Abstract
The aim of the present study was to verify cerebrospinal fluid (CSF) levels of glial cell line-derived neurotrophic factor (GDNF) and somatostatin, both measured by sensitive immunoassay, in: 16 chronic migraine (CM) patients, 15 patients with an antecedent history of migraine without aura diagnosed as having probable chronic migraine (PCM) and probable analgesic-abuse headache (PAAH), 20 patients affected by primary fibromyalgia syndrome (PFMS), and 20 control subjects. Significantly lower levels of GDNF and somatostatin were found in the CSF of both CM and PCM + PAAH patients compared with controls (GDNF = P < 0.001, P < 0.002; somatostatin = P < 0.002, P < 0.0003), without significant difference between the two groups. PFMS patients, with and without analgesic abuse, also had significantly lower levels of both somatostatin and GDNF (P < 0.0002, P < 0.001), which did not differ from those of CM and PCM + PAAH patients. A significant positive correlation emerged between CSF values of GDNF and those of somatostatin in CM (r = 0.70, P < 0.02), PCM + PAAH (r = 0.78, P < 0.004), and PFMS patients (r = 0.68, P < 0.008). Based on experimental findings, it can be postulated that reduced CSF levels of GDNF and somatostatin in both CM and PCM + PAAH patients can contribute to sustained central sensitization underlying chronic head pain. The abuse of simple or combination analgesics does not seem to influence the biochemical changes investigated, which appear to be more strictly related to the chronic pain state, as demonstrated also for fibromyalgia.
Keywords
Introduction
Data obtained in experimental animal models support the role of certain neurotrophins as putative candidates implicated in the pathogenic mechanisms underlying chronic pain, including chronic head pain. Few studies have been performed to establish their role in maintaining pain states in humans. Increased levels of nerve growth factor (NGF) have been demonstrated in the cerebrospinal fluid (CSF) of patients with fibromyalgia and patients affected by chronic daily headache (CDH) without significant difference between patients with and without analgesic abuse in the latter (1, 2).
More recently, a significant correlation has emerged between the levels of NGF and brain-derived neurotrophic factor (BDNF) and also glutamate in the CSF of CDH patients with a previous history of migraine, suggesting the putative intervention of both neurotrophins in enhancing glutamatergic transmission underlying chronic sensitization in CDH (3, 4).
Other members of the neurotrophic factor family have never been investigated in primary chronic headaches, including glial cell line-derived neurotrophic factor (GDNF). GDNF belongs to the superfamily of transforming growth factor (TGF)-β and exerts its action through the binding to specific GDNF family receptor α (GFRα) proteins and signals through the c-ret receptor tyrosine kinase (5). Its trophic action on sensory neurons is well known and it has been recognized to promote the survival of a subgroup of developing sensory neurons, which express GDNF receptors and possess the binding sites for isolectin Griffonia Simplicifolia (IB4) in the postnatal period, instead of expressing the TrkA high-affinity receptors of NGF (6, 7).
Immunohistochemical studies have revealed the presence of GDNF in the superficial layers of the spinal cord, which are closely related to pain (8, 9). GDNF also appeared to have a pain-relief effect in neuropathic pain models (10–12), possibly through regulation of the expression of sodium channel subunits (13), capsaicin VR1 receptors, and substance P (SP) release (14).
The relevance of GDNF in nociceptive neurons in both the human spinal trigeminal nucleus and nucleus caudalis has been shown by the histochemical findings of GDNF-immunostained fibres and terminal-like structures, codistributed with SP and calcitonin gene-related peptide (CGRP) in these nuclei (15). The occurrence of GDNF in the superficial layers of the trigeminal nucleus caudalis supports its contribution in modulating nociceptive processing in the trigeminal system, as proposed in experimental animals, and its involvement in changes of its expression and release in chronic migraine (CM).
