Abstract
Evidence for a disturbed maturation of information processing in migraine came recently from evoked and event-related potential studies during childhood. In adult migraineurs, deficient habituation is proposed as principal interictal abnormality and was found inter alia for Visual Evoked Potentials (VEPs). This study investigated response and habituation to pattern-reversal VEPs and its maturation in 102 children with primary headache (migraine with and without aura, tension-type headache) and 79 healthy controls from 6 to 18 years. A reduction of N180 latency from pre- to postpubertal age reflects maturation and was clearly present in controls but lessened in migraineurs. N180 latency was prolonged in migraineurs without aura from 12 years onwards. Habituation did not differ between groups. In conclusion, diminished N180 latency reduction with age in migraineurs gives further evidence that maturation of visual information processing is altered in migraine. Deficient habituation to pattern-reversal VEPs could not be confirmed during childhood migraine.
Introduction
Visual symptoms such as scotoma and photophobia are clinical features of migraine attacks, and psycho-physical studies of the visual system give evidence for subtle deficits even in the migraine-free interval, e.g. more intense illusions to grating patterns (1) and faster low-level performance (2). In migraine, the visual system undergoes interictal excitability changes that might be linked to a deficient habituation which is proposed as central interictal abnormality of sensory processing (3). Habituation is defined as a waning in the organism's response to a repetitive stimulus (4) and allows to ignore stimuli that have lost novelty or meaning (5). In evoked and event-related potentials, the within-run quasi exponential amplitude decline is the typical signature of habituation (6), but habituation may be reflected in peak latency changes as well (7).
A lack of habituation has been demonstrated in migraineurs for Visual Evoked Potentials (VEPs) (8–11), Acoustic Evoked Potentials (12–14), Somatosensory Evoked Potentials (15) as well as for event-related components as the Contingent Negative Variation (CNV) (16–19) and the P3 component (20, 21). Time of examination is relevant as the deficient habituation undergoes periodic changes with a tendency to normalization just before and during the attack (22).
Nearly all previous studies addressing habituation to visual stimuli in migraine were restricted to adults. Only one study investigating familial influences demonstrated an abnormal VEP habituation in adolescents (23). Investigation of evoked potentials over a broad age range during childhood and adolescence allows insights in maturation processes which are reflected by reduction of latencies and amplitudes with increasing age. Especially during childhood, amplitude changes are harder to interpret than latency changes because the amplitude decrease with age is due mostly to cranial changes (24) and amplitude shows higher variability (within and between subjects) than latency (25). As latency is a more reliable index of visual maturation we focused on the course of both amplitudes and latencies during ongoing stimulation to characterize habituation.
Age-dependent development was recently demonstrated to be altered in migraine by Kropp et al. (26) and by our group (27) in CNV. We found also deviant age-dependent development for the N135 latency of high contrast pattern-reversal VEPs over different spatial frequencies in children with primary headache (28). All these results argue for an altered cerebral maturation of information processing in migraine.
The aim of this study was (i) to investigate maturation of early visual processing in a habituation pattern-reversal VEP paradigm, (ii) to verify the presence (or absence) of a habituation deficit in children with migraine and (iii) to investigate the age-dependent development of habituation.
