Abstract
A growing body of evidence supports the pivotal role of the hypothalamus in the pathophysiology of cluster headache (CH). On the basis of animal studies, it has been suggested that a hypothalamic dysfunction can lead to a habituation deficit of brainstem reflex responses, as result of a stress-like condition. Taking into account these findings, we tested the hypothesis that habituation of brainstem reflexes may be impaired in CH patients. The habituation phenomenon of the late components (R2 and R3) of the blink reflex was studied in 27 CH patients during the cluster period, in 22 migraine patients interictally and in 20 control subjects. A significant habituation deficit in the R2 and R3 components was found in CH compared with both controls and migraineurs. The lack of habituation in CH, more pronounced than in migraine, points to abnormal processing of sensory stimuli at the trigeminal level that could be driven by hypothalamic dysfunction during the cluster period.
Introduction
The pathogenesis of cluster headache (CH) is still not completely understood, even though several studies have provided evidence suggesting that peripheral and central structures are involved (1–5).
The pain in CH, as in migraine, is thought to be mediated by activation of the trigeminal nerve, whereas the autonomic symptoms are due to activation of the cranial parasympathetic outflow from the seventh cranial nerve (6). This latter activation is, in many ways, the clinical signature of CH, differentiating it from migraine. Several lines of study suggest that central nervous system mechanisms play an important role in CH pathogenesis. In view of the striking circadian rhythm shown by CH attacks, the hypothalamus, a biological clock, is strongly suspected to play a role in triggering CH pain (6). During the past decade, indeed, functional neuroimaging investigations have highlighted increased blood flow during the pain attacks (3, 7) and structural changes (8) in the hypothalamic grey matter in CH. Furthermore, there is a growing body of evidence indicating clinical efficacy of deep brain stimulation of the posterior hypothalamus in intractable forms of chronic CH (9–11).
However, further investigations are needed in order to establish whether the hypothalamus acts as the primary generator of CH, via direct modulation of the activity of neural circuits involved in pain transmission, including the trigeminal nucleus caudalis (3, 6, 12), or whether it is a pain modulatory structure activated by primary nociceptive trigeminal inputs (4, 9).
Studying trigeminal reflexes is a means of exploring directly the connection between the trigeminal nerve and the facial nerve, and a useful method for investigating the functional state of the trigeminal brainstem complex (13, 14). The blink reflex (BR), in particular, has been shown to be highly sensitive to changes in trigeminal activity. The BR consists of two principal components: an early ipsilateral oligosynaptic one (R1), and a late bilateral polysynaptic one (R2). Moreover, a later reflex component, R3, is sporadically elicited when applying strong electrical pulses or by innocuous stimuli, such as an acoustic tone. The R3 component of the BR is considered part of the startle reaction (15) and a fast-adapting model of the trigeminal reflex response (16).
During the past 5 years, analysis of R2 habituation has proved to be a particularly sensitive tool for revealing functional changes in the excitability of the trigeminal system in migraine patients (17–21). In the interictal period, in particular, deficient habituation of the R2 component of the BR has been clearly demonstrated in migraineurs (17–21), mimicking that usually observed at cortical level (22).
Habituation (a reduction of a response as a result of repeated stimulation) is a form of non-associative learning and is one of the most well-documented and fundamental forms of nervous system plasticity (23).
Interestingly, in mice, dysfunction of the hypothalamus–pituitary–adrenal (HPA) axis has been found to cause a lack of habituation of the startle reflex (24). It has been suggested that this dysfunction produces a chronic stress-like condition leading to abnormal processing of relevant environmental stimuli (24). Eliciting the BR through electrical stimulation, Formisano et al. (25) have also found abnormal habituation mechanisms in a small number of CH patients during the attack, but comparison with control subjects is lacking.
We therefore looked for an abnormal habituation pattern of the R2 component of the BR in CH patients during bouts, and compared the results with findings in healthy volunteers. We also analysed the R3 component of the BR because, as mentioned above, it seems to be part of the startle reaction and may be seen as a fast-adapting model of trigeminal reflex response (16) that could yield further information on habituation phenomena involving the trigeminal circuits. Our working hypothesis was that CH patients, during a bout, may show impaired habituation of both the R2 and the R3 components of the BR. Moreover, it was decided to compare the CH patients' and healthy volunteers' reflex responses with those of an additional group of subjects affected by migraine without aura in order to look for evidence of possible differential impairment of habituation mechanisms in these two types of primary headache.