Details of patients
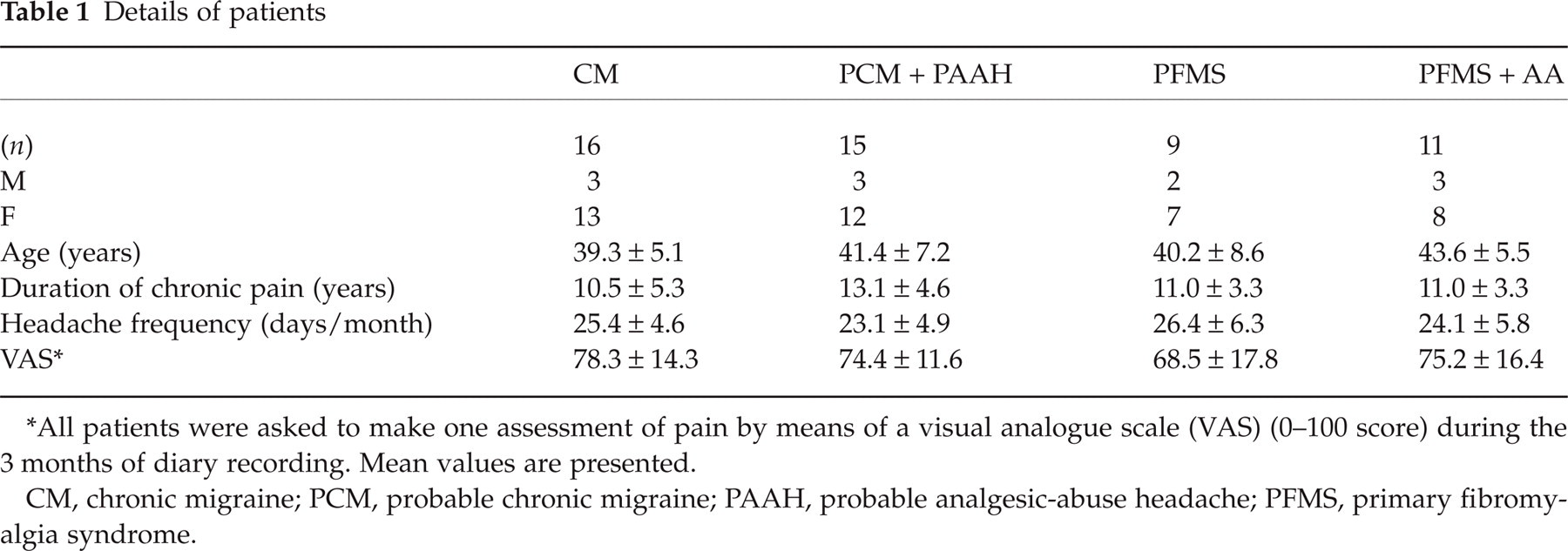
All patients were asked to make one assessment of pain by means of a visual analogue scale (VAS) (0–100 score) during the 3 months of diary recording. Mean values are presented.
CM, chronic migraine; PCM, probable chronic migraine; PAAH, probable analgesic-abuse headache; PFMS, primary fibromyalgia syndrome.
The expression of somatostatin, a neuropeptide with a well-known antinociceptive effect, is influenced by GDNF. Recent findings have shown that GDNF modulates the activity-induced release of endogenous somatostatin from both the central and peripheral terminals of adult sensory neurons (16). This novel mechanism deserves exploration as a putative therapeutic strategy to control not only inflammation but also pain (17).
All the above evidence from experimental models prompted us to investigate the involvement of both GDNF and somatostatin in CM and probable chronic migraine plus probable analgesic-abuse headache (PCM + PAAH).
Materials and methods
Patients and controls
The study protocol was approved by the Ethics Committee of the Azienda of Umbria and all patients gave their written consent to the study. Thirty-one consecutive patients affected by CM (n = 16) or PCM + PAAH (n = 15), attending the Headache Centre of the Neuroscience Department of the University of Perugia, were admitted to the study. According to the ICHD 2nd edition (18), inclusion criteria for 1.5.1 Chronic migraine were headache for at least 15 days per month for at least 3 months, and headache characteristics fulfilling those of migraine without aura in the absence of drug abuse. In the case of analgesic abuse, patients were coded according to the antecedent migraine subtype 1.1 Migraine without aura plus 1.6.5 Probable chronic migraine plus 8.2.7 Probable medication-overuse headache.
