Abstract
Six patients with alternating hemiplegia of childhood (AHC) underwent motor evoked potentials (MEPs), somatosensory evoked potentials (SEPs) and blink reflex recording. No SEP abnormality was found. As for MEP recording, central conduction time did not differ between patients studied during either interictal or ictal phase, and normal subjects, suggesting that the pyramidal system function is spared. In blink reflex recording, the latencies of both the ipsilateral (iR2) and contralateral (cR2) R2 components were significantly longer (P < 0.01) in patients during the interictal phase than in normal subjects. Moreover, the iR2 and cR2 areas were significantly reduced (P < 0.01) in patients during the interictal phase, compared with normal subjects. During the ictal phase, the ipsilateral R2 latency was significantly decreased after stimulation of the hemiplegic side, compared with the interictal phase (P < 0.05). The blink reflex abnormalities suggest a brainstem dysfunction, which may be linked to the pathophysiological mechanisms of the disease.
Introduction
Alternating hemiplegia of childhood (AHC) is a rare disorder characterized by transient recurrent episodes of hemiplegia involving alternatively either side of the body associated with tonic or dystonic attacks, abnormal ocular movement and autonomic abnormalities. The onset of AHC is usually before the age of 18 months. The clinical course is apparently progressive and patients develop fixed neurological deficits as choreoathetosis, dystonia, ataxia and neurodevelopmental disturbances. A few patients show signs of pyramidal tract involvement. Epileptic seizures are reported in 19–50% of patients (1–4). Neuroimaging studies are normal in a variety of patients and autoptic examination has not shown pathognomonic lesions of the brain (3, 5–7). A few family cases have been reported (8, 9). The pathophysiology of this disorder is unknown, but an identifiable molecular basis, perhaps at the level of an ion channel defect, can be assumed (7). The International Headache Society considered AHC in a recent classification of headaches (10). Although no gene mutations of familial hemiplegic migraine (FHM) were found in AHC patients (11, 12), a novel mutation in a kindred with features that bridge the phenotypic spectrum between AHC and FHM syndromes has been found (13, 14).
AHC has rarely been investigated using neurophysiological techniques, which may be useful in localizing the lesion site in the brain, thus making the pathophysiological mechanisms of the disease more clear. Moreover, these studies were often in disagreement. The somatosensory evoked potential (SEP) generated in the primary somatosensory area (N20) was found absent during the ictal phase, but it recovered normal amplitude and latency after diazepam injection (15). In one patient, the N20 SEP component showed low amplitude during the interictal phase, but it recovered after flunarizine treatment (16). Santanelli et al. did not find any modification of the brainstem auditory evoked potentials (BAEPs) in one child between the ictal and interictal phases (17). Di Capua et al. found abnormalities of both the ipsi- and contralateral R2 components of the blink reflex in two patients during the interictal phase, while SEPs, visual evoked potentials, motor evoked potentials (MEPs) and BAEPs were within normal limits during both the ictal and interictal phases (18).
The aim of our study was to investigate possible neurophysiological abnormalities at both the cerebral cortex and the subcortical structure levels in six AHC patients during the interictal phase and the attack. Therefore, we recorded SEPs and MEPs to assess the function of the somatosensory and motor pathways. Moreover, the blink reflex recording was addressed to identify dysfunctions at the brainstem level.
Patients and methods
Subjects
Six patients (three females and three males, mean age 16.17 years, range 5–31 years) suffering from AHC and five healthy subjects (four females and one male, mean age 17.2 years, range 11–30 years) were studied. All patients had normal brain magnetic resonance imaging (MRI). Table 1 summarizes the interictal clinical features of patients and the drugs they assumed. All patients during the interictal phase and all normal subjects underwent MEP, SEP and blink reflex recording. In five patients, MEPs, SEPs and blink reflex were also recorded during the ictal phase. Neurophysiological results were statistically compared between five interictal patients and five normal subjects, while one 5-year-old patient was not considered for the statistical analysis since the myelination process is not complete before the age of approximately 10 years (19–21). In the ictal phase, two patients showed left hemiplegia, three patients right hemiplegia and three patients showed unilateral nystagmus. The hemiplegia never affected the facial muscles and lasted from several hours to several days. It was not associated with modification of tendinous reflexes or with Babinski sign. During the attack, the muscle tone passed from an initial hypotonia to a later hypertonia. The neurophysiological tests were performed while the patient was hypotonic. One patient (patient 2 in Table 1) had headache before and during the attack and another patient (patient 6 in Table 1) rarely had headache before or during the ictal phase.