Methods
Subjects
Two hundred and five children and adolescents (98 females, 107 males) aged 6–18 years participated in the study: 82 headache-free controls (CO) and 123 headache sufferers who presented with headache as their primary problem. Headache was categorized according to the criteria of the International Headache Society (29). Inclusion criteria for the headache subgroups were migraine without aura (MO, code 1.1), migraine with aura (MA, code 1.2) or tension-type headache (TH, code 2.1 or 2.2) since at least one year and three or more attacks in the last quarter. Multiple diagnoses were possible, so most patients suffering from MA additionally had MO attacks. Beside migraineurs with and without aura, patients with TH were included to ascertain if a disturbed maturation or a habituation deficit, if verifiable, is specific for migraine. Diagnosis was established by a self-designed, structured interview with the child and a parent (30). Headache intensity was assessed using a 10 mm visual analogue scale. Headache characteristics were assessed using a child adapted prospective daily headache diary (31) over an 8-week-period from the first examination on. Exclusion criteria were other headache subtypes (IHS codes 1.3–1.7, 2.3, 3–13), neurological, ophtalmological or systemic diseases known to cause headache or VEP abnormalities, preventive therapy for migraine, and continuous medication with CNS effects. Only subjects reporting neither recurrent headaches nor ongoing medication and without any first degree relatives suffering from migraine were included into the control group. Patients were recorded in the headache-free interval, i.e. at least 72 h before and after an attack. The headache-free interval after the recording was ensured by a telephone interview three days later. 17 children had to be excluded from analysis because of headache attacks within this time frame, 7 other children had to be excluded from analysis because their data were incomplete or they produced too many artefacts. Demographic data of the remaining 181 subjects are given in Table 1. Headache characteristics (headache history, intensity and frequency of attacks) were comparable between subgroups except for headache intensity which was higher in both migraine subgroups compared to tension-type headache. This is in good accordance with the intensity criterion of the IHS (29). In agreement with other studies (32), about 1/3 of children with migraine suffered from clinically relevant behavioural and emotional problems (mainly internalizing disorders). These findings have been discussed elsewhere (33).
Demographic data

In the TH group, 4 patients (17.4%) had chronic TH.
In the MO group, 33 patients (57.9%) had additionally TH, 6 of them (10.5%) chronic TH.
In the MA group, all patients suffered also from attacks without aura. 11 patients (50%) had additionally TH.
Univariat analysis of variance (
Demographic data for headache-free controls (CO) and headache subgroups: tension-type headache (TH), migraine without aura (MO), migraine with aura (MA). For age, headache history and severity, mean and standard deviation (in parentheses) are given. For frequency, number and percentage of headache subgroup (in parentheses) are given.
The study was conducted according to the Declaration of Helsinki (current version, 1996) on biomedical research involving human subjects (Tokyo amendment) and the protocol was approved by the University of Heidelberg Ethical Committee. All children and their parents were instructed about the study and written informed consent was obtained.
Experimental design and recordings
Black-and-white checkerboard patterns with a check size of 7.5 min of arc and a contrast of 30% were binocularly presented on a 17′ video screen (800 × 600 pixel) subtending 12 × 15 degrees of visual angle. A reversal rate of 1 per second was chosen to allow proper investigation of the N180 component and its return to baseline. Subjects were seated in a distance of 1 m and instructed to fix a point 1.5 cm below and in the middle of the upper border of the screen as reported previously (34). To be able to reference to average of all electrodes, Neuroscan Synamp Amplifiers (Neuroscan Inc., USA) were used to record continuous DC 64 channel EEG with a sampling rate of 250Hz. Surface Ag-AgCl sintered electrodes were fixed using an equidistant electrode cap (Easycap, FMS, Germany). The right mastoid was used as ground and Cz′ as reference electrode for recording. Vertical and horizontal electrooculogram were also recorded. Impedances were kept below 5 kΩ. The EEG was processed off-line. During uninterrupted stimulation, five sequential blocks of 50 responses each were averaged. Data were transformed to average reference, averages were digitally low-pass filtered at 30 Hz (24dB/octave roll-off) and segmented into epochs of 640 ms (128 ms pre- to 512 ms post-stimulus). Recordings 128 ms pre stimulus were taken as baseline. The EEG-signal was corrected for DC-drifts (linear function), eye movements and blinks (algorithm described by Gratton et al. (35)). Artefacts were rejected automatically if the signal amplitude exceeded 150 µV and controlled by visual inspection. This way we made sure to exclude all severe artifacts but not to remove any signal systematically because background EEG amplitudes are larger in children and adolescents than in adults. Only artefact-free trials entered further analyses. Averages were analysed at Oz of the international 10/20 system for peak latencies (N80, P100, N180) and interpeak amplitudes (N80-P100, P100-N180). Peaks were defined as maximal negative or positive deflections in the following time ranges: 60–110 ms for N80, 90–160 ms for P100, 140–220 ms for N180. Peaks were detected automatically and confirmed manually by an experienced investigator who was blinded with regard to age and diagnosis.
