Abstract
Our group has previously shown that migraineurs, as opposed to individuals with other headaches, are more likely to have headache during the bright arctic summer than during the polar night season. We set out to investigate the impact of seasonal light exposure in migraine with and without aura. We performed a questionnaire-based study of 169 female volunteer migraineurs in an arctic area where light conditions during summer and winter seasons are extreme. We included 98 patients with migraine with aura (MA) and 71 with migraine without aura (MoA). One hundred and seven patients (63%) reported seasonal variation in migraine attack frequency. Close to half (47%) of patients with aura, but only 17% of patients without aura, reported more frequent attacks during the light season (P < 0.001). Patients with MA reported interictal light hypersensitivity and light exposure as an attack precipitating factor significantly more often than individuals with MoA. They also reported significantly more frequent use of sunglasses to prevent attacks. We found no significant differences between MA and MoA as regards sleep disturbances, use of oral contraceptives, impact of headache or circadian variations. Seasonal periodicity of migraine in an arctic population with more frequent attacks during the light season is a convincing phenomenon in MA but not in MoA. The amount of light exposure seems to be pivotal to this variation.
Introduction
Hypothalamic dysfunction may be present in migraine (1). Specific trigger factors, premonitory symptoms (2), association with sleep (3) and menstruation (4), female preponderance (5), and some observations of circadian periodicity of migraine attacks (6) may be compatible with a disturbance of hypothalamic function in migraineurs. More specifically, Zurak suggested a pivotal role of the suprachiasmatic nucleus (SCN) and melatonin in the pathogenesis of migraine attacks (7).
A link between exposure to light and circadian and seasonal rhythms driven via the hypothalamus is well established (8). No prospective study concerning seasonal variations in migraine has been performed. A retrospective review of 214 admissions due to migraine in South Carolina during a period of 20 years disclosed that most admissions occurred during springtime (21 March to 20 June) for females (9). Fox and Davis retrospectively analysed 3583 well-documented migraine attacks and found support for a circadian pattern with increased susceptibility to attack between 04.00 h and 09.00 h. They also observed a monthly cycle with more migraine attacks on the day of onset of menses. Even if they observed an overrepresentation of attacks during summer months, the difference was not statistically significant (10). Our group has previously shown that migraineurs in an arctic area, compared with patients with other headaches, are more likely to have headache during the bright arctic summer (11). The aim of the present study was to compare self-reported seasonal periodicity in migraine with (MA) and without aura (MoA) and correlate this with light exposure and factors influencing biological rhythms.
Subjects and methods
We performed a questionnaire-based study of volunteer migraineurs in northern Norway during March and April 2004.
Location
North Norway comprises Nordland, Troms and Finnmark counties with 460 000 inhabitants. Almost this entire region is located north of the Arctic Circle, and accordingly variations in light conditions during the year are huge. During the winter there are long periods of low sun and in the period around midwinter the sun never rises above the horizon. Equivalent to this period with polar night (dark season) there are several months with bright daylight (light season), even at midnight in the summer period.
Subjects
Participants were recruited through announcement in several local newspapers and the internet site of Nordlandssykehuset Bodø. The study was promoted as to investigate the ‘role of the midnight sun’ (formulation in the announcement). There was no payment for participation, but 10 received a lithograph by lottery.
Two hundred and nine women who fulfilled the inclusion criteria volunteered and received a comprehensive postal questionnaire. Inclusion required: (i) female sex, (ii) age 16–45 years, (iii) mean one to six migraine attacks per month during the last 3 months before study entry, (iv) migraine with or without aura according to the International Headache Society (IHS) criteria, (v) settlement for at least 1 year in north Norway prior to study entry, and (vi) lack of other serious medical conditions. Patients with probable medication overuse headache and patients who were not able to distinguish between migraine and other types of headache were excluded from the study.
Questionnaire
Patient characteristics
We recorded age, age at headache onset, family history of migraine, education, and social benefit from the National Health Insurance due to migraine and whether the patient had previously consulted a neurologist. We also included HIT-6 (12) to assess the impact of headache.
