Abstract
Recent studies of the visual cortex in patients with migraine have generally concluded that migraine (particularly migraine with aura) is associated with a state of functional cortical hyperexcitability. The mechanisms giving rise to this hyperexcitability have hitherto been unclear. This paper reports two studies that used a novel investigative technique, derived from basic research in vision science, to examine specific deficits of inhibitory processing in primary visual cortex. The technique is termed the metacontrast test, and it examines visual masking under highly specified conditions. In Study 1, 12 migraine with aura patients (MA), 12 age-matched migraine without aura patients (MO) and 12 age- and sex-matched headache-free control subjects (C) were compared using the metacontrast test. MA patients were significantly less susceptible to visual masking in the metacontrast test than both MO and C groups: this result is highly consistent with a deficit in cortical inhibitory processing in MA patients. Study 2 examined MA patients taking a variety of migraine prophylactics, again using the metacontrast test. Test results normalized in those MA patients taking sodium valproate, but not in those taking other prophylactics. Sodium valproate is a GABA-A agonist that is known to cross the blood–brain barrier: GABA-ergic networks act as the primary inhibitory mechanism in visual cortex. Taken together, the results of these studies argue that cortical hyperexcitability, at least in MA patients, is likely to be a result of deficient intracortical inhibitory processes.
Introduction
Recent interest in brain mechanisms of the early stages of a migraine attack has focused on the complex relationship between the inter-ictal status of cortical neural networks, the physiological events of cortical spreading depression (CSD), and the phenomenology of the visual aura. Since the time of Lashley (1), a compelling hypothesis has been that CSD is responsible for the visual hallucinations of classical visual aura (2). Although there is no direct evidence for the existence of CSD in human cortex, very recent investigations using neuroimaging techniques have tended to confirm a spreading neurovascular disturbance originating in the visual cortex during aura (3). It has been postulated that the headache phase of migraine may follow the aura via activation of a trigeminovascular response which has been demonstrated to occur after induced CSD in an animal model (4) (although this model has been criticised on technical grounds (5)).
What is currently a major topic of interest is why the cortex, especially the visual cortex, in migraine patients may be particularly susceptible to the initiation of CSD-like episodes. A number of research groups have successfully used the method of transcranial magnetic stimulation (TMS) to demonstrate that the visual cortex of migraine patients is functionally hyperexcitable. Aurora et al. (6) demonstrated clearly that the threshold level of magnetic stimulus required to evoke reports of phosphenes in subjects stimulated over the occiput was significantly lower in patients than in controls. This finding has been replicated by other investigators (7), although it must be pointed out that one group found evidence for decreased excitability in patients (8). Overall however, TMS evidence of increased excitability to stimulation is highly consistent with the notion that the cortex of migraine patients may be susceptible to episodes of CSD.
TMS does not at present allow detailed mapping of particular neural systems, and it is therefore not clear how the excitability to magnetic stimulation occurs in patients. Given the morphology of the primary visual cortex (V1), it is plausible to argue that there are (at least) two routes to functional hyperexcitability: (i) over-excitation in excitatory systems, (ii) under-inhibition in inhibitory systems. We have argued on a number of occasions that there are good reasons to implicate inhibitory systems in cortical dysfunction in migraine (9, 10). Furthermore, it is notable that CSD is more likely in under-inhibited cortex (11) and may indeed be triggered by trivial environmental changes in appropriate models (12). Circumstantial evidence also comes from the success of centrally acting GABA-agonists as migraine prophylactics: sodium valproate crosses the blood–brain barrier (13), reduces cortical excitability at least in patients with epilepsy (14) and has been demonstrated to reduce significantly the frequency of migraine attacks in double-blind trials (15, 16). However, to date there is no direct evidence to support the notion that inhibitory systems in V1 of migraine patients are impaired, although findings relating to the functional status of V1 in migraine are consistent with it (10, 17). We therefore investigated patients with migraine using a functional psychophysical test that has recently been shown to depend crucially upon inhibitory interactions at a cortical level. We next describe this task.
