Abstract
The present study was aimed at verifying the clinical characteristics of a typical attack in 20 migraine patients, 10 responders and 10 non-responders to rizatriptan, and at investigating any differences in the levels of neuropeptides of the trigeminovascular or parasympathetic systems [calcitonin gene-related peptide (CGRP), neurokinin A (NKA) and vasoactive intestinal peptide (VIP) measured by radio-immunoassay methods in external jugular blood] between responders and nonresponders. In all responders to rizatriptan, pain was unilateral, severe, and pulsating, and in five of them at least one sign suggestive of parasympathetic system activation was recorded. Five patients who were non-responders to rizatriptan referred bilateral and non-pulsating pain, even though severe in most of them. CGRP and NKA levels measured before rizatriptan administration were significantly higher in responders than in non-responders (P < 0.0001 and P < 0.002, respectively). In the five patients with autonomic signs among rizatriptan responders, detectable VIP levels were found at baseline. One hour after rizatriptan administration, a decrease in CGRP and NKA levels was evident in the external jugular venous blood of rizatriptan responders, and this corresponded to a significant pain relief and alleviation of accompanying symptoms. VIP levels were also significantly reduced at the same time in the five patients with autonomic signs. After rizatriptan administration, CGRP and NKA levels in non-responder patients showed less significant variations at all time points after rizatriptan administration compared with rizatriptan responders. The present study, although carried out on a limited number of patients, supports recent clinical evidence of increased trigeminal activation associated with a better triptan response in migraine patients accompanied by parasympathetic activation in a subgroup of patients with autonomic signs. In contrast, the poor response seems to be correlated with a lesser degree of trigeminal activation, lower variations of trigeminal neuropeptides after triptan administration, and no evidence of parasympathetic activation at baseline.
Introduction
Although triptans represent first-choice drugs for migraine attacks, the percentage of non-responders in clinical trials ranges from 25 to 35% (1). Furthermore, even in the case of migraine patients considered responders to a particular triptan, analyses of within-patient consistency have demonstrated that less than 2/3 of patients respond to the same triptan in 3/3 attacks (2–4). The lack of a response to triptan treatment for migraine crises has been attributed to a variety of factors, including low and inconsistent absorption of oral formulation, unrecognized analgesic overuse, inadequate dosing, and variability in individual response based on 5-HT1 receptor polymorphisms. Also to be considered is the possibility that the attack treated is not a migraine attack but an attack of tension-type headache, which coexists with migraine in many patients and is unlikely to respond to triptan treatment (5). Moreover, it has been emphasized that in some patients the poor response to triptan treatment may actually be the result of treatment too late in the progression of migraine attack when head pain is of moderate or severe intensity or cutaneous allodynia is already established (6–8). The need for early treatment has been affirmed based on several clinical trials for reducing the unsatisfactory response to a triptan and achieving maximum efficacy in patients with an incomplete response. This approach would reduce the number of true triptan non-responder patients (9–13).
In clinical practice, when the diagnosis of migraine is confirmed in at least three attacks, the poor response to one particular triptan does not exclude the possibility of successfully using another triptan. Recent open studies have in fact demonstrated, in the case of a poor response to a triptan, the efficacy of other members of the triptan family on the same patients (14–16). This implies that patients with a poor response to a triptan can be successfully switched to another triptan and some practical, useful clinical tools have been developed in this regard, based on questions which explore the most relevant expectation of patients with acute treatment (17, 18).
To date, no studies are available that have either focused on the clinical characteristics of true non-responder migraineurs to a particular triptan or investigated the particular biochemical characteristics correlated with trigeminovascular activation. Recent evidence suggests that unilateral cranial autonomic symptoms, such as lacrimation, conjunctival injection, eyelid oedema and nasal congestion may occur in a discrete proportion of migraine patients during attacks. Their presence supports an activation of the trigemino-autonomic reflex, probably related to over-activity of trigeminovascular afferents (19).
This observation has been hypothesized to have therapeutic implications, based on the observation that the presence of unilateral cranial autonomic symptoms consequent to activation of the trigeminal autonomic reflex in such patients may be predictive of a positive response to the 5-HT1B/1D receptor agonist sumatriptan (20). The present study was aimed at investigating the clinical and biochemical correlates of a satisfactory and poor response to a new generation triptan, rizatriptan, with particular regard to trigemino-parasympathetic activation during a migraine attack.