Data analysis
Statistical analysis was carried out using SPSS for windows 10.0 software and Stata 8.0. For demographic data see Table 1. Children were divided into two age groups: 6–11 years (pre and early puberty) and 12–18 years (late and post puberty). Age groups and diagnostic groups were balanced with regard to gender (χ2 test age group × gender P = 0.402, diagnosis × gender P = 0.716). Results were compared by multiway analysis of variance (
For measuring habituation, a linear regression analysis of the interpeak amplitudes N80-P100 and P100-N180 was performed over the 5 blocks in each subject with Stata 8.0 software. Habituation was indicated by a negative slope of the regression line, potentiation by a positive slope. In the same way, a linear regression analysis was performed for the latencies N80, P100 and N180. Slopes were compared by analysis of variance (
Additionally, the frequency of subjects showing potentiation (positive slope) or habituation (negative slope) in the diagnostic groups was compared using the χ2-test. The α-level was set to 0.05.
Results
N80, P100 and N180 could be clearly identified in all recordings.
Interpeak amplitudes
Interpeak amplitudes N80-P100 and P100-N180 were not significantly different between diagnostic groups in the first block nor in the successive blocks (

Interpeak amplitude of P100-N180 in the younger (6–11y; □) and older (12–18y; ▴) age group of (a) healthy controls (CO) and patients with (b) tension-type headache (TH), (c) migraine without aura (MO) or (d) migraine with aura (MA). Results for interpeak amplitude N80-P100 are similar (not depicted).
Slope of the regression line
For P100-N180, the slope of the regression line was more negative in the younger age group indicating stronger habituation in children than in adolescents (P = 0.003). Diagnosis (P = 0.779) and interaction between diagnosis and age group (P = 0.662) had no significant effects (Fig. 2). For N80-P100, the slope of the regression line over the five blocks was not significantly different between headache groups (P = 0.523) or age groups (P = 0.309) and there was no significant interaction between diagnosis and age group (P = 0.318).

Habituation regression line: Slope of the regression line (mean ± standard error of the mean) of the interpeak amplitude (a) N80-P100 and (b) P100-N180 over block 1–5 in the younger (6–11y; □) and older (12–18y; ▪) age group of healthy controls (CO) and patients with tension-type headache (TH), migraine without aura (MO) or migraine with aura (MA). The number of subjects is given for each subgroup. For P100-N180, the slope of the regression line was more negative in the younger age group indicating stronger habituation in children (P = 0.003).
Slope of the regression line was significantly negatively correlated with the respective amplitude at the first block for both interpeak amplitudes: a high amplitude at the first block corresponded to a more negative slope (N80-P100: Pearson's correlation coefficient −0.332, P < 0.001; P100-N180: Pearson's correlation coefficient −0.524, P < 0.001). Because of this correlation, an
To investigate the occurrence of a rather qualitatively than quantitatively altered habituation response, the percentage of subjects showing habituation (negative regression slope) and potentiation (positive regression slope) was calculated in each subgroup for N80-P100 and P100-N180 (Fig. 3). For both interpeak amplitudes, the habituation-to-potentiation ratio did not differ significantly between headache groups, neither for the complete sample (N80-P100: P = 0.671; P100-N180: P = 0.824) nor in the younger (N80-P100: P = 0.860; P100-N180: P = 0.995) or the older age group (N80-P100: P = 0.581; P100-N180: P = 0.361).