A further patient group included 20 patients affected by primary fibromyalgia syndrome (PFMS). Diagnosis of PFMS was made according to the American College of Rheumatology Criteria (1990) (19). These criteria included widespread pain (axial plus upper and lower segments plus left- and right-side pain) in combination with tenderness in at least 11 of 18 specific tender point sites. Widespread pain was scored with a visual analogue scale (VAS) (1–100). None of the PFMS patients suffered from episodic headaches (both tension-type and migraine types) or CM or chronic tension-type headache. In all patient groups women were in the premenopausal period. Eleven PFMS patients took analgesics for ≥15 days/month for at least 3 months and were classified as analgesic abusers (PFMS + AA).
The abused analgesic drugs in the PCM + PAAH and PFMS + AA groups are listed in Table 2. The monthly drug intake averaged 64.3 ± 13.5 (mean ± SD) and 68.3 ± 13.5 (mean ± SD) tablets or suppositories in the two patient groups, respectively. None of the patients used drugs containing codeine and/or took preventive medication for at least 1 month. They were assessed when free from prophylactic treatment for at least 20 days, and therefore before beginning a new prophylactic treatment cycle and/or undergoing detoxification in the case of analgesic abuse.
Analgesics abused by (PCM + PAAH) and (PFMS + AA) patient groups

The sum of the percentages exceeds 100% because the majority of patients used more than one simple analgesic or combination analgesic in association with one simple analgesic.
PCM, probable chronic migraine; PAAH, probable analgesic-abuse headache; PFMS, primary fibromyalgia syndrome; NSAID, non-steroidal anti-inflammatory drug.
All patients were admitted to the Neurologic Clinic of the Neuroscience Department of the University of Perugia to undergo lumbar puncture. Control CSF specimens were also obtained from 20 age-matched subjects who underwent lumbar puncture for diagnostic purposes. In all these subjects CSF and blood tests excluded central nervous system (CNS) or systemic diseases. All control subjects were drug-free for at least 2 months and none of them was taking any medication at the time of CSF sampling or had a personal or family history of migraine or suffered from tension-type headache. None of the above controls developed postlumbar puncture headache.
Methods
Quantification of GDNF in CSF
Quantitative analysis of human GDNF in the CSF was performed using an immunoassay system (Promega, Madison, WI, USA), in an antibody sandwich format according to the supplier's instructions. In this system, 96-well plates are coated with anti-GDNF monoclonal antibody (mAb) which binds soluble GDNF. The captured GDNF is bound by a second specific polyclonal antibody. After washing, the amount of specifically found polyclonal antibody is then detected using a species-specific antibody conjugated to horseradish peroxidase as a tertiary reactant. The unbound conjugate is removed by washing, and following incubation with a chromogenic substrate, the colour change is measured, recording the absorbance at 450 nm on a plate reader within 30 min of stopping the reaction. Using this system GDNF can be quantified in biological fluids, culture supernatants and tissue homogenates in the range of 10–1000 pg/ml. The specificity of the GDNF immunoassay system has been confirmed by the lack of detectable binding of functionally related proteins (natural and recombinant human TGF-α, NGF and BDNF) at concentrations as high as 10 ng/ml (cross-reactivity <3%). The coefficients of variance for intra-assay and interassay variabilities were 6.2% and 5.2%, respectively. Data were expressed as pg/ml.
Quantification of somatostatin in CSF
Immunoreactivity of somatostatin was determined by initial elution with 60% acetonitrile in 0.1% trifluoroacetic acid (TFA) on SEP-C18 columns activated with 0.1% TFA and 60% acetonitrile in 0.1% TFA. Eluates were desiccated after centrifugal concentration (Supervap PL-CC-180). Residues were reconstituted in 200 μl of assay buffer and 50 μl were assayed by radioimmunoassay kit following the manufacturer's instructions (Immunobiological Laboratories, Hamburg, Germany). Extracts were analysed by competitive assay, in which somatostatin in standards and samples competed with 125I-Tyr1-labelled somatostatin in binding to the antibodies. The 125I-Tyr1-somatostatin bound in a reverse proportion to the concentration of somatostatin in standards and samples, which were run in duplicate and triplicate, respectively. Antibody-bound 125I-Tyr1-somatostatin was separated from the unbound fraction using the double antibody–polyethylene glycol precipitation technique. The radioactivity of the precipitates was measured. Somatostatin concentrations in the samples were corrected for the per cent recovery calculated in the recovery controls. Somatostatin human antiserum was obtained from rabbit. The immunogen was cyclic somatostatin conjugated to bovine thyroglobulin. Somatostatin antiserum showed a 100% cross-reactivity for cyclic somatostatin and Tyr1-somatostatin, and no cross-reactivity for prosomatostatin-32, SP, amylin, cholecystokinin octapeptide (desulphated), vasoactive intestinal polypeptide (human, bovine, porcine, rat), and insulin (human). The intra- and interassay variabilities were 5% and 10%, respectively. A sensitivity of 10 pg/ml was calculated from a 2 SD decrease in binding of the zero standard. Data were expressed as pg/ml.