Materials and methods
Subjects
Forty-nine patients (13 female and 36 male; age 23–67 years, mean 36.4 ± 11.5 years) were recruited among those seeking treatment at the University Centre for Adaptive Disorders and Headache (IRCCS ‘C. Mondino Institute of Neurology’ Foundation, University of Pavia, Italy). According to the diagnostic criteria set out in the 2nd edition of the International Classification of Headache Disorders (1), 27 patients had episodic cluster headache (CH, code 3.1.1; five female and 22 male; age 24–67 years, mean 36.9 ± 12.6 years; mean attack frequency 2.8 ± 1.2/day; mean cluster duration 6 ± 2 weeks; mean attack duration 43 ± 24 min) and 22 patients migraine without aura (MoA, code 1.1; eight female and 14 male; age 23–58 years, mean 35.5 ± 10.0 years; mean attack frequency 4.13 ± 1.95/month; mean disease duration 22 ± 8.1 years). Patients with CH experienced strictly unilateral pain (12 right side; 15 left side), whereas patients with MoA had a higher attack prevalence on one side (10 right side; 12 left side). Multiple diagnoses were not allowed. Other primary headaches or secondary headaches were excluded by clinical and/or instrumental evaluation, as appropriate. Exclusion criteria included any serious systemic or neurological disease or psychiatric disorder. CH patients with family history (first-degree relatives) of migraine were excluded. The patients were compared with 20 sex- and age-matched healthy volunteers (HVs, five female and 15 male, age 23–49 years, mean 32.02 ± 6.6 years), who were recruited among students of the medical school and healthcare professionals. They were studied in parallel with patients. The HVs had never presented neurological disorders and had no personal or family (first- or second-degree relatives) history of disabling headaches or aura-like symptoms. No patient had been taking prophylactic medication for headache in the previous 3 months, and all were self-medicated with triptans (CH or MoA) or with oxygen therapy (CH). The CH patients were recorded during a bout, but outside the attacks, and the MoA patients had to be attack-free for ≤ 3 days prior to and after the recording sessions (they were telephoned to verify this).
All participants were given a complete description of the study and gave their informed consent. The local ethics committee approved the study.
Blink reflex measurement
The subjects were comfortably settled in an armchair in a quiet room and were asked to sit back and relax, keeping their eyes open. All recording sessions were performed in the morning (between 09.00 and 11.00 h) always by the same expert neurophysiologist.
Stimulation
The BR was elicited by electrical stimulation (monopolar square-wave pulse with a duration of 0.3 ms, delivered by a constant current stimulator) at the emergence of the supraorbital nerve by means of surface electrodes placed longitudinally 2 cm apart. The stimuli were applied on the usual headache side in both the CH and MoA patients, and always on the right side in HVs.
In each participant, the sensory threshold (ST) and pain threshold (PT) were determined on the basis of a sequence of stimuli of increasing and decreasing intensity (increased or decreased in 1-mA steps) delivered at unpredictable intervals (of ≤ 10 s). Subjects were asked to indicate verbally the stimulation levels at which they became aware of sensory and pain sensations. The subjective pain sensation elicited by supraorbital nerve stimulation was graded on an 11-item numeric rating scale (NRS; 0, no pain; 10, severe pain).
Once the ST had been determined, the reflex threshold (RT) was separately defined for the R2 and R3 components by raising the stimulus intensity until a stable reflex response appeared and persisted over a series of five stimuli without producing pain. If no reflex responses (R2 or R3) were detected, although PT was reached, the reflex was considered not elicitable.
For the RT assessment, in order to avoid R2 and R3 habituation, the stimuli were delivered at pseudorandom frequencies between 0.033 and 0.025 Hz (16).
Electromyography recordings
Electromyographic (EMG) signals were recorded from both orbicularis oculi muscles via a standard pair of Ag/AgCl surface electrodes placed on the midline of the lower eyelid and on the bridge of the nose, near the inner canthus of the eye. The ground electrode was placed on the subject's forehead.
The filter bandpass settings were between 3 Hz and 3 kHz. The analysis time was 200 ms, sensitivity set at 100 µV (Medelec Synergy, Oxford UK). The signals were amplified and full-wave rectified.
The latency (L), visually determined as the take-off point from the baseline, and area under the curve (AUC) of the R2 and R3 components were automatically measured at the intensity corresponding to 1.3 times the RT and expressed in ms and in mV × ms, respectively. For each component, the time window to calculate the AUC was defined according to the measurable latencies of the best-defined template, both at the beginning and at the end of the component, and was then kept constant in each subject. For the L and AUC basal assessment, at least three to five successful responses were recorded and averaged in all participants.