Patients and methods
Patients
The protocol was approved by the Ethics Committees of the three Headache Centres (Perugia, Padua and Modena) in which the patients were enrolled. After written informed consent, 20 patients with a history of migraine without aura (MoA) for at least 2 years were enrolled from 200 consecutive triptan-naive MoA patients, who attended the three Headache Centres in the period January 2004 to November 2004. These patients had used only simple or combination analgesics or non-steroidal anti-inflammatory drugs (NSAIDs) for treating their migraine attacks: 80 of them judged the response to analgesics satisfactory, while the remainder judged it unsatisfactory. In the patients screened, the diagnosis was confirmed using a computerized record for verifying application of the International Classification of Headache Disorders-II criteria for primary headaches (21).
The number of attacks/month ranged from two to six in the last 6 months, and the duration of attacks from 10 to 48 h. Thirteen of these patients had a family history of migraine. Exclusion criteria for patients joining the study were: smoking habit, migraine duration <2 h, and systemic or concomitant pathologies (renal and cardiovascular diseases, including hypertension and epilepsy). Before inclusion in the study, all patients had been taking simple analgesics for symptomatic treatment of their usual attacks with satisfactory relief of headache.
Patients were selected on the basis of a satisfactory or poor response to rizatriptan in non-responders or responders [nine in the Headache Centre of Perugia (four responders and five non-responders), six in the Headache Centre of Modena (three responders and three non-responders), and five in the Headache Centre of Padua (three responders and two non-responders)]. Inefficacy or efficacy of rizatriptan was verified in the treatment of at least three consecutive migraine attacks.
Clinical assessment
To verify the response to rizatriptan, the 200 initially screened patients were allowed to keep a headache diary in three consecutive attacks. They were asked to record the time of headache onset and when rizatriptan was taken, the intensity of headache, the presence of accompanying symptoms (nausea, vomiting, phono- and photophobia), functional disability, and also the presence of autonomic signs and symptoms (eyelid oedema, lacrimation, conjunctival injection, nasal congestion and rhinorrhoea).
All parameters were recorded at defined time points: at 1, 2, 4, 6, and 12 h post dosing. The patients should also have recorded the time of headache cessation as well as headache recurrence within 24 h and the use of rescue medication, a NSAID, which was allowed after the first 12 h of recording headache characteristics. The time chosen for rescue medication was late compared with other migraine studies; however, we followed the course of typical attacks of patients without medication other than rizatriptan in the first 12 h. All patients were informed of our purpose and, as was specified in their informed consent, they accepted to wait for a rescue medication at this set time.
On the basis of the clinical assessment in six consecutive attacks (three were treated when pain was mild at baseline at the time of rizatriptan administration, and three were treated when pain was moderate or severe at baseline), 10 patients were identified as satisfactory responders to rizatriptan and 10 patients as poor responders to rizatriptan. A satisfactory response to rizatriptan was defined as a reduction in pain from severe or moderate to mild or absent within 2 h after rizatriptan administration without a recurrence in the next 48 h. In contrast, an unsatisfactory response was defined as a non-significant reduction in pain intensity within 2 h (as defined above) or, in the case of a significant reduction, a recurrence of pain within 48 h after administration. In all six attacks examined, non-responders to rizatriptan showed no significant reduction in pain intensity within 24 h after rizatriptan administration.
After having confirmed a satisfactory or poor response to rizatriptan in six consecutive attacks, all patients were invited to reach their corresponding Headache Centre within 2 h of onset of another attack. The attacks resembled those typically experienced by our study patients. Before taking any symptomatic medication, they underwent clinical assessment of the attack characteristics, presence of autonomic symptoms, and their response to 10 mg rizatriptan benzoate (RPD formulation) every hour for the following 12 h as previously reported. The clinicians examining autonomic activation were blind to the biochemical data and response to rizatriptan.
Jugular venous blood procedure during a typical migraine without aura attack
Immediately after being admitted to our Headache Centre, patients underwent catheterization of the external jugular vein. Access to the right or left internal jugular vein was subsequently performed (ipsilaterally to head pain; in eight patients the right jugular vein, in the remaining patients the left jugular vein). Details are reported in previous papers concerning the internal jugular venous procedure (6, 7). Jugular venous blood samples were drawn immediately after catheter positioning, and at 1, 2, 4, 6 and 12 h after triptan administration. For ethical reasons, it was not possible to take additional jugular venous blood samples after 12 h of sampling, in particular, when the patients were without pain.