Comorbidity and use of medication
We asked whether the patient had suffered from stroke, epilepsy, hypertension or ‘other diseases’ which were specified. All types of medication were recorded. Concerning triptans, other migraine medications and general analgesics, patients had to tick off one of the following: ‘daily use’, ‘more than 3 days per week’, ‘1–2 days per week’, ‘less than once per week’ and ‘less than once per second week’.
Sleep
A general complaint of sleep during the last year prior to the study was recorded. Such complaints were further assessed by questions concerning circannual variations and relationship to migraine attacks. Patients who reported reduced ability to fall asleep or to maintain sleep more than three nights a week for more than 1 month, and did not feel refreshed the next day, were considered to have insomnia. To investigate the relationship between migraine and sleep, we asked the patients how often they woke up during the night due to migraine attacks and whether migraine onset occurred in the morning after waking up. Answers to both questions were categorized into ‘every migraine attack’, ‘more than 50% of the migraine attacks’, ‘less than 50% of the migraine attacks’ and ‘infrequently’. We also asked the patients whether their sleep problem varied with season and if the light season was considered worse with respect to this.
Precipitating factors
Patients were asked whether specific factors had a tendency to trigger their migraine attacks and, if so, how often (‘more than 50% of the attacks’, ‘less than 50% of the attacks’ and ‘infrequently’). They were able to choose from a standard list of 20 factors and there was a possibility for free text entry.
Sensitivity to light
We recorded whether the patients used sunglasses to prevent headache (‘always, ‘often’, ‘infrequently’, ‘never’) and whether the patients experienced interictal light hypersensitivity (‘yes’ or ‘no’).
Seasonal variation
We asked the patients whether their headache varied with the season and, if so, which season entailed most suffering. We also assessed which headache characteristics the patients perceived as worse (‘longer-lasting attacks’, ‘more frequent attacks’, ‘stronger attacks’).
Telephone interview
All participants were interviewed by one of the authors (K.B.A., R.S. and S.B.) to ensure the clinical diagnosis applied the IHS criteria (13).
Data analysis
All data were entered onto a computer. Data were analysed with SPSS software (version 12.0 for Windows; SPSS Inc., Chicago, IL, USA). Most of the data were ordinal and categorical. When possible and most reasonable, data were dichotomized. To compare frequencies in the two samples of patients with and without aura, the χ2 test was performed. For ordinal data that were not dichotomized, the samples of MA and MoA were considered independent, and the Mann–Whitney U-test was used. The t-test was used for comparing means. The level of significance was set at 5%.
Ethical approval
The regional ethics committee of northern Norway reviewed the protocol and recommended the study.
Results
One hundred and eighty-two (87%) individuals filled in and returned the questionnaire. Four were excluded since they did not fulfil the IHS criteria for migraine. Five were excluded because they were not able to distinguish migraine from other types of headache, and another four patients were excluded due to possible medication overuse headache. Of the remaining 169 volunteers, 98 had MA (predominantly visual aura in 87%) and 71 had only migraine attacks without aura. Of patients with MA, 78 of 98 (80%) had co-occurrence of MoA, while 20 of 98 (20%) reported that headache was always preceded by aura. Fifteen patients in our sample had occupations where shift work must be regarded as common.
As shown in Tables 1 and 2, the groups compared were similar regarding demographic profile, impact of headache, comorbidity, sleep disorders and use of oral contraceptives. One hundred and seven (63%) of the patients reported seasonal variation in migraine attack frequency. With respect to this, we found a significant difference between MA and MoA patients (P < 0.001). We found no significant difference with respect to circadian periodicity (P = 0.09). Nor was there a significant difference with respect to more frequent attacks during the dark season (P = 0.42). On the other hand, close to half (47%) of MA patients, but only 17% of MoA patients, reported more frequent attacks during the light season (P < 0.001). Table 3 summarizes information regarding migraine periodicity. Patients with MA reported interictal light hypersensitivity and light exposure as an attack precipitating factor significantly more often than individuals with MoA (P = 0.006 and P < 0.001, respectively). They also reported significantly more frequent use of sunglasses to prevent migraine attacks than MoA patients (P = 0.012). Table 4 summarizes information regarding light exposure.