Habituation
To evaluate the habituation phenomenon of the R2 component of the BR, a series of electrical stimuli delivered at different, randomly chosen stimulation frequencies (0.2, 0.3, 0.5, 0.7 and 1 Hz) were used. We chose to vary the stimulation frequency parameter because it has been shown that it could influence R2 response area and, consequently, habituation rate (18). The stimulus intensity was set at 1.3 times the R2 RT. A sequence of 16 consecutive rectified EMG responses was recorded for each stimulation frequency (SF). The first sweep of each sequence of responses was excluded from further analysis to avoid contamination with a startle response. In off-line analysis, the sequence of responses for each SF was subdivided into three blocks of five and the R2 AUC values were calculated and averaged for each block of responses. The mean AUC values of the second and third blocks, expressed as the percentage of the mean AUC value of the first block, were taken as an index of habituation for each SF.
Moreover, in a separate session following that of R2 by 20 min, habituation of the R3 component was assessed by evaluating the reflex responses to stimuli delivered at two randomly chosen SFs (0.066 and 0.033 Hz). The intensity of the stimulus was set at 1.3 times the R3 RT. For each SF, five consecutive rectified EMG responses were recorded, and the AUC values of the second to the fifth responses, expressed as a percentage of the first response, were taken as an index of habituation. This method makes it possible to analyse the habituation phenomenon of the R3 component, which is not possible with short interstimulus intervals, because of its fast adaptation (16).