Determination of sensory neuropeptides and nitrites
Plasma samples were stored at −70°C until determination. The trigeminal markers calcitonin gene-related peptide (CGRP) and neurokinin A (NKA), and the parasympathetic marker vasoactive intestinal peptide (VIP) were determined by the radioimmunoassay (RIA) method (Peninsula Laboratories, Belmont, CA, USA for CGRP and NKA; and Phoenix Laboratories, Karlsruhe, Germany for VIP).
CGRP, NKA and VIP immunoreactivities were eluted with 60% acetonitrile in 0.1% trifluoroacetic acid in SEP-C18 columns activated with 0.1% trifluoroacetic acid and 60% acetonitrile in 0.1% trifluoroacetic acid. Eluates were associated with a centrifuge concentrator (Supervap PL-CC-180). Residues were dissolved in buffer and determined with RIA kits. Data were expressed as pmol/l for neuropeptides. Standards for the above substances were dissolved in 0.1 mol/l phosphate buffer, pH 7.5, containing 0.1% bovine albumin, 0.01% sodium azide, and 500 kIU/ml kallikrein. Details of the methods for CGRP and NKA are reported in previous papers of our group (22, 23).
VIP human antiserum showed a weak cross-reactivity for human PACAP-27-NH2 (2%), PACAP-28 (0.4%), and PHM-27 (0.9%) but not for substance P, endothelin-1, glucagon, galanin, and somatostatin. The linearity range of the kit was 1–128 pg/tube, the sensitivity IC50 approximately 7 pg/tube. The detection limit of the assay was <4.3 pg/tube. The intra- and interassay variabilities were 3 and 5%, respectively.
Statistical analysis
Pain characteristics, associated symptoms and autonomic signs were recorded at each time point and results were expressed as number and percentage of patients. The data concerning trigeminal and parasympathetic neuropeptides at each time point were expressed as mean ± SEM and compared using
Results
In all patients with a satisfactory response to rizatriptan, the head pain at baseline was unilateral, pulsating, and of severe intensity, whereas of patients who were non-responders to rizatriptan, only half had head pain with a unilateral location, although in the majority it was of severe or moderate intensity. Pearson's χ2 test showed a significant difference in pulsating pain quality and unilateral location between responder and non-responder patients to rizatriptan (P = 0.05 and P = 0.009, respectively). No difference at baseline emerged in the proportion of patients with aggravation with routine physical activity between responders and non-responders to rizatriptan (Table 1).
Pain characteristics (unilaterality, pain quality, pain intensity, aggravation with routine physical activity) at different time points in migraine patient responders and non-responders to rizatriptan

The percentages can be calculated by multiplying each value by 10.
Within 2 h of rizatriptan administration, the 10 patients considered responders to rizatriptan, on the basis of the assessment of six previous consecutive attacks, had a significant response to rizatriptan. This can be verified in Table 1, which shows only two patients ‘responders’ with mild pain not limiting routine physical activity at 2 h. In none of the responder patients was headache recurrence recorded within 48 h.
Patients believed to be non-responders to rizatriptan continued to complain of pain of severe or moderate intensity at 2, 4, 6 and 12 h after rizatriptan administration. Rescue medication was administered to all of them 12 h post dose. Seven of these patients were pain free at 24 h, whereas three continued to refer pain of mild intensity at this time.
No difference emerged in the proportion of associated symptoms in migraine patients who were responders and non-responders to rizatriptan at baseline. Within 2 h after rizatriptan administration, only two of 10 patient responders had photophobia and/or phonophobia. Non-responder patients continued to experience at least two associated symptoms until 12 h before rescue medication was administered (Table 2).