Statistical analysis
Data of each group (patients and controls) were expressed as mean ± 2 SEM and compared using the Mann–Whitney U-test. Correlation between variables was determined using Spearman's rank correlation test. P-values < 0.05 were considered significant.
Results
Chemical and cell examinations were performed immediately after lumbar puncture, and all values of both patient and control groups were found to be normal. No significant correlations emerged between the levels of GDNF and somatostatin in the CSF and age in all patient groups and controls. Patients with CM showed significantly lower levels of GDNF and somatostatin compared with those of control subjects (P < 0.001 and P < 0.002) (Table 3). A decrease in both GDNF and somatostatin values was also evident in PCM + PAAH patient groups compared with controls (P < 0.002 and P < 0.004). No significant differences emerged between CSF values of GDNF or somatostatin between these two groups. A significant positive correlation emerged between GDNF and somatostatin values in the CSF of CDH patients but not in control subjects (Fig. 1a,b). No significant correlations were found between CSF levels of both GDNF and somatostatin and duration of chronic headache as well as number of days with headache/month and VAS scores for pain. Significantly lower values of GDNF and somatostatin were also detected in the CSF of PFMS and PFMS + AA patient groups compared with controls (GDNF, P < 0.0008, P < 0.001; somatostatin, P < 0.0001, P < 0.002, for PFMS and PFMS + AA patients, respectively), without significant differences between the two patient groups (Table 3).

Plot of the levels of glial cell line-derived neurotrophic factor (GDNF) vs. somatostatin, both expressed as pg/ml, in patients with chronic migraine (CM) (a) and in patients with probable chronic migraine (PCM) + probable analgesic-abuse headache (PAAH) (b).
Cerebrospinal fluid levels of GDNF and somatostatin (mean ± 2 SEM) in patient and control groups

CM, chronic migraine; PCM, probable chronic migraine; PAAH, probable analgesic-abuse headache; PFMS, primary fibromyalgia syndrome.
For the correlation concerning PFMS patients, we considered the entire group independently from the presence of drug abuse, due to the limited number of patients and the lack of differences between PFMS patients with or without analgesic abuse. We found a significant correlation between CSF levels of GDNF and somatostatin in CM (r = 0.70, P < 0.02) and PCM + PAAH (r = 0.78, P < 0.004) groups, as in the whole group of PFMS and PFMS + AA patients (r = 0.68, P < 0.008) (Fig. 1a,b; Fig. 2). In both PFMS patient groups with or without analgesic abuse, CSF values of GDNF and somatostatin were not correlated with the duration of PFMS or intensity of pain as measured by VAS.