Statistical methods
The Statistical Package for the Social Sciences (SPSS) for Windows, version 13.0, was used for all analyses (SPSS Inc., Chicago, IL, USA).
The Mann–Whitney test for independent samples was used to compare the baseline neurophysiological parameters (ST, PT, RT and L and AUC of the R2 and R3) between the three groups of subjects participating (HVs, MoA, CH). The Wilcoxon signed ranks test for related samples was used to check for ipsilateral–contralateral differences. In addition, to compare habituation differences between the three groups of subjects participating (HVs, MoA, CH), multivariate analysis of variance (
Furthermore, in order to compare the different R2 and R3 components' habituation rates between the stimulation frequencies, a
The level of significance was set at 0.05. All values were reported as means ±
Results
Blink reflex baseline parameters
The R2 response was elicited in all subjects. Conversely, the R3 component was clearly detected in 17 (73.9%) of the 20 HVs, in 19 (86.3%) of the 22 patients with MoA and in 25 (92.5%) of the 27 CH patients.
No statistically significant differences emerged in the ST, PT and RT values, AUC and L of the R2 and R3 components of the BR (ipsi- and contralateral) between the three groups of subjects (HVs, MoA, CH) (Table 1).
Mean values ±

HV, healthy volunteers; MoA, migraine without aura patients; CH cluster headache patients; ST, sensory threshold; PT, pain threshold; RT, reflex threshold; L, latency; AUC, area under the curve.