Metacontrast masking is a type of visual masking that occurs when judgements about a target are impaired because of a subsequently presented, spatially non-overlapping mask. For example, a briefly (approx. 20 ms) presented capital letter of normal size (the target), followed at an interval of 75 ms by a ring shape (the mask) surrounding the letter but not overlapping it in either space or time, will be invisible to observers. However, the same letter presented in the absence of the mask is plainly visible. The masking effect is strongest when the time interval between the presentation of the target and the mask is between 50 and 100 ms (18). This results in a characteristic U-shaped masking function, where there is little or no masking when the target and the mask are presented simultaneously, maximal masking at a 50–100 ms time interval, and a return to little or no masking with time intervals above about 150 ms. Macknik & Livingstone (19) investigated the responses of V1 cells in monkey during the presentation of visual stimuli that give rise to metacontrast masking in humans. Macknik & Livingstone demonstrated, using single and multi-unit recording from V1 neurones, that the metacontrast phenomenon was specifically associated with inhibition of the transient after-discharge to the target stimulus. In essence, it appears to be the case that the cells responsible for processing features of the mask send inhibitory messages to the cells responsible for the target, before sufficient time has elapsed for processing of the target to be complete, and for it thus to be discriminable. As inhibition between adjacent columns is a crucial feature of the stimulus-processing capability of V1, and a primary reason for the preponderance of GABA-ergic cells in this cortical area (20), it is reasonable to assume that the metacontrast function provides an index of its overall inhibitory potential.
We therefore predicted (Study 1) that migraine patients with aura should be less susceptible to metacontrast masking if the overall inhibitory function of V1 was compromised because of their condition, and therefore less inhibition of transient after-discharge would occur. Furthermore, we predicted (Study 2) that in migraine patients being treated with prophylactic agents likely to increase inhibitory potential in cortical areas, metacontrast curves would show equivalence with those of migraine-free controls.
Study 1
Patients and controls
Twelve migraine with visual aura patients (nine female, three male), 12 age-matched migraine without aura patients (10 female, two male) and 12 age- andsex-matched headache-free controls participated in the study (Table 1). Participants were not matched for handedness. The migraine patients were recruited from the local area and were diagnosed according to the diagnostic criteria of the International Headache Society (IHS) (21). All but one of the migraine with aura patients reported that their attacks were always preceded by visual aura. The other migraine with aura patient had recently started waking up with a migraine headache, although she still experienced aura before the majority of attacks (approx. 90%). The migraine without aura patients all reported exclusively attacks without aura. All participants had normal colour vision, normal or corrected to normal visual acuity and normal spatial contrast sensitivity. None of the participants who were included in this study was taking prophylactic medication for migraine, either at the time of testing or for at least 1 month before. They took only acute medication to control attacks as necessary. All patients were tested inter-ictally, at least 24 h since the last attack had cleared and 48 h before the start of the next one. Ethical approval for Study 1 was obtained from the Department of Psychology, Lancaster University. Written informed consent was obtained from all patients prior to testing.
Age, age of onset and migraine chronicity for the three experimental groups

Methods
Stimuli and apparatus
The stimuli consisted of an inner target disk and an outer ring-shaped mask. The stimuli were black (mean luminance 1.066 cd/m2) presented against a grey background (mean luminance 107.7 cd/m2, contrast 98%) on a 75-Hz monitor. There were two types of target stimuli, a complete disk (whole target) or one with the right side cut off (cut target). Examples of the stimuli are shown in Fig. 1 along with sizes and visual angles. A viewing distance of 50 cm was maintained with a chin rest. Throughout the experiment participants were asked to focus on a small fixation point, a black square 1 × 1 mm subtending 0.11° of visual angle, which appeared in the centre of the screen. The target and the mask stimuli then appeared either above or below the fixation point with the outside edge of the mask appearing 10 mm (1.15°) from the centre of the fixation point.