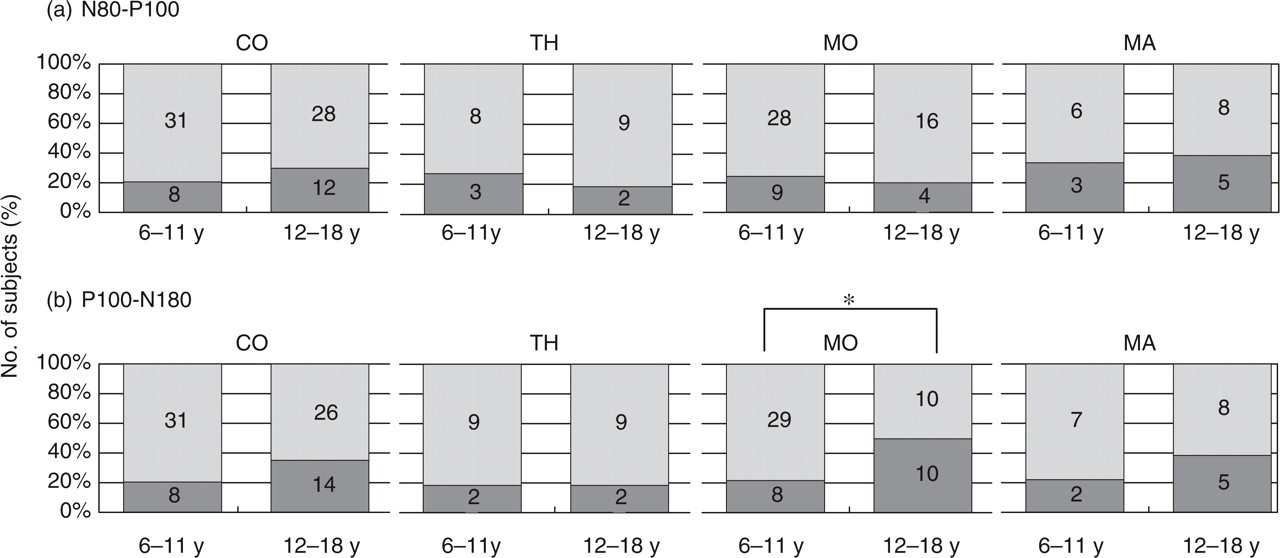
Percentage of subjects with habituation (light grey, regression slope < 0) and potentiation (dark grey, regression slope > 0) of N80-P100 (a) and P100-N180 (b) in both age groups in healthy controls (CO), patients with tension-type headache (TH), migraine without (MO) and with aura (MA). Absolute number of subjects is given inside the bars. Only in MO patients, the percentage of subjects showing P100-N180 potentiation was higher in the older age group (50% vs. 22%, ∗P = 0.028).
To detect possible changes of age-dependent development of habituation of one headache subgroup, the frequency of subjects showing habituation and potentiation was compared using the χ2-test within each headache subgroup. Only in the MO subgroup, the percentage of subjects showing P100-N180 potentiation was higher in the older age group (50% vs. 22%, P = 0.028, see Fig. 3 ). For the other headache subgroups, age had no significant effect on the habituation-to-potentiation-ratio within headache subgroups.
Latencies
Latencies of N80 and P100 increased significantly with ongoing stimulation (effect of factor block P < 0.001). There was a statistical trend for a reduction of P100 latency (but not N80) from the younger to the older age group (P = 0.064) and for an interaction between diagnosis, age group and block also for P100 (P = 0.071) but not for N80 (P = 0.428). P100 latency increased most over the five blocks in 12–18 years old migraineurs with aura (MA), latency reduction with age was clearly found in TH patients and controls but not in migraineurs without aura (MO) (Fig. 4).

Latencies of N80 (a,d), P100 (b,e) and N180 (c,f) over 5 blocks in the 6–11 years old (a–c) and 12–18 years old (d–f) control subjects (CO; •), patients with tension-type headache (TH; ▪), migraine without (MO; ○) or with aura (MA; □).
In contrast, N180 latency decreased with ongoing stimulation (effect of factor block P < 0.001). Reduction of N180 latency with age was also highly significant (effect of factor age group P < 0.001). N180 latency differed significantly between headache subgroups (effect of factor diagnosis P < 0.035). Post-hoc tests (Scheffé) showed significantly longer N180 latencies in MO when compared with CO (P = 0.025). Interaction between diagnosis, age group and block was significant (P < 0.01), N180 latency was prolonged in 12–18 years old migraineurs without aura (MO), latency reduction with age was smaller than in controls (Fig. 4). The remaining interactions between factors had no significant effects.