Blink reflex habituation
Instead, clear differences emerged between the three groups of subjects in the habituation of the ipsilateral R2 and R3 components of the BR. Since no ipsilateral vs. contralateral differences in habituation rate of the R2 and R3 components were detected in any group at any SF, we here report only the ipsilateral responses (Tables 1 and 2).
Mean area under curve values ±
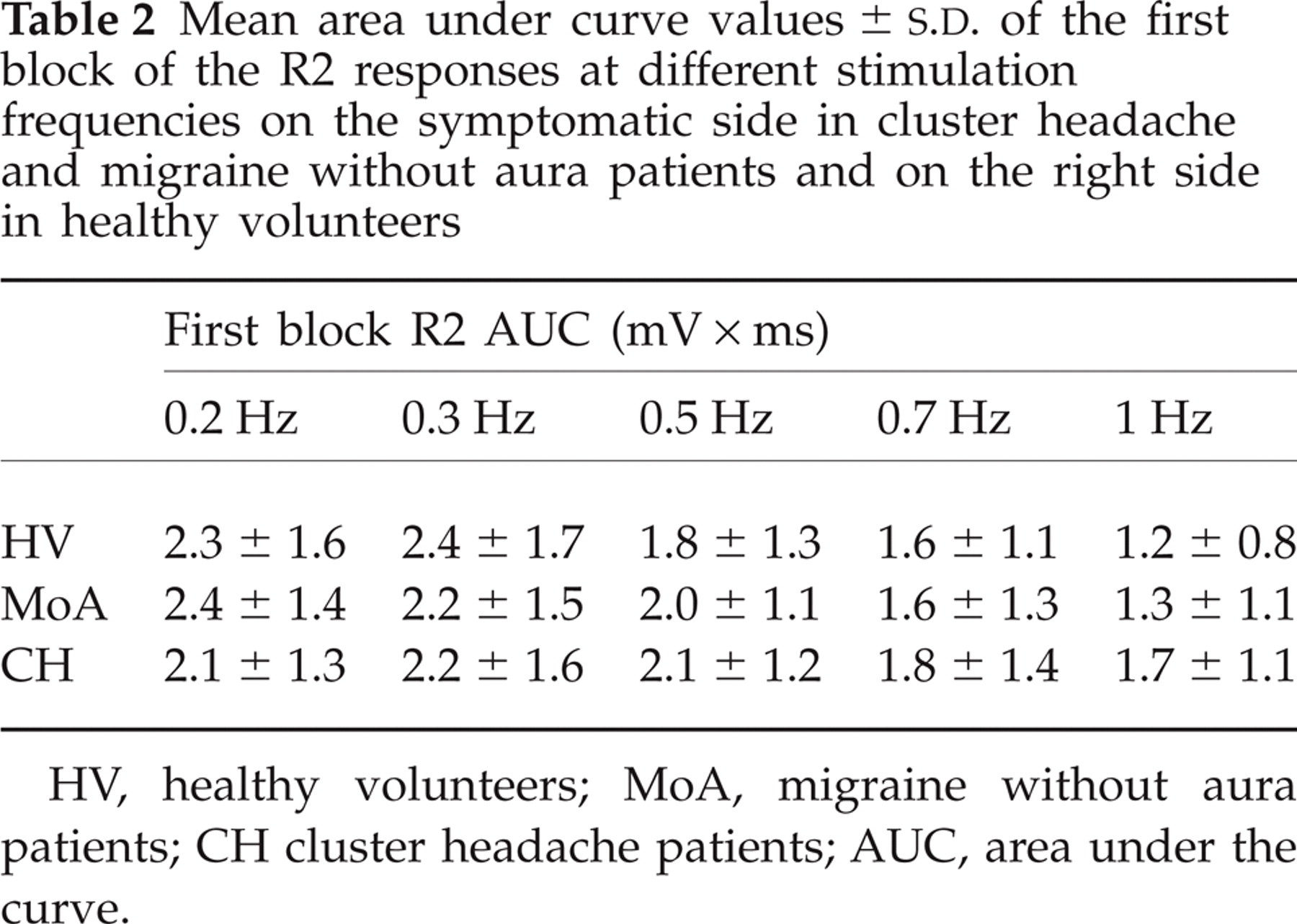
HV, healthy volunteers; MoA, migraine without aura patients; CH cluster headache patients; AUC, area under the curve.
A clear habituation of the R2 and R3 components of the BR was observed in the HVs across the three blocks and at every SF considered (Figs 1 and 2).
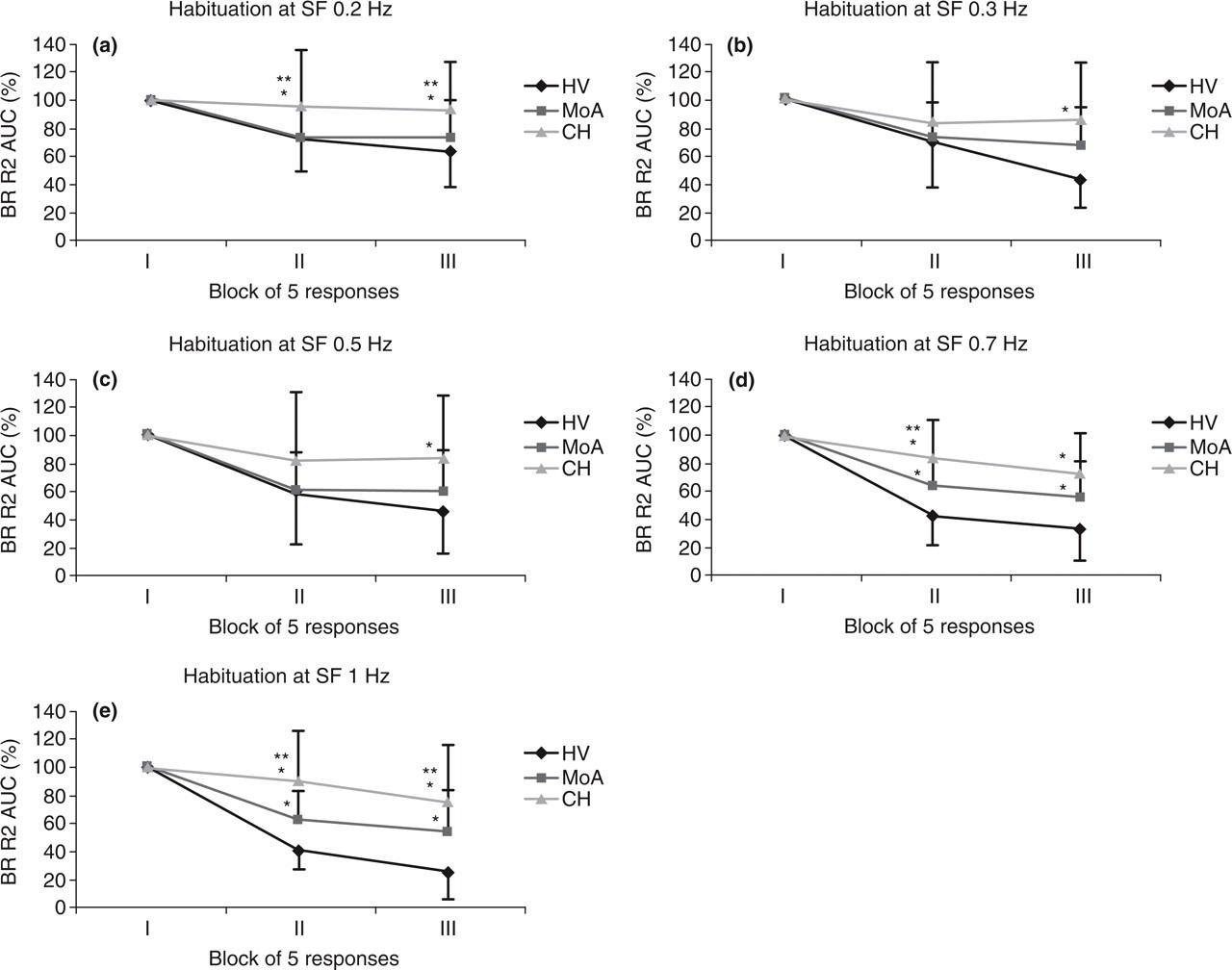
Habituation of the ipsilateral R2 area under the curve (AUC) in three blocks of five averagings at increasing stimulation frequencies (SF) (a, 0.2 Hz; b, 0.3 Hz; c, 0.5 Hz; d, 0.7 Hz; e, 1 Hz) expressed as a percentage of the first block. Data are shown as mean values and standard deviations of the mean. BR, blink reflex; CH, cluster headache patients; MoA, migraine without aura patients; HVs, healthy volunteers. Bonferroni test ∗P < 0.05 vs. HVs; ∗∗P < 0.05 vs. MoA.
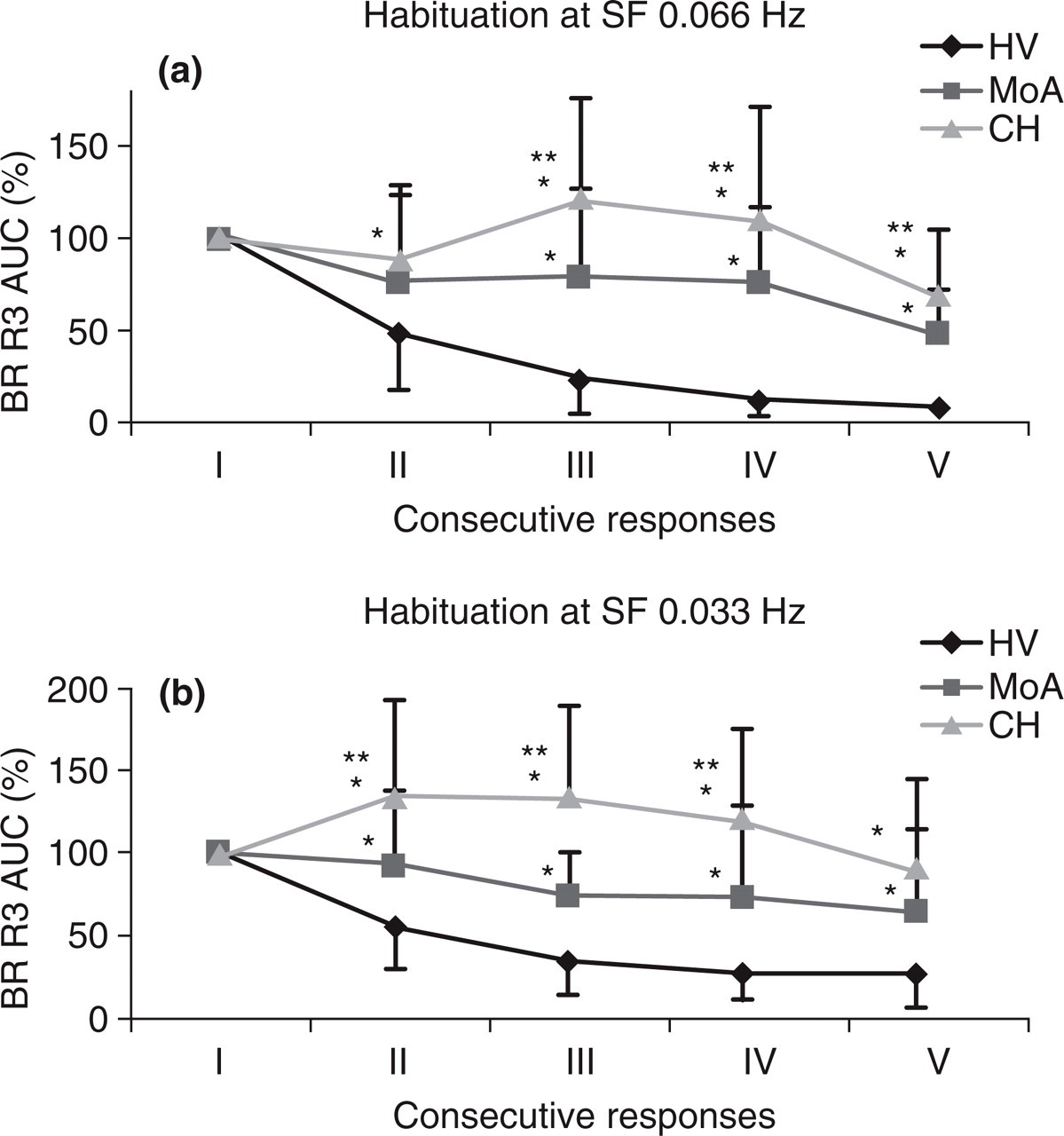
Habituation of the ipsilateral R3 area under the curve (AUC) of five consecutive responses at two stimulus frequencies (SF), 0.066 Hz (a) and 0.033 Hz (b), expressed as a percentage of the first response. Data are shown as mean values and standard deviations of the mean. BR, blink reflex; CH, cluster headache patients; MoA, migraine without aura patients; HVs, healthy volunteers. Bonferroni test ∗P < 0.05 vs. HVs; ∗∗P < 0.05 vs. MoA.