Clinical features of patients

Blink reflex and evoked potential recording technique
During all the recording sessions, subjects were supine and relaxed in a quiet room. All neurophysiological examinations were performed using a Medelec Synergy system (Oxford Instruments, Eynsham, UK).
The blink reflex was evoked by electrical stimulation of the supraorbital nerve. The stimulus intensity (12.2 ± 0.8 mA) was sufficient to produce a relatively constant response with repeated stimulations. In no case was the stimulus perceived as painful. To avoid habituation, shocks were delivered arrhythmically at time intervals of 30 s. Active surface Ag/AgCl electrodes were placed over the left and right mid-lower half of the orbicularis oculi muscle. Reference electrodes were placed over both the lateral edges of the orbita. The EMG signal was amplified and filtered (bandpass 50–5000 Hz). Ipsilateral (R1 and iR2) and contralateral (cR2) responses were recorded. Five responses were recorded for both the left and right supraorbital nerve stimulation and the minimal R1 and R2 latencies were measured (19, 20). Then, the five sweeps were rectified and averaged. In the average traces, the areas of both the iR2 and cR2 components were calculated.
For MEP recording, the motor cortex was stimulated by a magnetoelectric stimulator (Cadwell MES10) producing a maximum magnetic field of 2 T, pulsed for 100 μs, with a bipolar pulse shape. Cortical stimulation was performed with a circular coil (diameter 9.5 cm) positioned over Fz for the lower limb MEPs and over Cz for the upper limb MEPs. Radicular stimulation was performed with the coil positioned over C7 for the upper limbs and over L5 for the lower limbs. The MEPs were recorded by surface electrodes on the abductor pollicis brevis and tibialis anterior muscles bilaterally, referred to the electrodes on the corresponding distal tendon. Cortical MEPs were recorded at rest and following voluntary muscle contraction (10% of maximal muscle contraction), while radicular MEPs were obtained at rest. The electromyographic (EMG) filter bandpass was between 2 Hz and 5 kHz. Motor threshold was measured at rest for upper and lower limbs in all subjects as the lowest stimulator intensity evoking clearly discernible and reproducible muscle responses. Stimulus intensity for MEP recording was 120–150% above threshold. The onset latency of the negative potentials and the peak-to-peak amplitude were measured; the motor central conduction time (CCT) was calculated for upper and lower limbs by subtracting the latency of the cervical and lumbar MEPs from the latency of the cortical MEPs, respectively (21, 22).
For SEP recording, both the right and left median nerves were stimulated in all subjects. Monophasic square wave electrical pulses of 200 μs were delivered at a rate of 3 Hz through surface Ag/AgCl electrodes over the median nerve at the wrist. Stimulus intensity was set slightly above motor threshold, eliciting a visible and stable twitching of the thenar eminence muscles. Active surface recording electrodes were placed at Erb's point ipsilateral to stimulation (referred to contralateral Erb's), over the spinous process of the sixth cervical vertebra (Cv6) (referred to an electrode located immediately above the thyroid cartilage), in the scalp parietal region contralateral to stimulation (P3/P4) and at Fz (referred to the earlobe ipsilateral to the stimulation). Electrode impedance was less than 5 hΩ. Analysis time was 100 ms, sampling rate 20 kHz and filtering bandpass 5–1500 Hz. Responses to 800 stimuli were averaged, two repetitions were made for the left and right median nerve stimulation. Peak latency and peak-to-peak amplitude were measured for the peripheral N9 potential, for the cervical N13 response, and for the parietal N20 and the frontal N30 waves (23, 24).
Statistical analysis
The Shapiro–Wilks test was used to ensure the Gaussian distribution of the data. Unpaired t-test was used to compare the neurophsyiological values between patients and normal subjects. In the AHC patients, paired t-test was used to compare the neurophysiological values between the ictal and interictal phases. The significance level was fixed at P < 0.05.