Slope of the regression line
Slopes of the regression lines of N80, P100 and N180 latency and the P-values for the influence of the factors diagnosis, age group and the interaction between diagnosis and age group are given in Table 2. Slope for N180 latency is usually negative indicating shortening with ongoing stimulation, whereas slope for N80 and P100 is usually positive. The only significant effect was the interaction of diagnosis and age group on the slope of the regression line for P100.
Slope of the regression lines for latencies N80, P100 and N180
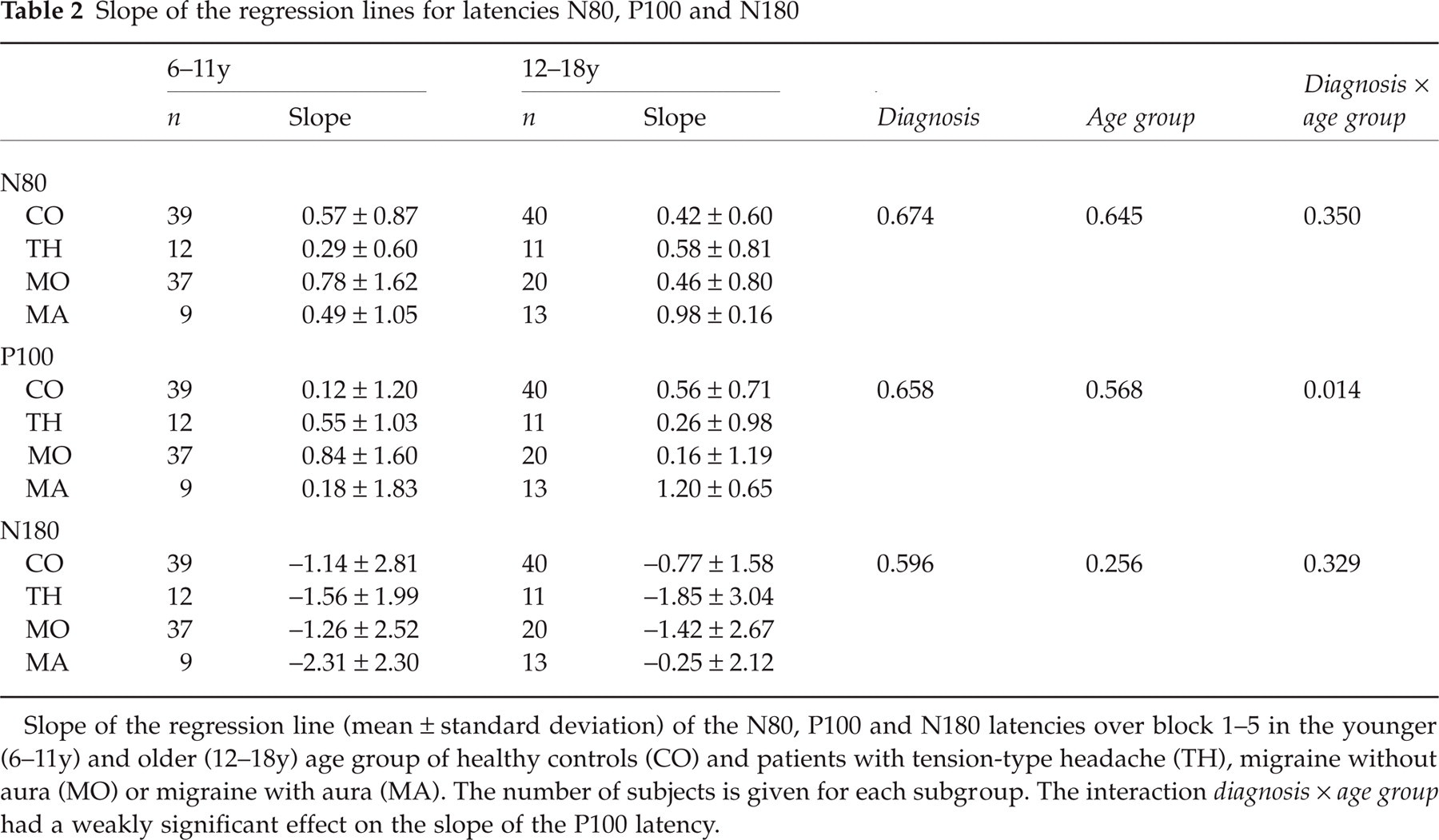
Slope of the regression line (mean ± standard deviation) of the N80, P100 and N180 latencies over block 1–5 in the younger (6–11y) and older (12–18y) age group of healthy controls (CO) and patients with tension-type headache (TH), migraine without aura (MO) or migraine with aura (MA). The number of subjects is given for each subgroup. The interaction diagnosis ×age group had a weakly significant effect on the slope of the P100 latency.