Multivariate analysis revealed significant between-group (HVs, MoA, CH) differences in R2 habituation at 0.2 Hz (F2,66 = 4.62, P = 0.001), 0.7 Hz (F2,66 = 18.73, P = 0.001) and 1 Hz (F2,66 = 20.71, P = 0.001) in the second block of responses (Fig. 1a,d,e), and at 0.2 Hz (F2,66 = 6.69, P = 0.002), 0.3 Hz (F2,66 = 9.29, P = 0.0001), 0.5 Hz (F2,66 = 7.23, P = 0.001), 0.7 Hz (F2,66 = 11.46, P = 0.0001) and 1 Hz (F2,66 = 11.95, P = 0.0001) in the third block (Fig. 1a–e).
In comparison with the HVs, CH patients showed a lack of habituation of the R2 component at every SF (Fig. 1), with significant differences at 0.2, 0.7 and 1 Hz (all P < 0.05) in the second block of responses (Fig. 1a,d,e), and at 0.2, 0.3, 0.5, 0.7 and 1 Hz (all P < 0.05) in the third block (Fig. 1a–e).
Similarly, multivariate analysis revealed significant between-group (HVs, MoA, CH) differences in R3 habituation at two SFs: 0.066 Hz (2nd F2,60 = 4.52, P = 0.015; 3rd F2,60 = 22.70, P = 0.0001; 4th F2,60 = 24.55, P = 0.0001; 5th F2,60 = 25.82, P = 0.0001) (Fig. 2a) and 0.033 Hz (2nd F2,60 = 14.71, P = 0.0001; 3rd F2,60 = 33.62, P = 0.0001; 4th F2,60 = 18.70, P = 0.0001; 5th F2,60 = 10.09, P = 0.0001) (Fig. 2b).
The R3 component showed marked lack of habituation in the patients with CH compared with the HVs, with significant differences at both 0.066 and 0.033 Hz in all SFs (2nd to 5th) (all P < 0.05) (Fig. 2a,b).