(a) Stimuli flow chart for the experiment: each trial consisted of the sequence of events in either the top row or the bottom row (selection was quasi-random). Panel 1 shows the ‘whole’ target (top) and ‘cut’ target (bottom). Panel 2 shows the four stimulus onset asynchronies (SOAs) employed (SOA was measured from the onset of the target to the onset of the mask). Panel 3 shows the masking stimulus, which was identical for both target types and whose inner edges did not overlap any contours of the target. Panel 4 gives the response required of the subject. At all values of SOA, target and mask were each displayed for 13 ms. (b) Sizes and visual angles of target and mask.
Procedure
Subjects were required to decide whether or not the target circle was cut, pressing a key on the keyboard if they thought it was cut and remaining passive if the circle was whole. The instructions emphasized that the task was like a visual illusion, in that the presence of outer mask would make it difficult and sometimes impossible for them to perceive the target circle clearly, but that they were to try and complete the task as accurately as possible. Each trial started with the fixation point in the centre of the screen for 1000 ms, and then the target and the mask appeared for 13 ms each. After the offset of the mask, the fixation point remained in the centre of the screen for a further 1500 ms. During this time the participants responded by pressing a key if they thought the circle had been cut. The target and the mask appeared either simultaneously or with a time interval between the onset of the target and the onset of the mask (stimulus onset asynchrony (SOA)) of 39, 78 or 156 ms. Overall, subjects completed 720 trials, 180 trials for each SOA (60 practice, 120 experimental). The total number of trials was divided equally between three blocks to help reduce any possible effects due to fatigue. The order of trials was randomized over blocks and subjects.
Results
Following an appropriate statistical transformation, mean proportions correct were submitted to a group (migraine with aura, migraine without aura, control) × SOA (0, 39, 78, 156) analysis of variance with group as the only between factor. This analysis included the data from the two experimental blocks only (data from practice trials were discarded). Mean proportions correct are shown in Fig. 2.

(a) Overall proportion of correct target judgements in the metacontrast test, separated by diagnostic group. In this and all subsequent fiaures, error bars show
There was a main effect of group, F(2, 33) = 3.48, P < 0.05; the migraine with aura patients made more correct responses than either the migraine without aura patients or the headache-free control subjects. This effect is also shown in Fig. 2. Planned comparisons were conducted to investigate this group difference further. The mean performance of the migraine with aura patients was significantly better than the mean performance of the migraine without aura patients and the controls (F(1, 33) = 6.379, P < 0.05). The accuracy of the migraine without aura patients and the accuracy of the control subjects was not significantly different (F(1, 33) = 0.577, P = 0.45). There was also a main effect of SOA (F(3, 99) = 105.33, P < 0.01) resulting in a U-shaped masking function overall, as expected. The interaction between group and SOA was not significant.
A secondary analysis of the data examined the relationship between task performance and frequency of migraine attacks. A standard regression of frequency of migraine on overall target judgement accuracy was performed for each patient group separately. Figure 3 shows the scatterplots and regression lines. For the migraine without aura group, there was no significant effect of attack frequency on accuracy. For the migraine with aura group, overall accuracy increasedwith frequency of migraine (r 2 = 3.73, F(1, 10) = 5.937, P < 0.05).