Demographic profile
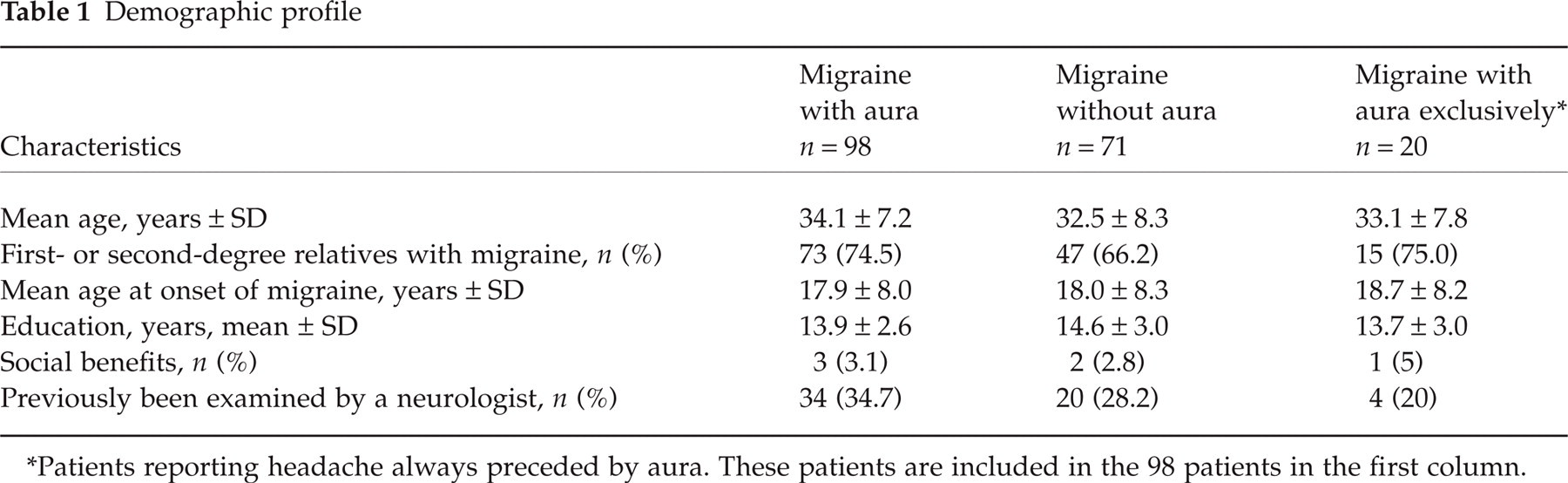
Patients reporting headache always preceded by aura. These patients are included in the 98 patients in the first column.
Comorbidity, users of oral contraceptives, impact of headache and insomnia

Patients reporting headache always preceded by aura. These patients are included in the 98 patients in the first column.
One patient reported polycystic ovarian disease, two reported depression, six reported hypothyroidism, and 16 reported moderate asthma and/or allergy. Other conditions included hypertension, psoriasis, fibromyalgia, and vitiligo.
Percentage of patients reporting periodicity of migraine attacks

Patients reporting headache always preceded by aura. These patients are included in the 98 patients in the first column.
P < 0.001 for the difference between migraine with aura and migraine without aura.
No significant difference.
Interictal light hypersensitivity and light exposure as an attack precipitating factor

Patients reporting headache always preceded by aura. These patients are included in the 98 patients in the first column.
P < 0.001 for the difference between migraine with aura and migraine without aura.
P = 0.012 for the difference between migraine with aura and migraine without aura.
P = 0.006 for the difference between migraine with aura and migraine without aura.