The CH patients also showed a more pronounced habituation deficit of the R2 and R3 components with respect to MoA patients (Figs 1 and 2). The habituation deficit of the R2 component was statistically significant at the 0.2-, 0.7- and 1-Hz SFs in the second block of responses (P < 0.05, Fig. 1a,d,e) and at the 0.2- and 1-Hz SFs in the third block (P < 0.05, Fig. 1a,e) in the CH vs. MoA patients. The habituation deficit of the R3 component in CH patients differed significantly from that shown by MoA patients from the third to the fifth response at the SF of 0.066 Hz (P < 0.05, Fig. 2a), as well as from the second to the fourth response at the SF of 0.033 Hz (P < 0.05, Fig. 2b).
MoA patients showed significantly reduced R2 habituation at the 0.7- and 1-Hz SFs both in the second and third block of responses (P < 0.05, Fig. 1d,e) compared with the HVs. Moreover, in MoA patients the habituation of the R3 component too was also found to be reduced compared with that recorded in HVs, from the third to the fifth response at 0.066 Hz and from the second to the fifth response at 0.033 Hz (P < 0.05, Fig. 2a,b).
In patients groups (MoA and CH), multivariate analysis was unable to show differences in R2 habituation rate between the different SFs in either second or third block of responses. This was not the case in the HV group, where significant differences were found in both second (F4,99 = 6.054; P < 0.001) and third (F4,99 = 6.506; P < 0.001) blocks of responses. Post-hoc analysis revealed a significant habituation effect at 0.7 and 1 Hz with respect to both 0.2 Hz (P < 0.01 for both) and 0.3 Hz (P < 0.01 and P < 0.05, respectively) in the second block and with respect to 0.2 Hz (P < 0.01 for both) in the third block.
As regards the R3 component, in patients (both CH and MoA) a significantly higher degree of habituation at 0.066 Hz with respect to 0.033 Hz in the second (t 1,24 = 4.857, P < 0.01 in CH and t 1,18 = 3.028, P < 0.01 in MoA) and fifth (t 1,24 = 3.054, P < 0.05 in CH and t 1,18 = 2.710, P < 0.05 in MoA) responses was found. In the HV group a significantly higher degree of habituation at 0.066 Hz with respect to 0.033 Hz was found in all responses (2nd t 1,16 = 3.189; 3rd t 1,16 = 3.785; 4th t 1,16 = 5.020; 5th t 1,16 = 4.825, all P < 0.01).
Discussion
The main finding of this study was that CH patients, during cluster periods, showed habituation deficits of the R2 and R3 components of the BR elicited on the symptomatic side. These deficits were even more pronounced than those observed in MoA patients.
Moreover, both the R2 and R3 components showed a significant increase of the habituation rate at the higher frequencies of stimulation (0.7 and 1 Hz for R2 and 0.066 Hz for R3) with respect to the lower frequencies (0.2 and 0.3 Hz for R2 and 0.033 Hz for R3) in HVs (higher habituation rate), with almost no change in MoA and CH (reduced or no habituation). These data resemble the findings obtained by other authors in normal subjects as well as in migraineurs (18).
In addition, no group differences were observed in baseline parameters, such as thresholds, latencies and AUC of either the R2 or R3 responses. These findings reflect those of previous studies that have shown a segmental functional integrity of the trigemino-facial circuits in CH patients (25–28).
Taken together, these observations suggest an abnormal suprasegmental modulation of trigemino-facial reflex excitability in CH during the cluster period.
To the best of our knowledge, this is the first study to explore habituation of the R2 and R3 components of the BR in CH patients during cluster periods and to compare them with those of healthy subjects and MoA patients.
The neural network that mediates the polysynaptic components (R2 and R3) of the BR includes the nucleus trigeminalis caudalis, excitatory interneurons of the bulbo-pontine lateral reticular formation, and motor neurons in the pontine facial nuclei innervating the orbicularis oculi muscles (13). These polysynaptic reflex components are modulated by segmental and suprasegmental mechanisms, and are subject to habituation phenomena (14).
Short-term habituation is a multifactorial and still poorly understood event, defined as ‘a response decrement as a result of repeated stimulation’, and it usually exhibits an exponential course (23).
The habituation of spinal reflexes has been suggested to be mediated by a variety of segmental (‘intrinsic’) mechanisms, including synaptic inhibition (pre or post), homosynaptic depression and membrane desensitization (23, 29–33). Moreover, suprasegmental (‘extrinsic’) mechanisms are also essential in determining the degree of response reduction (23, 34). In mammals, including humans, a complex descending modulatory pathway possibly involving the reticular formation and related structures, such as brainstem monoaminergic nuclei, may be important in mediating habituation of several kinds of sensory stimulus (e.g. auditory, visual, and so on) (23, 35–37).