Number of patient responders and non-responders to rizatriptan experiencing migraine-associated symptoms and with autonomic signs at each time point of the study
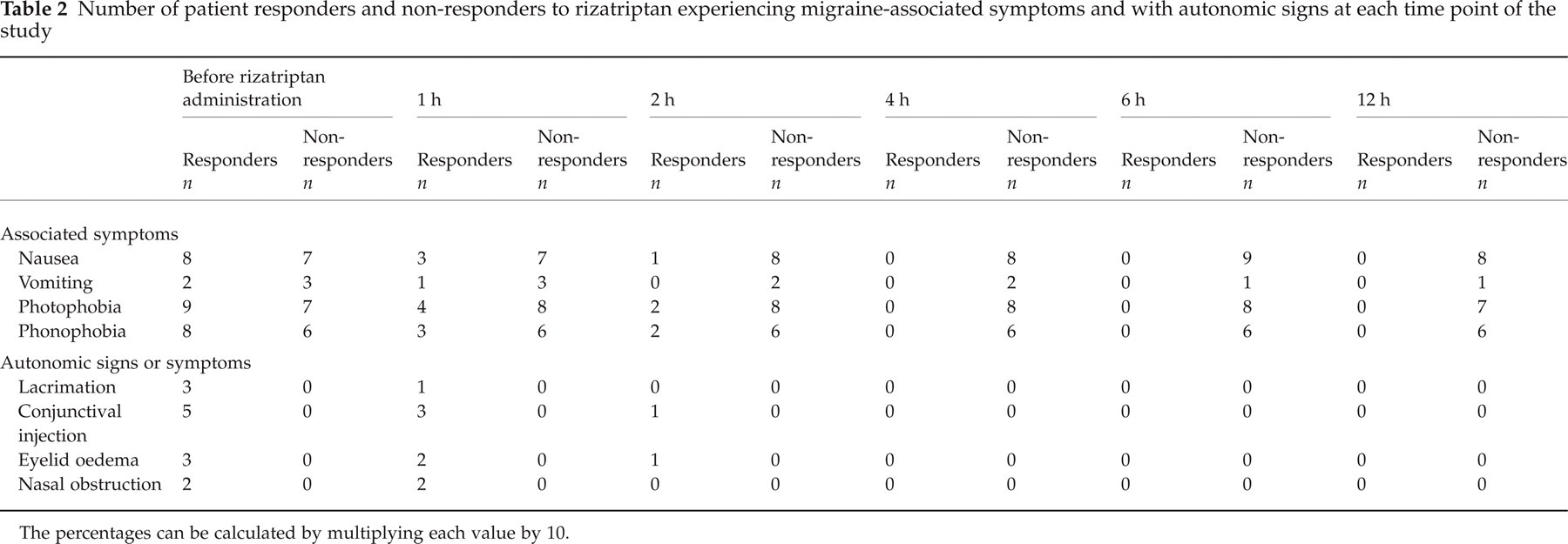
The percentages can be calculated by multiplying each value by 10.
In five patient responders to rizatriptan, at least one unilateral sign indicative of parasympathetic activation (lacrimation, conjunctival injection, eyelid oedema and nasal congestion) was found at baseline. They tended to disappear within 2 h after rizatriptan administration, with the exception of one patient with mild pain at 2 h who had residual autonomic signs and symptoms indicative of parasympathetic activation, which were never identified in patients who were non-responders to rizatriptan at any time point of the study (Table 2).
Pearson's χ2 test showed a statistical significance of 0.009 in the proportion of patients with and without conjunctival injection among responder and non-responder patients, whereas the difference for the occurrence of lacrimation, eyelid oedema and nasal congestion did not reach the minimum level of significance (P < 0.06 for all).
Patient non-responders to rizatriptan showed significantly lower levels of CGRP and NKA compared with responders at baseline (P < 0.0001 and P < 0.002). In these patients, the levels of the two trigeminal peptides showed no significant variations following rizatriptan administration, which was instead evident in patient responders to rizatriptan within the first 2 h. In contrast, patient responders to rizatriptan with signs of parasympathetic activation showed detectable levels of VIP before rizatriptan administration. These levels tended to decrease significantly in the first 2 h post dose (Table 3).
Levels of neuropeptides calcitonin gene-related peptide (CGRP), neurokinin A (NKA) and vasoactive intestinal peptide (VIP) in the internal jugular blood of patient responders and non-responders to rizatriptan assessed during a migraine attack at each time point of the study

Statistical significance: patient responders to rizatriptan vs. baseline at 1 h: CGRP =
P < 0.002; 2, 4, 6, and 12 h:
P < 0.0001; at 1 h: NKA =
P < 0.001; 2 h:
P < 0.0002; 4, 6, and 12 h:
P < 0.0001.
For VIP it was impossible to calculate a significant difference at the different time points with respect to baseline because the levels of this neuropeptide were undetectable.
Patient non-responders to rizatriptan vs. baseline at 1, 2, 4, 6, and 12 h: CGRP and NKA: P not significant.