Like for amplitudes, the slope of the regression line was significantly correlated with the respective latency at the first block for N180 (Pearson's correlation coefficient −0.460, P < 0.001) and for P100 (Pearson's correlation coefficient −0.298, P < 0.001), but not for N80 (Pearson's correlation coefficient −0.115, P = 0.126). Because of this correlation, for slopes of N180 and P100 latency an
Data of this study were collected in parallel with data on high contrast pattern-reversal VEPs of different spatial frequencies which have been published elsewhere (28).
Discussion
This is the first study to investigate maturation of early visual processing in a habituation pattern-reversal VEP paradigm. In children, shortening of ERP latencies with age is usually taken as sign of maturation of receiving networks (38) whereas the amplitude decrease with age is attributed mainly to cranial changes (24). In the pattern-reversal habituation VEP paradigm used, N180 latency and P100-N180 amplitude decrease significantly with age. For P100 latency, only a trend for reduction with age was found. Our data are in line with previous results: In pattern-reversal VEPs over different spatial frequencies with a higher contrast we found a reduction of P100 and N135 latencies as well as interpeak amplitudes N80-P100 and P100-N135 with age (28). In general, our data extend previous findings of greater interpeak amplitudes in early childhood compared to adulthood (39) and a reduction of P100 latency in early childhood (25, 36), all these studies focused mainly on the first years of life and did not include school children.
In this study, latency maturation was different between diagnostic groups: The reduction of N180 latency with increasing age was smaller in MO patients than in headache-free controls. Headache-free controls showed pronounced reduction of P100 and N180 latency from pre- to postpubertal age. In patients with MO and MA, this latency reduction expressing maturation is reduced (statistically significant for N180; Fig. 4). N180 latency is under our stimulating conditions prolonged in children and adolescents with migraine without aura, this is in line with a prolonged N2 latency at high spatial frequency which we could demonstrate in adult migraineurs (40). TH patients seem to undergo normal maturation processes, but the small number of subjects makes any conclusion for TH only tentative. In a study using different check sizes with high contrast for stimulation, we found recently evidence for an altered maturation pattern of N135 latency in children with primary headache with a normal or even advanced maturation at low spatial frequency and a disturbed or delayed maturation at high spatial frequency (28). A lack of maturational changes with age in migraine was also found for the amplitude/stimulus intensity function (ASF slope) of the amplitudes N1P2 of the auditory evoked potential (AEP) (41). All these results fit in with evidence for a disturbed cerebral maturation of information processing in migraine coming from an altered age-dependent development of the habituation slope as well as the amplitude of the early and the late component of the contingent negative variation (CNV) (26, 27).
During childhood and adolescence, a habituation deficit to pattern-reversal VEP was not confirmed in our study. Neither the interpeak amplitudes N80-P100 and P100-N180 nor their regression lines over the five blocks were affected noticeably by headache diagnosis. The detection of the habituation deficit seems to depend on stimulating conditions, thus deficient habituation to VEPs could not be demonstrated in adult migraineurs in two studies (40, 42) with stimulating conditions slightly different from those used by Afra et al. (8) and Schoenen et al. (10): Lower pattern contrast and higher temporal (6) or spatial frequency (43) are known to enhance habituation effects. In the current study, pattern contrast and temporal frequency were lower than in the studies conducted by the Schoenen's group (8, 10), these conditions might contribute to the differing results (the lower temporal frequency was chosen to allow proper investigation of the N180 component and its return to baseline). Otherwise, incomplete maturation of neuronal circuits during childhood and adolescence probably adds to the absence of a deficient habituation in underage migraine patients, as habituation itself seems to be subject to maturation.
