reflects slower changes. Data are expressed as means (SD). ECH: episodic cluster headache; CCH: chronic cluster headache; n.s.: not significant.
Spectrum analysis of response to tilt. Absolute changes in power spectra including normalized units and ratio between LF and HF.

Data are expressed as means (SD). LF: low frequency; HF: high frequency; n.s.: not significant.
Effect of smoking
In our sample there were no differences in resting HR (p = 0.8590) or BP (SBP: p = 0.6229, DBP: p = 0.7888) between smoking and non-smoking patients. In controls, DBP was lower in smokers (49 vs. 60 mmHg, p = 0.0219) whereas there was no difference in HR (p = 0.1625) or SBP (p = 0.0859).
Effect of medication
Eleven patients were taking verapamil, an L-type calcium-channel blocker, as cluster headache prophylaxis. HR (p = 0.2267), SBP (p = 0.1088) and DBP (p = 0.1088) did not differ and there were no differences in ΔAbs. power LF (p = 0.2039), ΔAbs. power HF (p = 0.4160), ΔLF n.u. (p = 0.6634), ΔHF n.u. (p = 0.6595) or ΔLF/HF (p = 0.2555) between those using prophylactic treatment and those not. HR, SBP and DBP in the patients who had used triptans (n = 4) compared to those who had not (n = 23) did not differ (respectively: 66 vs. 70 (p = 0.9728), 117 vs. 122 (p = 0.2323), 74 vs. 79 (p = 0.2901)).
Other clinical features
No differences were found in a subgroup analysis stratifying patients according to annual or diurnal rhythmicity, time since last attack, headache severity or whether the patients suffered primarily sleep or wake attacks.
Discussion
In a population of CCH and ECH patients in bout, we found changes in the non-linear and frequency domain in an HRV analysis of RR intervals during HUT in the attack-free state. The main finding was a significantly lower increase in the LF power and correspondingly a lower decrease in HF power in patients compared to controls in response to orthostatic challenge. This result was to a certain extent mirrored in the non-linear analysis. The results may be interpreted as a blunted autonomic response to the transition from the supine to standing position in patients during active bout. Comparing the two groups of CH, we also found a larger response in change in HR in CCH patients compared to ECH in the first five minutes of tilt after adjusting for baseline.
It must be stressed that the changes observed in the present study are not indicative of clinically relevant autonomic dysfunction and none of the patients presented symptoms of autonomic dysfunction or neurodegenerative disorders. They may, however, be related to the processes underlying the severe headache attacks. That major dysfunction in cardiovascular control should be found was not part of the hypothesis tested, as CH patients in general have no complaints of autonomic dysregulation besides the prominent ictal facial symptoms. Thus, three scenarios can be suggested: 1. The findings reflect dysfunction of the autonomic nervous system which may be involved in triggering CH attacks. 2. As with other phenomena observed in CH, the findings are epiphenomena and the results of dysregulation on a more complex level. 3. CH pathology is seated somewhere else entirely, and the observed changes are the result of other, non-identified differences between patients and controls.
Normal HR and BP responses to HUT are mediated by sympathetic activation through baroreceptor reflexes and a central command involving the posterior hypothalamus. It is hypothesized that the (posterior) hypothalamus (dorsomedial and paraventricular nucleus) acts as a central hub regulating autonomic response (22). The observed inability to decrease HF and increase LF activity during HUT would typically be interpreted as a blunting of baroreceptor activity. This is mirrored in the non-linear analysis where SD1 represents the fast beat-to-beat variation mediated through vagal stimulation and SD2 reflects slower changes predominantly mediated by the sympathetic nervous system in the baroreflex arc.
In CH, a number of older studies have investigated cardiovascular responses to various tests and during attacks. In 61% of attacks occurring from wake mean HR decreased, whereas in 67.5% of attacks beginning from sleep, no change or an increase in HR was observed (15). Further, Russell and Storstein found that the increase in HR at the onset of attacks was dependent on the HR before the attack (14). They theorized that a low level of sympathetic activity beforehand provides greater possibility for a larger relative increase in sympathetic activity or similarly a greater decrease in parasympathetic tone. In a subsequent analysis they found an increase of HR at the onset of attacks, a relative decrease during the attack itself and again a relative increase at the end of attacks (14). In one patient subjected to Holter monitoring, Attanasio et al. found bradycardia along with phases of junctional rhythm beginning roughly five minutes prior to the attack and lasting almost throughout (11). Increased sinus arrhythmia during deep breathing but normal response to the Valsalva maneuver have also been described (13). Further, higher ictal HRV has been described in a single case along with a blunted HR response to tilt (10).
In these findings, increased sinus arrhythmia points toward a more active parasympathetic system, which is substantiated by, firstly, the case report of bradycardia and junctional rhythm reported by Attanasio (11), secondly, the bradycardia and sinoatrial block reverted by atropine in one patient (14) and thirdly, the attack-related atrial fibrillation in another patient (14). The changes in HR at the onset and at the end of an attack are compatible with on-off transients in parasympathetic tone, i.e. a fast withdrawal with a subsequent normalization causing temporary increments in HR followed by a leveling off to pre-attack values. The increase in standard deviation of HR found by Russell and Storstein during attacks expresses an increase in total HRV due to either increased parasympathetic activity or increased patient activity. The more pronounced changes in HR in those with attacks during sleep are compatible with the generally higher parasympathetic activity during this stage and are reflected in the lower HR prior to attacks.