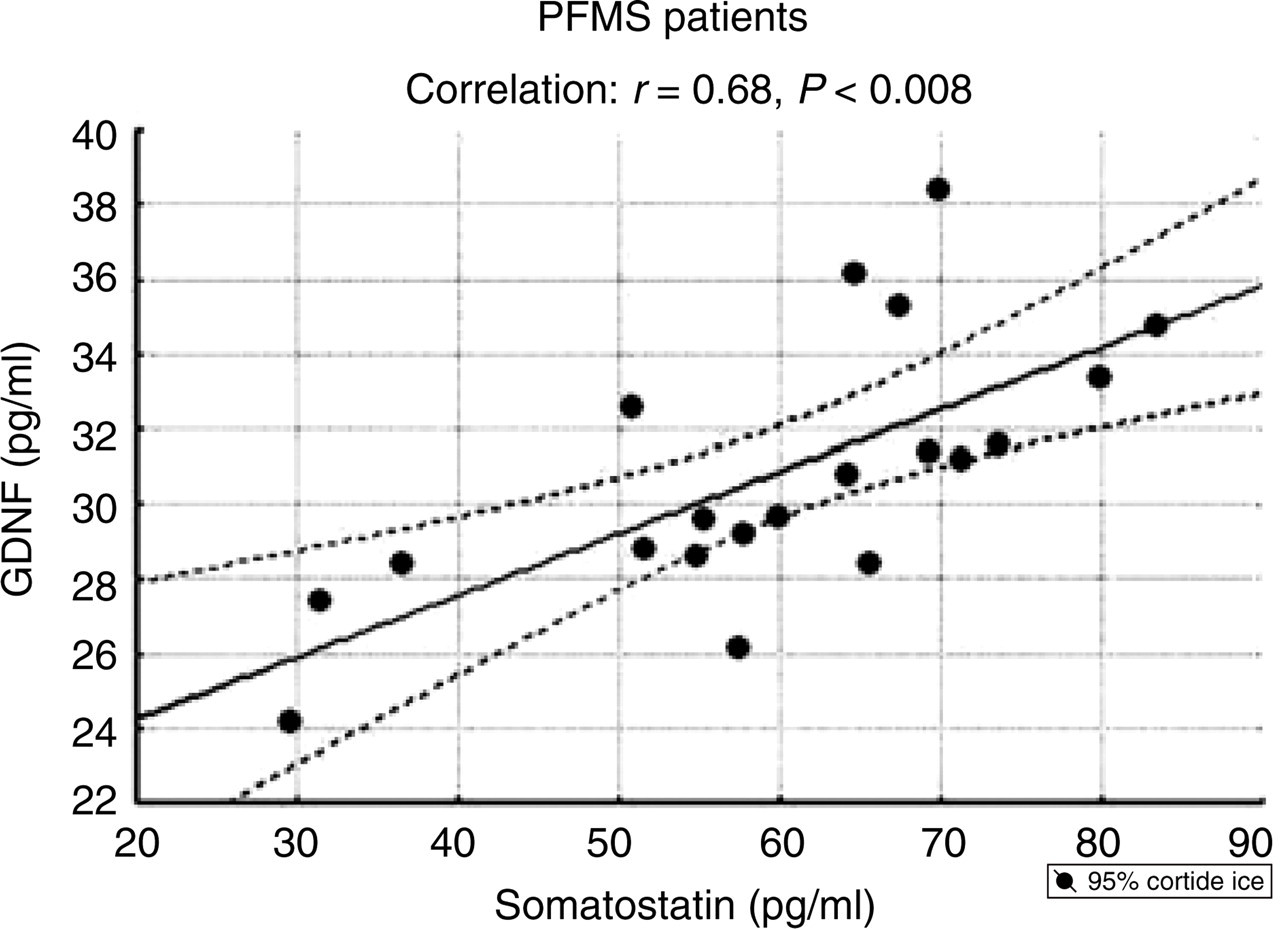
Plot of the levels of glial cell line-derived neurotrophic factor (GDNF) vs. somatostatin, both expressed as pg/ml, in patients with primary fibromyalgia syndrome (PFMS).
Discussion
Results of the present study support the reduction in CSF levels of the neurotrophic factor GDNF in patients with CM. This reduction seems unrelated to analgesic abuse, whereas a similar reduction is evident in patients with PCM or PAAH with a previous history of episodic migraine without aura, and seems therefore more strictly associated with chronic pain per se. This was further supported by the finding of reduced levels of GDNF in patients with primary fibromyalgia, which challenged the specificity of our finding for CM.
The reduction in GDNF levels found in patients with CM and fibromyalgia could be related to the pivotal role of this neurotrophic factor in preventing and reversing abnormalities that develop in chronic pain conditions. This has been clearly shown in experimental models of peripheral nerve injury and neuropathic pain, where GDNF is able to counteract the up-regulation of TTX-sensitive type III α sodium channels and rescue the down-regulation of the nonpeptidergic P2X3 receptors (13, 20).