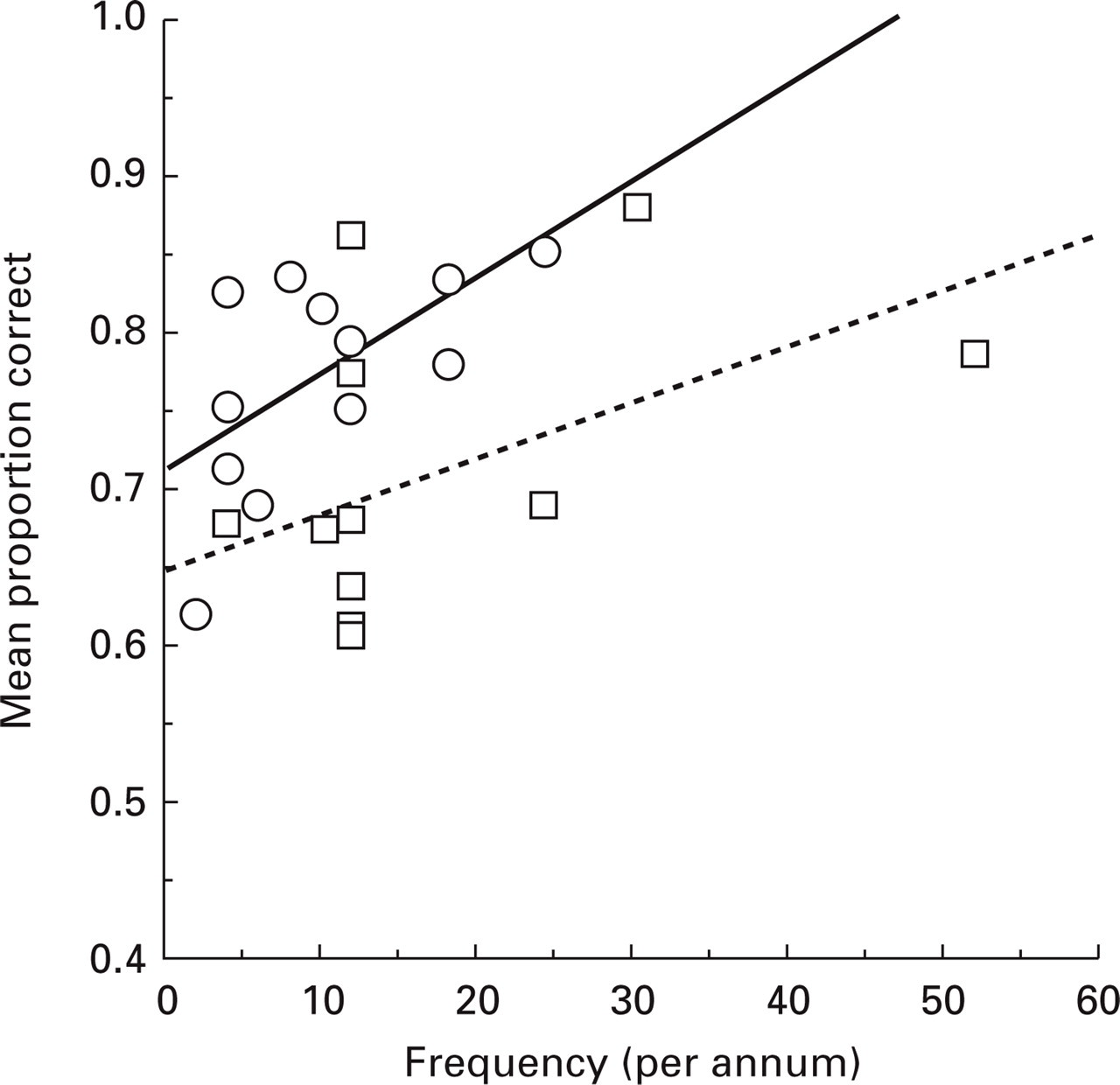
Overall proportion of correct target judgements (pooled over stimulus onset asynchronies (SOA)) as a function of frequency of migraine (self-reported number of attacks per annum). The solid regression line is fitted through the points from migraine with aura patients; the dotted regression line through the points from migraine without aura patients. ○, MA; □, MO.
Discussion
The main effect of diagnostic group (Fig. 2) demonstrates that migraine with aura patients are better able to make target judgements than normal control subjects. This is a powerful dissociation, as a performance enhancement is unlikely to be explainable in terms of non-specific factors such as the experiment's demand characteristics. It is supportive of impaired inhibitory function in V1 of these patients: reduced inhibition of transient channel after-discharge (19) is likely to reduce the metacontrast masking phenomenon. The effect of attack frequency on target judgement accuracy in migraine with aura patients possibly reflects a differential loss of inhibitory function in patients with varying headache frequency: greater levels of inhibitory impairment render the cortex more likely to support an episode of a CSD-like event and hence headache frequency is increased. We comment further on this issue below.