In a spectral analysis of the response to HUT, De Marinis et al. found that three of their eight patients did present an increased HF component during rest and tilt; however, overall, there was no difference between CH patients and controls (8). Cortelli et al. studied responses to various autonomic tests in eight CCH patients implanted with a deep brain stimulator (5). Analyzing HRV, they found increased LF/HF in the HUT after initiation of deep brain stimulation (DBS) interpreted as an enhanced sympathoexcitatory drive. It seems that DBS, in producing a state of CH remission, also changes the autonomic response in the HUT (but not in other autonomic tests). These findings are in line with those in the present study as we found a lower LF/HF ratio in CH, and older studies have shown that stimulation of the posterior hypothalamus produces increased sympathetic drive whereas lesioning of this area decreases sympathetic activity or increases parasympathetic activity (23,24). This could explain the results obtained by Cortelli et al., and, in the setting of a theory of dysregulation of the posterior hypothalamus in CH, it could also explain ours.
In CH there is clinical evidence that ANS dysfunction plays a role in the pathogenesis, in part due to reports of the timing of attacks. Russell showed that daytime attacks primarily occur when patients are physically relaxed (relatively high parasympathetic tone) (25). It can be theorized that, in susceptible individuals, the normal fluctuations of sympathetic and parasympathetic activity may trigger a dysregulation of nociception perhaps resulting in disinhibition of descending antinociceptive input to the trigeminal complex, in turn resulting in a free-running trigeminal-autonomic reflex. In light of this, it seems that the possibility that the present results are simply epiphenomena is low.
Strengths and limitations
The present study is strengthened by the fact that it included a large number of well-characterized patients in the active period of the disorder and compared these to age-, sex- and BMI-matched controls. However, complete matching for smoking was not achieved as our CH patients (and CH patients in general) were heavy smokers to a degree that proved very difficult to account for. Several lifestyle factors are known to influence cardiovascular health and baroreflex sensitivity (BRS). Smoking increases sympathetic activity (26) and studies have demonstrated that—in comparison to nonsmokers—smokers have higher HR, diminished HRV, and reduced cardiovagal BRS (27) but similar or even lower BP (28). We did, in fact, observe lower DBP in smoking controls, a known phenomenon (29). Even though we succeeded in matching our patients so that there were no statistical difference in pack-years, it is still possible that the changes we observed may be attributable to differences in lifestyles, perhaps also in smoking habits.
Some of our patients were on headache medication including verapamil and triptans. A post hoc analysis showed that this did not produce significant differences compared to those patients who were off medication in analyzed parameters but we cannot exclude that it did alter the results. Previous studies in patients with cardiovascular disease have shown a shift toward vagal dominance or sympathetic attenuation during treatment with verapamil (30–34). In migraine, one study showed that verapamil reduced an exaggerated sympathetic response (35). Such a shift toward vagal dominance induced by verapamil could partially explain the present findings; however, since there was no difference in response to tilt between patients taking verapamil and those not, it seems an unlikely scenario. Further, the use of triptans did not affect BP in the patient group. Of course ideally the patients would have been free of medication; however, investigating CH produces practical and ethical dilemmas as tapering of medical prophylaxis and/or triptans produces severe worsening of the pain disorder.
Conclusion
In conclusion, we have demonstrated that CH patients exhibited a blunted shift toward sympathodominant activity in the HUT, suggesting a decreased sympathetic drive or a failure to withdraw parasympathetic tone. The results support hypothalamic involvement and the theory of central dysregulation in CH. Future endeavors in this area should focus on possible changes outside the cluster period, since even though there is no manifest headache, ongoing pathological processes may still be present (36).
Clinical implications
Cluster headache (CH) patients have normal cardiovascular responses to change from the supine to upright position. In a heart rate variability analysis, CH patients exhibited a blunted sympathetic response indicative of a decreased sympathetic drive or continued parasympathetic dominance at that head-up tilt table test. The results support a theory of central dysregulation in CH.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
None declared.
Referees
Pietro Cortelli, Department of Neurological Sciences, University of Bologna, Bologna, Italy, pietro.cortelli@unibo.it Alessandro Silvani, Department of Neurological Sciences, University of Bologna, Bologna, Italy, alessandro.silvani3@unibo.it Tim Jürgens, University Medical Center Hamburg – Eppendorf, Hamburg, Germany, t.juergens@uke.de A. Glaros, Division of Basic Medical Sciences, Kansas City University of Medicine and Biosciences, 1750 Independence Ave., Kansas City, MO 64106, USA, aglaros@kcumb.edu J.A. van Vliet, Department of Neurology and Clinical Neurophysiology, Leiden University Medical Centre, PO Box 9600, 2300 RC Leiden, the Netherlands, javanvliet@lumc.nl