VIP levels were detectable in only five patients with autonomic signs at baseline in responders to rizatriptan. n.d., Not detectable.
All values in pmol/l.
Discussion
Few studies have been conducted until now specifically investigating the clinical characteristics of non-responders to triptans, and these have mostly regarded sumatriptan. In a study conducted on sumatriptan non-responders, a higher body mass index and migraine onset at an earlier age have been found and, most importantly, these patients treated their migraine attacks earlier. In the oral group, non-responders had attacks associated with more severe vomiting and photophobia, more often went to sleep or rest, and more frequently experienced initial worsening of the headache after sumatriptan administration. Within patients, no differences were found between attacks with and without response (24).
In another study, sumatriptan non-responders indicated less influence on their migraine of menstrual factors, had a higher lifetime prevalence of generalized anxiety, and showed 16 Personality Factor scores indicating greater shyness, self-sufficiency and perfectionism. Non-responders were also more imaginative and less socially outgoing. Although they must be interpreted with caution due to small sample size and the multiple comparisons made, these results suggest that biological differences exist between the two patient groups, which probably account for both the differences in their responses to sumatriptan and in the related clinical features (25).
In a recent review, Dodick (5) critically considered the trials in which patients with a poor response to sumatriptan were switched to treatment with other triptans. He observed difficulties in the comparison of findings among them, based on the consideration that placebo-controlled studies are more rigorous than open-label trials, and that prospective screening of patients for poor sumatriptan response identifies patient populations that are more recalcitrant to triptan therapy. According to him, this may explain why a lower level of pain relief was obtained in patients examined with a placebo-controlled design study, which prospectively screened out patients who achieved pain relief with sumatriptan in prerandomization attacks (2, 3), than in those patients studied with a less rigorous design or which relied on a retrospective history of poor sumatriptan response (rizatriptan/zolmitriptan trials) (1).
Aside from these considerations, Dodick affirmed that baseline headache intensity varied widely across the sumatriptan non-responder studies, and could also have influenced the response to sumatriptan at baseline (5). Patients with severe intensity of pain are clearly more difficult to treat and condition the definition of poor response, which could eventually be revisited when the attack was treated when pain was of mild intensity (11).
Although few studies have focused on the clinical features of attacks in non-responder patients, the published experience concerning a better response to triptans is limited to sumatriptan and the findings of open studies are still under investigation. Concerning sumatriptan, the above findings support the occurrence of autonomic signs during migraine attacks in a subgroup of migraine patients, and this peculiar clinical phenotype has been shown to be associated with a better response to triptans (19).
This evidence suggests the activation of a trigemino-parasympathetic reflex and the release of both potential trigeminal markers, CGRP and NKA, and that of the parasympathetic marker, VIP, during a migraine attack in these patients. Until now, no studies have been carried out investigating at the same time the clinical characteristics of migraineur responders or non-responders to rizatriptan and their biochemical phenotype with particular reference to the markers of trigeminal and autonomic system activation. This aspect has been investigated in the present study in a limited number of patients (10 responders and 10 non-responders), due to the difficulty in obtaining samples from the external jugular venous blood in a greater number of patients.
In our study, the poor response to rizatriptan was verified in three attacks when pain was moderate or severe at baseline before rizatriptan administration. Three additional attacks were assessed when pain was mild at baseline and the poor response to rizatriptan was also confirmed. Patients included in the study can therefore be considered true rizatriptan non-responders.
We found that these patients have ‘purer’ migraine attacks and more clinical autonomic activation than rizatriptan non-responders. At least 25% of the 20 MoA patients had autonomic unilateral symptoms and they represent 50% of rizatriptan responders.
Our findings, although not in agreement with previous reports of controlled studies, which showed autonomic dysfunction only in migraine with aura patients (26), concur with the more recent findings of Barbanti et al. (19), which demonstrated the occurrence of unilateral cranial autonomic symptoms, such as lacrimation, conjunctival injection, eyelid oedema and nasal congestion, all hallmarks of trigeminal autonomic cephalgias, in 81 of 177 consecutive migraine patients (45.8%) with ocular symptoms alone or in combination with nasal symptoms being the most frequent. In the same study, the headache was more severe and more strictly unilateral in patients with unilateral autonomic symptoms than in those without them. The authors hypothesized that the presence of these symptoms is suggestive of an activation of the trigeminal-autonomic reflex, probably related to an over-activation of the trigeminal afferent arm, and that this evidence may have therapeutic implications, given the greater possibility of recruitment of peripheral neurovascular 5-HT1B/1D receptors (the target of acute migraine treatment) in such patients.