As the major inhibitory neurotransmitter in visual cortex is GABA, and neurotoxins for cortical GABA-ergic inhibitory systems render visual cortex highly susceptible to spreading depression (22), Study 2 examined responses to the metacontrast test in a small number of patients who were established on sodium valproate for the prophylaxis of migraine. These patients were compared with others taking prophylactics with no known action on cortical GABA-ergic systems, and the drug-free patients tested in Study 1. It was predicted (i) that the metacontrast results of patients taking valproate would be similar to normal subjects, and (ii) that the results of patients taking other prophylactics would be similar to drug-free patients.
Study 2
Participants
Six migraine with visual aura patients and one migraine aura without headache patient, who were taking prophylactic medication for migraine at the time of testing, were recruited from the Head Pain Clinics at Manchester Royal Infirmary and the Royal Preston Hospital. All patients were diagnosed according to the diagnostic criteria of the IHS (21). Clinical details are shown in Table 2.
Clinical characteristics of patients in Study 2

?, Patient unsure of answer;
Patient who is colour blind. Headache and aura frequency are both given currently, since the patient started taking prophylactic medication, and previously, prior to the commencement of current prophylactic treatment. Aura frequency is a percentage of attacks on which visual aura is present.
The one patient with migraine aura without headache had previously had a headache phase following his aura. Three of the patients were taking sodium valproate, two were taking propanolol, one was taking pizotifen and the final patient was taking methysergide. As it was the effect of taking sodium valproate on metacontrast masking that was of particular interest, and as there was a range of different prophylactic medications being taken by the patients, two experimental groups were formed. The first group included only those patients taking sodium valproate (Valproate), whilst the second included the patients on any other prophylactic medication which is not known to affect GABA levels (Other). The results of these two groups were then compared with the 12 drug-free migraine with visual aura patients who were tested in Study 1.
All of the patients had normal or corrected to normal visual acuity. Colour vision was normal in all of the patients, except the patient who was taking pizotifen, whose scores were consistent with deuteranopia. Participants were not matched for handedness. Ethical approval for Study 2 was obtained from the Department of Psychology, Lancaster University, the Royal Preston Hospital Ethical Committee and the Central Manchester Health Care Trust Ethical Committee. Written informed consent was obtained from all patients prior to testing.
Method
The study used the same metacontrast masking test described in Study 1.
Results
Following an appropriate statistical transformation, mean proportions of correct responses were submitted to a medication (valproate, other prophylactic, none) × SOA (0, 39, 78, 156) analysis of variance with medication as the only between factor. There was a main effect of medication, F(2, 16) = 5.36, P < 0.05; the patients taking sodium valproate made more errors than either of the other two groups (Fig. 4).

(a) Overall proportion of correct target judgements, separated by medication type. ‘Acute’ refers to those patients taking only acute treatment for attacks, and are the same data as reported in Study 1. ‘Valproate’ refers to patients taking sodium valproate for prophylaxis of migraine; ‘Other’ refers to patients taking other prophylactics. (b) Proportion of correct target judgements plotted on a stimulus onset asynchrony scale and separated by medication type. The control group data from Study 1 are included in the plot for ease of comparison. •, Acute; □, valproate; Δ, other; ○, controls; ·· · · ·, chance.
Planned comparisons were conducted to investigate this group difference further. The mean performance of the migraine with aura patients taking sodium valproate was significantly less accurate than the mean performance of the migraine without aura patients who were taking either no prophylactic or ones which are not known to affect GABA (propanolol, pizotifen, methysergide) (F(1, 16) = 8.43, P < 0.05). The accuracy of the two migraine with aura patient groups not taking sodium valproate was not significantly different (F (1, 16) = 0.681, P = 0.43). There was also a main effect of SOA (F(3, 48) = 22.06, P < 0.01) with the results once again showing the characteristic effect of SOA in producing a U-shaped function.