Results
Blink reflex recording
Table 2 shows the blink reflex data in five normal subjects and five interictal patients. We calculated the normal latency limits for the R1, iR2 and cR2 responses as the mean values ± 2.5 SD. The upper normal limit was 11.34 ms for the R1 component, 30.35 ms and 32.74 ms for the iR2 and cR2 responses, respectively. While the R1 latency in patients was within normal limits, the iR2 latency was abnormally prolonged in four patients after right supraorbital nerve stimulation and in three patients after left supraorbital nerve stimulation, and the cR2 latency was abnormally prolonged in four patients to right supraorbital nerve stimulation and in three patients to left supraorbital nerve stimulation. Our upper normal limits for R2 latencies were lower than the ones reported in literature, perhaps due to the limited number of our normal subjects. However, the abnormal R2 latency values in our patients overtook even the commonly used upper normal R2 latency limits (25). R2 latencies were significantly longer in the interictal patients than in normal subjects (P < 0.01). The iR2 latency after stimulation of the supraorbital nerve ipsilateral to the hemiplegia was significantly earlier during the ictal phase than during the interictal phase (P < 0.05). Moreover, during the attack the iR2 latency to stimulation of the hemiplegic side became significantly shorter than the one to contralateral stimulation (P < 0.01) (Figs 1 and 2). The iR2 and cR2 areas were significantly larger in normal subjects than in patients during both the ictal or interictal phases (P < 0.01) (Figs 1 and 2), while in patients no difference was found between the interictal phase and the attack (P > 0.05).

Blink reflex in a healthy subject and in a patient during the interictal phase. Notice the delayed R2 latencies in the patient.

Blink reflex after left side stimulation in a patient during the interictal and the ictal phases (left hemiplegia). Notice the reduction of the iR2 latency in the ictal phase.
Interictal blink reflex

Motor evoked potentials
At rest, MEPs were recorded in all normal subjects, while no response was obtained in two patients from lower limbs. Table 3 shows the MEP data in five interictal patients and five normal subjects. Motor threshold was significantly higher in patients than in healthy subjects for both upper and lower limb motor area stimulation (P < 0.05) (Fig. 3). MEP amplitude measured during active muscle contraction tended to be lower in patients than in normal subjects, although the difference was not statistically significant (upper limbs P = 0.15, lower limbs P = 0.19). Central motor conduction time was not different between patients and normal subjects (upper limbs P = 0.31; lower limbs P = 0.89). Lastly, central motor conduction time obtained during the interictal phase did not significantly change during the attack after recording from both the hemiplegic and non-hemiplegic sides (upper limbs P = 0.25; lower limbs P = 0.81).

Histogram comparing the mean motor threshold for upper and lower limb motor evoked potential recording between patients (▪) and healthy subjects (□). Vertical lines show the standard deviations. ∗P < 0.05. Notice the higher motor threshold in patients than in normal subjects.