To confirm this hypothesis, the same authors, in a more recent study, investigated the response to sumatriptan 50 mg in 72 consecutive migraineurs with unilateral cranial autonomic symptoms (20). Pain relief was recorded in 47 (65.3%) of these patients at 1 h and in 59 (81.9%) at 2 h. Pain-free response was reported by 22 patients (30.6%) at 1 h and by 44 (61.1%) at 2 h. Responsiveness to sumatriptan did not correlate with the type or number of unilateral cranial autonomic symptoms. The finding of at least one autonomic sign in 50% of our rizatriptan responders is therefore in line with the above results, suggesting that the occurrence of unilateral cranial autonomic symptoms (independent of the number or type) may predict a positive response to the triptans in a selected population of migraine patients.
In our study, neuropeptide measurements confirmed the relationship between the greater activation of the trigeminal system in migraine patient responders to rizatriptan. The better response to rizatriptan treatment corresponds to a greater release in the external jugular blood of CGRP and NKA, and can furnish the basis for understanding the biochemical ictal phenotype of these patients. One-half of rizatriptan responders also showed detectable levels of parasympathetic activation, VIP, and all of them had at least one sign of unilateral autonomic activation at baseline.
In contrast, the lack of a response to rizatriptan seems to be associated with a lesser degree of trigeminal activation and the lack of parasympathetic activation at baseline. This was clinically confirmed by evidence of the greater proportion of patients with a bilateral location of pain, and pain of a non-pulsating quality, although of severe intensity (in 90%), and the lack of unilateral autonomic signs in any of them throughout the study.
Biochemically, this clinical phenotype was reflected by lower levels of the markers of trigeminal activation, CGRP and NKA, compared with responders to rizatriptan, and undetectable levels of the marker of parasympathetic activation, VIP. At the moment, it is difficult to explain the discrepancy between the clinical and biochemical findings in these patients. It can be hypothesized that, although there was a smaller release of sensory neuropeptides in these patients, a peripheral and perhaps central sensitization of first- and second-order trigeminal neurons occurred to explain the severe intensity of pain and its maintenance in the majority of patients. Pulsating quality, which is believed to be related to peripheral sensitization, according to Burstein et al. (27), was found in 50% of our non-responder patients. This suggests a peculiarity of these patients related to a different response to trigeminal neuropeptide release. Allodynia, which is related to central sensitization by the same authors, was not evaluated in our study and needs to be assessed in future research in triptan non-responders compared with responders. The poor response to rizatriptan treatment in these patients was not accompanied by any significant variations in trigeminal neuropeptides within 12 h of rizatriptan administration. Thus, it cannot be excluded that the lack of a rizatriptan effect on neuropeptide levels may be secondary to the fact that rizatriptan is ineffective in these patients.
The biochemical data obtained from non-responders to triptans could be interpreted as if these patients had a ‘softer’ trigeminovascular activation. However, this does not mean that treatment of their attacks with a triptan should be easier than treatment of patients with more severe attacks and more severe autonomic activation, considering that one of the main targets of the recognized mechanism of action of triptans is the presynaptic blockade of neuropeptide activation from activated trigeminal endings.
The findings of this open-label study, carried out on a limited number of migraine patients, are among the first evidence to delineate the biochemical characteristics of responders and non-responders to a triptan and need to be confirmed for other drugs of this class. The levels of other neurotransmitters should also be investigated in future research to verify other aspects of migraine pathogenic mechanisms which can influence the response to triptans.
No placebo effect was assessed in our study, which was limited to the assessment of pain relief of rizatriptan only. Therefore, it cannot be excluded that the finding of unresponsiveness to rizatriptan may also imply unresponsiveness to placebo. This is particularly noteworthy, since three of six previously assessed attacks were of mild pain, whereas a placebo response of mild pain to pain free was seen in about 30% of treated subjects in trials of mild pain treatment with another triptan, i.e. sumatriptan (28). It may therefore be appropriate to design studies with a placebo arm involving patients with a poor response to triptans to investigate any resistance to the placebo effect.
Footnotes
Acknowledgements
The authors express their gratitude to John A. Toomey for editing the English and Marisa M. Morson for technical assistance.