Other mechanisms, advocated to explain the potent analgesic effect exerted by GDNF in pain states, include modification in the expression of SP and of the capsaicin receptor, VR1, two nociceptive properties already known to be NGF-regulated, and increase in the expression of somatostatin in nociceptive neurons dependent on activation of voltage-sensitive calcium channels (VSCCs) (14, 21).
We focused our attention particularly on somatostatin levels in the CSF, which appeared to be reduced in all patient groups and significantly correlated with reduced levels of GDNF.
This strict correlation suggests that central sensitization occurring in chronic pain states is, at least in part, attributed to the decrease in somatostatin release due to the failure of GDNF mechanisms, which can favour the prevalence of NGF-mediated nociceptive effects, as previously shown by our group (2, 3).
We did not directly investigate in the present study markers of the trigeminovascular system, such as CGRP, neurokinin A and SP, which appeared to be significantly increased in previous research carried out by our group in patients with CDH evolving from a previous history of episodic migraine without aura (2). With the known effect of GDNF on sensory neuropeptide expression, however, it can be hypothesized that reduction of this neuropeptide may contribute to their increased release, at least in CM. There is no clear proof of an increase in CGRP in patients with fibromyalgia. Levels of this sensory neuropeptide and of SP were measured in only one dated study, in which no reference was made to comparison with levels in control CSF samples (22). The authors concluded that the anatomical coexistence of pain-related neuropeptides in neurons was not necessarily reflected by the levels of these peptides measured in the CSF, and that the presence of CGRP in the CSF could be important, since it can enhance the nociceptive activity of tachykinins. No data are available on GDNF levels in the CSF of fibromyalgia patients. Moreover, there is no evidence in the literature concerning low back pain, and although a significant release of CGRP as well as VIP occurred in spontaneous or nitroglycerin-induced attacks in the external jugular blood of cluster headache patients during bouts, data from the CSF are lacking in both episodic and chronic forms (23, 24).
The role of somatostatin as a regulatory antinociceptive and anti-inflammatory peptide produced throughout the CNS and in most major peripheral organs is well known (25–28). The observation of the potent analgesic effect of somatostatin and its stable analogues (octreotide, lanreotide, vapreotide, etc.) in animal models prompted researchers to propose both somatostatin and its analogues for the treatment of opioid-resistant or postsurgery pain (29–31). This novel mechanism deserves exploration as a potential new therapeutic strategy also in the chronic treatment of refractory sine materia pain conditions, such as CM and fibromyalgia, especially in light of the finding of reduced peptide levels in the CSF of patients affected by these two chronic pain conditions. There are at this time only two small randomized, double-blind trials suggesting efficacy of somatostatin in cluster headache and findings of a recent, double-blind control study demonstrating a significant analgesic effect of a somatostatin analogue, octreotide, in aborting cluster headache attacks. There is, however, no evidence of an effect of somatostatin as preventive treatment in cluster headache, in both episodic and chronic subtypes (32–34).
Both above conditions are characterized by an exhaustibility of the stress system, which coordinates the adaptive responses of the organism to chronic pain. The main components of this system are the corticotropin-releasing hormone (CRH) and locus ceruleus-norepinephrine (LC/NE)-autonomic systems and their peripheral effectors, the pituitary–adrenal axis, and the limbs of the autonomic system (35).
Although impaired growth hormone production previously shown in fibromyalgia patients has been attributed to augmented hypothalamic somatostatin tone rather than to its failure (36, 37), no evidence of this is available for CM, and it can be hypothesized that the chronic hyperactivity of the stress system, as a consequence of chronic pain, could be responsible for its functional failure. Reduced levels of somatostatin in the CSF of patients affected by both pain conditions could be interpreted therefore as an indirect expression of impairment of the stress system. In light of these considerations, the use of potentiators of CRH secretion/action can also be proposed, which could influence somatostatin tone both in pain processing pathways and the stress system.
Footnotes
Acknowledgements
The authors express their gratitude to John A. Toomey for editing the English and Marisa M. Morson for technical assistance.