Discussion
About 2/3 of migraineurs in this study reported seasonal variations in migraine and about 1/3 reported more frequent attacks during the light season. Based on a previous study from our group (11), these proportions were somewhat larger than expected. The reason for this is probably the use of questionnaires only allowing yes/no types of answers and the selection of only fertile women with no serious comorbidity and with a limited number of migraine attacks per month. This restriction was made at the expense of study power but it reduced confounding. Another reason for studying only young women is the age-dependent sexual dimorphism of the SCN (7) and the sexual dimorphism of migraine (5). The high number of patients with aura probably reflects a selection bias. Moreover, the patients’ introduction to the study may have influenced answers concerning seasonal variation and light hypersensitivity. Inferences regarding the general population should thus be made with caution.
When MA patients were compared with MoA patients, however, some significant differences, which cannot be attributed to selection bias or over-reporting, were found. More than 75% of MA patients reported seasonal variations in their migraine attack frequency, while this was the case in only 46% of MoA individuals (P < 0.001). More frequent attacks during the light season were reported in 47% of MA but in only 17% of MoA patients (P < 0.001). The frequent co-occurrence of MA and MoA observed in our study (80%) was also observed by Kelman in a study from tertiary care (14). To ensure a correct subgroup classification, we had undertaken a telephone interview to confirm the diagnosis of MA and MoA. As in the study of Kelman, most patients in our study had aura with headache relatively infrequently. Comparing patients who had only MA with patients without aura disclosed an even greater difference. Seasonal periodicity of migraine with more frequent attacks in the light season is thus a convincing phenomenon in MA but not in MoA.
A possible explanation of our findings may be an underlying hyperexcitability of the occipital cortex in migraineurs, suggested by Welch (15) and supported by evidence from Aurora et al. (16) and Young et al. (17). Interictal photophobia is common in migraine (18) and light exposure is a well-known trigger factor for migraine attacks (19). Russell et al. found that bright light is a precipitating factor in MA, but not in MoA (20). In our study, more than half of all patients reported interictal light hypersensitivity and 3/4 of patients reported strong light as a precipitating factor for migraine attacks, and the differences between MA and MoA in these respects were highly significant (Table 4). These observations may also indicate that the theory of ‘central neuronal hyperexcitability’ (15) is limited to MA. However, this opinion is not supported in a study of Mulleners et al. (21) and interictal hyperexcitability is probably not limited to the occipital cortex (22).
Seasonal and circadian fluctuations in biological rhythms are generated in the SCN. Variations in light exposure and in the length of the night probably trigger these fluctuations (8). The possible role of the SCN in the menstrual cycle and weekly rhythms is less clear (23). Key hormone of this system is melatonin, an indole compound derived from serotonin. Secretion of melatonin by the pineal gland is rhythmic and occurs almost exclusively during the night. It is secreted for longer periods, and sometimes in higher concentrations, during winter than during summer (8). Observed circardian and seasonal periodicity in migraine may support an involvement of the biological rhythms in the pathogenesis of migraine. However, many factors such as ageing, serious disease, use of contraceptives, sleep disorders, and round-the-clock work, influence our biological clocks (23), and are potentially confounders of such observations. The MA and MoA groups in the present study were similar concerning these factors. Patients who reported headache exclusively preceded by aura reported more insomnia and more frequent attacks during the early morning hours than the other patients, but this not significant compared with the non-aura group (P = 0.07 and 0.15, respectively). Overall, 1/3 of the patients reported attack onset primarily during the early morning hours, but this applied to both MA and MoA. There were no differences in susceptibility to attacks in the dark season.
Claustrat et al. studied the sensitivity to light of melatonin secretion in 12 female patients with migraine (eight MA and four MoA) compared with 12 female controls (24). They found that the migraineurs were supersensitive to light and concluded that this is in agreement with the concept of central neuronal excitability in migraine. There were no observed differences between patients with and without aura, but they emphasize that the sample was small and that this needs further investigation. In our study, the distinction of MA as regards ‘over-occurrence’ of attacks during the summer season is probably not related to biological rhythms. A prospective study in the same arctic area with female patients recording every migraine attack during a whole year should assess this and the periodicity of migraine further.
In conclusion, increased susceptibility to migraine attacks during the arctic summer season in female migrainurs is a feature of MA. Variations in light exposure, and not in biological rhythms per se, seem to be the key to this variation.