Interictal motor evoked potential recording during active muscle contraction in alternating hemiplegia of childhood patients and normal subjects
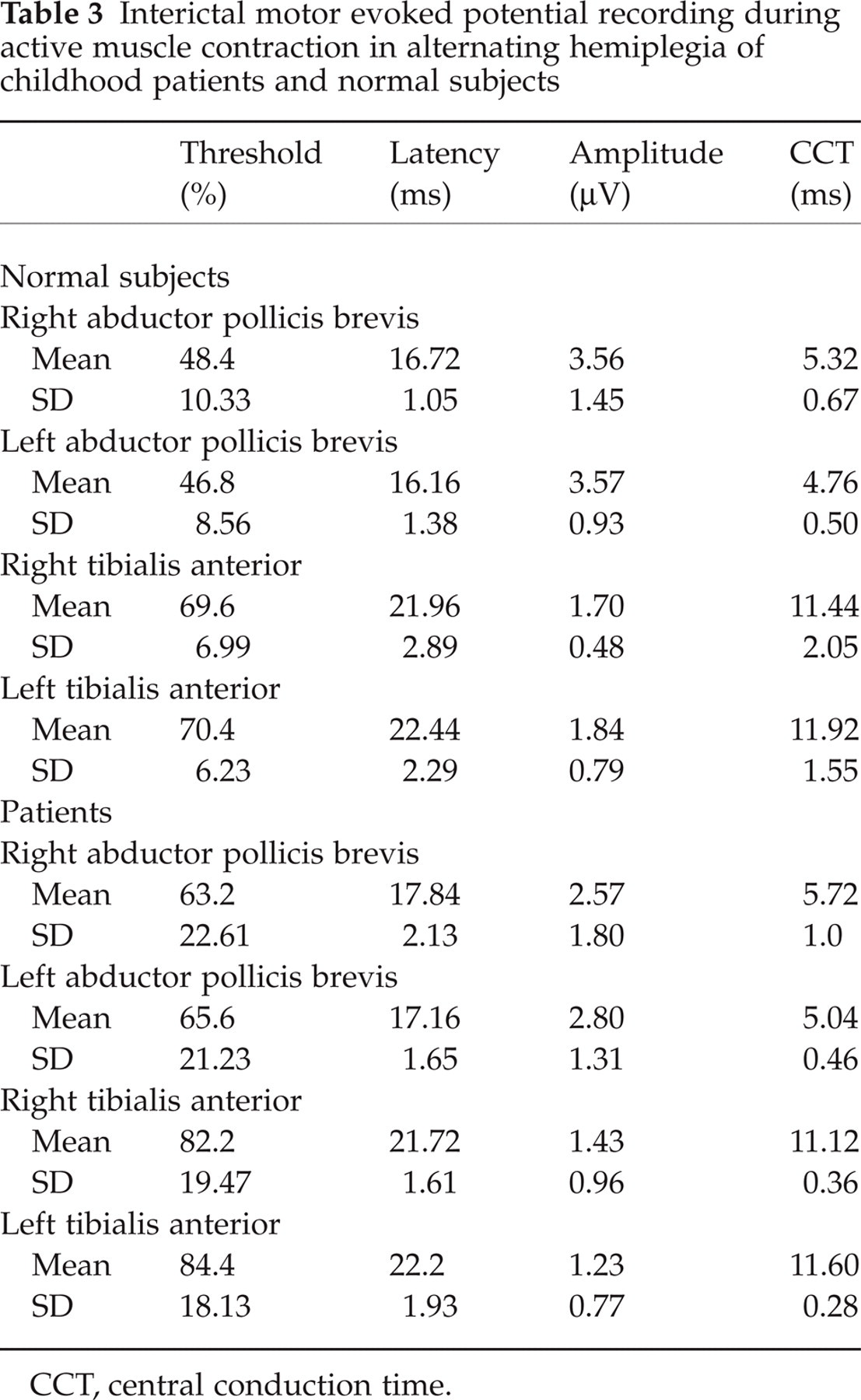
CCT, central conduction time.
Somatosensory evoked potentials
Table 4 shows the data obtained from patients in the interictal phase and from normal subjects. N9, N13, N20 and N30 latencies and amplitudes were not significantly different between patients and normal subjects (P > 0.05). Moreover, SEPs recorded during the interictal phase did not show any significant modification during the attack (P > 0.05).
Interictal somatosensory evoked potentials in alternating hemiplegia of childhood patients and normal subjects

Discussion
Our AHC patients showed two main neurophysiological features: (i) impaired brainstem circuits as suggested by the blink reflex abnormalities, and (ii) normal function of the motor pathways subserving both the upper and lower limb muscles.