Discussion
Inspection of Fig. 4 reveals clearly that the patients taking propanolol, pizotifen and methysergide performed similarly to drug-free patients on the metacontrast test: their overall performance was statistically indistinguishable. However, patients taking valproate were significantly less accurate overall: curves were if anything shallower than those of normal subjects. This pattern of data is entirely consistent with the predictions that stem from the known activity of sodium valproate on cortical GABA-ergic inhibitory systems. All three patients taking sodium valproate reported good therapeutic success with valproate: it is plausible to argue that this is perhaps because of restoration of normal cortical inhibitory potential in these patients. It is evident however, that sodium valproate has a rather diverse pharmacological profile of activity, and its efficacy as a prophylactic for migraine may result from ‘a combination of actions at different loci’ (13, 23). It remains possible, though in our opinion unlikely, that a valproate-mediated increase in brain GABA is uncorrelated with therapeutic outcome in migraine.
General discussion
In this study, a novel functional test—the metacontrast test—of the functioning of V1 has demonstrated a likely reduction of cortical inhibitory potential in migraine patients with aura. The relationship between attack frequency and results of the metacontrast test also suggests that the greater the impairment of inhibitory function, the more likely are attacks. Furthermore, the normalization of metacontrast test results in a small number of patients taking a known cortical GABA-agonist for the prophylaxis of migraine, but not in patients taking prophylactics with no known GABA-agonist activity, serves as a preliminary confirmation of the importance of this neurotransmitter system in the genesis of pathological cortical excitability in these patients. The data reported here are highly consistent with findings that cortical tissue that is deficient in inhibitory potential is susceptible to CSD (12)
A number of issues stem from these data. First, what might be the cause of inhibitory loss in V1 of migraine patients with aura? The authors (E.P.C. & W.M.M.) have previously argued that the cumulative physiological effects of many episodes of aura may cause ultrastructural changes to the selectively vulnerable GABA-ergic systems of V1 (9). Although there is no direct evidence as yet to support this view, it is interesting to note that, in animal models, repeated episodes of CSD have been shown to cause selective suppression of GABA-ergic inhibitory function (24). At present however, the notion of an intrinsic cortical hyperexcitability in migraine is equally attractive and plausible. The data we report here suggest that, if this second notion is correct, the functional hyperexcitability of V1 may still stem from an intrinsic disorder of inhibitory systems.
Second, it is apparent that the metacontrast test results are only significantly different from control subjects in patients with aura. Study 1 patients with aura were selected on the basis of having aura before all, or the vast majority of attacks, whereas patients without aura had never experienced a classical visual aura. It is possible therefore that cortical hyperexcitability is a matter of degree even within the migraine population, and is maximized in those patients who frequently have aura. This suggestion is bolstered by the finding of Study 1 of a significant correlation between attack frequency and overall probability of correct judgements in the metacontrast test. It will be of great interest in future to examine the interrelationships between the overall frequency of attacks, the ratio of attacks with to attacks without aura and metacontrast test results.
Study 2 used very small numbers of patients, and it may be argued that the results are not representative. Against this argument are the two points: (i) that the metacontrast testing procedure is effectively blinded: the testing is under computer control and neither patient nor experimenter knows what type of target will next appear; and (ii) a very large number (480) of data points are gained from each patient, and so the potential for error is considerably reduced compared with, say, the provision on a rating scale of a single headache intensity score. Nonetheless, it is clear that intervention studies will be required in order to examine the changes in metacontrast test results prior to and during therapy with valproate: such studies are currently underway in our laboratory.
Previous studies have proposed that migraine with aura is associated with cortical hyperexcitability, and have found evidence for disordered visual cortical processing. The threshold for the generation of phosphenes by transcranial magnetic stimulation of the occipital cortex is probably lower in migraine with aura patients than headache-free control subjects (6, 7). Migraine patients—in particular those with aura—also exhibit a variety of deficits on other visual tasks that is consistent with functional cortical hyperexcitability (10). The results of the present study extend and give further support to these findings, by providing evidence that specifically inhibitory mechanisms are disrupted in the visual cortex of migraine with aura patients. Furthermore, they suggest that the disruption of inhibition may be reversed pharmacologically: this may have implications for the future development of prophylactic agents.
Footnotes
Acknowledgements
The work reported here was supported by Project Grant 115 from The Migraine Trust and an ESRC Research Studentship awarded to J.E.P.