Reduced BR habituation is a well-documented abnormality in neurological diseases including Parkinson's disease (38) and migraine (17–21). Several hypotheses have been advanced that explain this lack of habituation as a consequence of centrally driven defective control of trigeminal excitability. In Parkinson's disease, clear and direct evidence has been provided in favour of dopaminergic modulation of BR habituation (38). Interestingly, it has been suggested that the dopaminergic system may play a role in migraine and CH pathogenesis (39–41). It has recently been shown in rats that dopamine can inhibit the activation of trigeminocervical neurons in response to middle meningeal artery stimulation (42).
From this perspective, dopaminergic dysfunction could explain the deficient habituation of the BR observed in migraine as well as in CH patients (18, 21). Moreover, it has been shown that the dopaminergic control pathways modulate BR excitability via serotoninergic transmission by the nucleus raphe magnus, which, in turn, inhibits the spinal trigeminal nucleus (43). Therefore, the deficient habituation observed in CH patients may, as hypothesized in migraine, be regarded as a consequence of low activity in central serotoninergic pathways.
The habituation deficit observed in both CH and migraine in our study suggests that these primary headaches share some pathophysiological mechanisms; however, the more pronounced habituation deficit found in CH with respect to the migraine group suggests that additional dysfunctional neurobiological factors could be at work in CH patients.
Since involvement of the hypothalamus in the pathogenesis of CH has been widely demonstrated at functional and biochemical level (3, 7, 44–46), it is reasonable to hypothesize that the hypothalamus could also be involved in the habituation deficit observed in our CH patients. In fact, the hypothalamus is known to have a modulatory effect on the trigeminovascular nociceptive system and on autonomic pathways (47, 48). Dirks et al. (24) have found that transgenic mice overexpressing corticotrophin-releasing hormone did not display any habituation of the startle reflex, suggesting that the HPA axis hyperactivity reduced startle reactivity and plasticity.
Taking these facts together, the habituation deficit of the BR observed in our CH patients supports the hypothesis of centrally driven disinhibition of the processing of sensory stimuli at trigeminal level. This could be due to several supraspinal dysfunctions, including dysmodulation of aminergic and hypothalamic pathways.
Moreover, we cannot a priori exclude that sensitization of the trigeminal pathways during the cluster period in CH patients may be one of the pathophysiological processes underlying the habituation deficit observed in our patients.
Lozza et al. (27) have shown reduced inhibition of the R2 recovery curve on the headache side in patients with CH after segmental conditioning stimulation, and on both sides after extrasegmental conditioning stimulation. The authors took these results to be indicative of hyperexcitability of the spinal trigeminal complex neurons (27).
The development of cutaneous allodynia and underlying central sensitization of the trigeminal structures in CH is a topic of debate given the contrasting results obtained in different studies (49, 50). However, it has been demonstrated in several animal models that sensitization and habituation phenomena may coexist (51–54).
The Groves and Thompson ‘dual-process’ theory suggests that the ‘dishabituation’ effect is ‘a separate facilitatory process superimposed upon the habituated system’ and that this dual process of habituation and sensitization is able to account for all the incrementing and decrementing behavioural responses (51, 53). Thus, dishabituation may be a form of sensitization that is gated by habituation (55). This behaviour may be finely modulated by descending pathways (23, 51, 55, 56).
On the basis of these findings, the sensitization of the trigeminal pathways during the cluster period in CH patients may be one of the pathophysiological processes underlying the deficient habituation observed in our patients. Furthermore, given that CH patients did not show a habituation deficit of event-related potentials (57, 58), it is possible that the neurobiological factors in the pathogenesis of CH could have an effect at cortical level that is different from that at the trigeminal pathway level.
Although the findings from the current study are promising, some limitations should be acknowledged: first, the lack of non-painful side stimulation in CH patients, which would have permitted additional hypothesis concerning the site of physiopathological abnormalities in CH patients, as well as the lack of blinding to diagnosis of the investigator who performed neurophysiological tests; second, the slightly, although not significant, between-group differences in age range.
In conclusion, taking into account the recent findings in CH pathophysiology, we have hypothesized that dysfunctioning descending aminergic and hypothalamic trigeminal control as well as sensitization phenomena occurring at the level of the nucleus trigeminalis caudalis could be the underlying cause of the abnormal trigeminal processing observed in our CH patients during the cluster period. However, literature data on the habituation phenomenon are still scant, and other studies on evoked potentials and brainstem reflexes are needed to clarify the role of habituation/sensitization processes in CH during the cluster period.