In our patients during the interictal phase, the blink reflex iR2 and cR2 latencies were significantly longer and the iR2 and cR2 areas were significantly decreased, compared with normal subjects. In patients, the iR2 latency after stimulation of the supraorbital nerve ipsilateral to hemiplegia was shortened during the attack, compared with the interictal phase. It is difficult to explain the blink reflex abnormalities in patients who did not show any evident brainstem lesion in MRI. However, an abnormal R2 response may be due not only to lesions directly affecting the reflex pathways, but also to dysfunctions out of brainstem which indirectly influence the excitability of the R2 polysynaptic connections. Bilaterally abnormal R2 responses with normal R1 component are rarely found in lesions affecting the brainstem (25). On the other hand, bilaterally prolonged R2 latency has been described in Huntington's disease (26, 27), in cerebral palsy due to perinatal asphyxia (28), in ischaemic-hypoxic brain damage during the neonatal period (29), in pseudobulbar palsy and in cerebral stroke (25). In Parkinson's disease the R2 latency reduction suggests an increase of interneuronal excitability compared with healthy subjects (30). There are two main hypotheses to explain our findings. First, the prolonged R2 latency during the interictal phase might be due to a basal ganglia dysfunction, the existence of which is suggested in AHC patients by the presence of chorea. Although no known direct projections from the basal ganglia to the lateral reticular formation at caudal brainstem level has ever been demonstrated, a primary basal ganglia dysfunction may secondarily perturb corticoreticular or rubroreticular projections (31). Indeed, the basal ganglia output structures can regulate the excitability of brainstem reflex blink circuits (32, 33). However, this hypothesis cannot account for the R2 latency reduction during the attack. Second, as in Huntington's disease and in hemiplegia (31), also in AHC the R2 latency delay can be due to the loss of a tonic facilitation from the postcentral cortical region onto the brainstem interneuronal pool. From this point of view, a unilateral abrupt recovery of this facilitatory influence might lead to both the appearance of the ictal neurological symptoms and the R2 latency reduction after stimulation of the hemiplegic side. This mechanism is similar to the one possibly operating in migraine, in which the painful attack is thought to represent a reset after the abnormality of the cerebral cortex excitability has reached a critical level (34). Although migraineurs during the interictal phase do not show any abnormality of either R2 latency or area (35, 36), Kaube et al. (37) showed a shortened latency of the R2 component to painful stimulation during the migraine attack.
Since it was shown that antiepileptic drugs used in polytheraphy increase the R2 latency (38), our findings might be due to patients' pharmacological treatment (see Table 1). However, even one patient who did not use any antiepileptic drug and another patient who used just phenobarbital showed a prolonged R2 latency during the interictal phase, thus suggesting that the R2 latency delay could not be attributed to the pharmacological treatment.
In our patients, during the interictal phase, the MEP threshold was increased with respect to healthy subjects. However, three out of five patients used antiepileptic drugs at the moment of the test. Since antiepileptic drugs increase motor threshold in normal subjects and in patients with epilepsy (39), our findings may be due merely to antiepileptic drug use. Indeed, the motor threshold was particularly high in two patients who used antiepileptic drugs in polytherapy (patients 3 and 4 in Table 1). Since MEP abnormalities were found to be related to pyramidal tract impairment in several diseases, such as stroke, multiple sclerosis, amyotrophic lateral sclerosis and cervical spondylotic myelopathy (40–43), normal MEPs in our patients suggest that the pyramidal tract function is spared during both the interictal phase and the attack. This is in agreement with the clinical findings, which showed neurological abnormalities such as choreoathetosis and dystonic postures, but no pyramidal signs.
SEPs recorded in all of our patients during both the interictal and ictal phases did not differ from those recorded in healthy subjects, thus confirming that the somatosensory pathways are not damaged in AHC (18). On the contrary, isolated cases of abnormal SEPs have been reported in one AHC patient during an epileptic episode (15) and in another patient during the interictal phase (16).
In conclusion, the normal motor central conduction time in AHC patients indicates that the pyramidal system is not impaired in this disease, thus the dysfunction site must be sought in the pyramidal system. The abnormal blink reflex during the interictal phase may be related to a brainstem dysfunction affecting the polysynaptic pathways mediating the R2 response. The R2 latency reduction during the attack suggests that the brainstem abnormality is due to excitability changes of suprasegmental circuits, possibly within the basal ganglia or the cerebral cortex. While we must be cautious in interpreting the R2 latency delay during the interictal phase, due to the relatively low number of patients in our series and the possible interference of antiepileptic drugs, the R2 latency reduction during the ictal phase strongly resembles what has been previously described during the migraine attack (37), thus suggesting similar mechanisms in AHC and migraine.
Footnotes
Acknowledgements
We thank the Associazione Italiana per la Sindrome di Emiplegia Alternante (AISEA) for its continuous organizational assistance and support.