Characteristics of habituation to VEPs undergo a change from pre- to postpubertal age. Habituation of the interpeak amplitude P100-N180 is reduced with increasing age. To our knowledge, the maturation of the habituation to VEPs during childhood and adolescence has not been investigated so far. Habituation expressed as a decrease of amplitudes during ongoing stimulation is (for P100-N180) higher in pre- than postpubertal subjects which might be related to the generally higher amplitudes which have been demonstrated in children (28, 39). In fact, habituation expressed as a negative slope of the regression lines is for both interpeak amplitudes N80-P100 and P100-N180 significantly correlated with the respective amplitude at the first block. Afra et al. (37) found a similar correlation between the first block amplitude and potentiation of VEPs and concluded that the response potentiation in migraine is likely to be due to a reduced preactivation level of sensory cortices.
Our VEP data show a 20–25% reduction of interpeak amplitudes in the younger and a reduction up to 10% in the older age group of healthy controls which is in line with Psatta describing a 20–60% reduction in normal children (44) and with Afra et al. (45) reporting a reduction of aproximately 5% in adults.
In the current study, the percentage of subjects showing potentiation increased significantly with age in the MO subgroup only. This finding converges with the tendency to ‘potentiation instead of habituation’ which was described to be characteristic for migraine (10) and with data reporting a high amount of ‘potentiating’ subjects in a MO adolescents group with a mean age of 17 years (23). The pre- to post-pubertal increase of subjects showing potentiation in the MO and MA subgroup makes it plausible that the malfunction of habituation in migraine itself is developing with the neuronal maturation during childhood and adolescence. For habituation of P300 amplitude, a similar increase of the habituation deficit with age has been described in (21).
During ongoing stimulation, amplitude as well as latency of the N180 component decreased. Latency reduction is contrary to the characteristics of the earlier components N80 and P100 which revealed increasing latency (but decreasing amplitudes) over the five blocks. During habituation, latency prolongation has been described to occur for the P3 component (46, 47), whereas in VEP habituation studies latencies were not investigated (42) or unchanged (8, 10). Differences to the latter may be due to the slower reversal frequency used in our study (see above) which allows proper investigation of the N180 component until return to baseline. A decrease of latency with ongoing stimulation has been described for interpeak latencies to flash VEPs (7).
The decrease of N180 latency with ongoing stimulation might be due to a superposition of different components which is in accordance with previous results from source analysis (48). Components with different habituation kinetics could explain the latency reduction: A later habituating component with a decreasing amplitude and an earlier non or less-habituating component with a stable amplitude would result in shortening of the resulting N180 with ongoing stimulation, as the earlier component is unmasked when the later component minimizes.
In summary, maturational changes with age were different between diagnostic groups. A lack of reduction of N180 latency from pre- to postpubertal age in migraine patients and a prolonged N180 latency especially in adolescents gives further evidence for an altered maturation of information processing in the visual system in migraine. The phenomenon of deficient habituation which has been proposed to be characteristic for migraine could not be reproduced in children and adolescents. This might not be due only to methodological differences but partly to the finding that habituation itself is subject to maturation and decreases with increasing age during childhood. Thus the habituation deficit also may undergo an age-dependent development. According to this, the percentage of subjects showing potentiation increased significantly with age in the MO subgroup. Further research should address the localization and characteristics of the deficient maturation in migraine.
Footnotes
Acknowledgements
This work was supported by the Pain Research Programme of the Medical Faculty, University of Heidelberg (F207040, E1) and the Deutsche Forschungsgemeinschaft (DFG), Klinische Forschergruppe KFO 107/5. The authors would like to thank Kerstin Herwig for helping to acquire the data and for preprocessing of the EEG recordings including removing of artefacts and Dr André Rupp for helpful discussion.
