Abstract
Although headache frequency is increasing in preschool age children, an extensive evaluation of the clinical features in affected patients has yet to be achieved. This retrospective study examined 243 patients who were separately analysed in two distinct groups according to the age of onset and the age of first clinical evaluation. Group 1 included preschool age children, while Group 2 consisted of pubertal age patients. In all the patients the importance of a positive family history for headache as a risk factor was confirmed. In addition, when compared with Group 2, Group 1 showed greater male gender prevalence and earlier onset of the attacks. Regarding clinical features, in Group 1, compared with Group 2, the attack duration was shorter with lower symptom association such as photo- or phonophobia, nausea and no pain increase during physical activity. In the same group, tension-type headache was the predominant diagnosis, in contrast to the high migraine prevalence of Group 2. This study also showed that the International Headache Society 1988 classification criteria are not fully adequate for juvenile headaches. In fact, the headaches of more than 10% of patients in Group 1 still remained unclassifiable, while those of all the subjects in Group 2 were properly classifiable.
Introduction
Primary recurrent headache disorders represent a pathology with high and increasing prevalence during developmental age. Recurrent headaches are becoming a focus in paediatrics, but a complete understanding of them has yet to be achieved (1–6).
In children between 3 and 4 years old, headache prevalence ranges from 3% to 6%, but this prevalence increases from 5.9% to 6.3% around 7 years of age (7, 8). Other authors report values higher than 19% at 5 years old (2, 4, 7, 9). Migraine prevalence in the preschool age ranges from 1.4% to 3.2% (7, 10). These values have recently increased to 3.4% and 7.4% for the ages of 5 and 6 years, respectively (11).
Recurrent headaches in the preschool period are therefore not infrequent: the onset of headache occurs before the age of 5 years in about 40% of subjects (11), and 25% of migraineurs have their first attack in the preschool period (12).
A differential diagnosis of headaches in patients under 6 years old may be difficult due to the limits of the child in describing the pain precisely and the great complexity of the physician's task in performing an accurate physical examination at that age.
As recently reported, current diagnostic criteria (13) distinguishing the primary headache syndromes at the paediatric age may not be appropriate in terms of sensitivity and specificity (14–22), and a new classification of headache has been recently proposed with some adjustments to take account of this age group (23).
The goals of the present study were: (i) to verify the applicability of the present headache classification criteria (13) to compare two different paediatric age groups of affected patients (pubertal vs. preschool age); (ii) to verify the validity of the proposed reviewed classification in the diagnosis of migraine without aura (24); and (iii) to evaluate the existence of predictive factors in the evolution of headache onset at preschool age.
Materials and methods
The research included two phases.
Cross-sectional study. This study consisted of a comparison between two groups of subjects: the first with an early headache onset (<6 years old) and the second with late onset (between 12 and 18 years old). The data included in this phase were obtained from headache history forms filled out by the specialty doctors in the first clinical evaluation at the out-patient care Centres of Diagnosis and Therapy of Headache for the Juvenile Age of the Paediatric Departments at the University of Padova and University of Ferrara, Italy. Several headache features were considered to classify the patients: the frequency and duration of attacks, the intensity, the localization and quality of pain, pain exacerbation with physical activity, associated symptoms, and family history.
Longitudinal study. This part of the study was related to the clinical evolution of the primary headache with preschool onset. The follow-up analysis of headache evolution was carried out by means of a brief questionnaire (see Appendix) with relevant data on headache patterns, sent to all the families of subjects belonging to Group 1. The clinical evolution of the headache was classified as ‘favourable’ (remission or improvement) or ‘unfavourable’ (stability or worsening of symptoms), in relation to attack frequency. Subjects were defined as in ‘remission’ if no attacks appeared in the last 6 months, ‘improved’ if the frequency of attacks decreased by at least 50%, ‘stable’ if symptoms were reduced by less than 50%, and as ‘worsened’ if attack frequency increased by more than 50%.
This research was conducted in accordance with the Helsinki agreement on research involving human subjects. All patients and their parents gave informed consent.
Statistical analysis was performed by means of SPSS software (Chicago, IL, USA). To analyse the statistical significance between samples, several statistical tests were performed: χ2, Fisher, Student's t-test, the non-parametric Mann–Withney U-test, and the Kruskal–Wallis test. The χ2 test was used to compare the two groups of patients in relation to qualitative variables: gender, frequency of attacks, pain intensity, localization and quality, pain exacerbation with physical activity, presence of associated symptoms and diagnoses prevalence. In some cases, when more than 20% of cells had lower weighted frequencies than 5, Fisher's exact test was used instead of χ2. The t-test was used to compare the two groups in relation to quantitative variables, in particular the parents’ age at headache onset. Finally, the Mann–Withney U-test was used to compare the two groups in relation to the duration of the attacks. A P-value < 0.05 was considered to be statistically significant.
Patients
As previously outlined, the study population of patients affected by headaches was divided into two different groups. All patients attending the specialist headache clinic during the 1992–2001 period were studied retrospectively.
Selection was based on the age of onset and on the age at which the first clinical evaluation had been made in the centres of diagnosis and therapy for juvenile headache in Padova (n = 132) and Ferrara (n = 111). Statistical analysis grouped all the patients evaluated in the two centres into one single patient population with 243 cases.
All patients included in the study underwent the same anamnestic protocol regarding pre-existing risk factors, evolution of headache features (frequency, intensity, duration, quality and type of pain, coexisting or pre-existing symptoms) and the presence of a family history of headache (including only first-degree relatives). Furthermore, all the included subjects had physical and neurological tests to exclude the existence of a secondary headache. The diagnosis was made based on the up-to-date criteria established by the International Headache Society (IHS) (13) (Table 1).
Classification of headaches

According to IHS (1988) criteria (13).
According to Winner IHS-R (1997) criteria (24).
Results
Cross-sectional study
Subject features
Based on age at the first evaluation, two groups were isolated (Table 2): Group 1 (‘juniors’, n = 163: 82 from Padova and 81 from Ferrara), which included all patients <6 years old, and Group 2 (‘seniors’, n = 80: 50 from Padova and 30 from Ferrara), which included randomized patients aged 12–18 years and with headache onset after the age of 12 years. The mean age at the first evaluation for the subjects belonging to Group 1 was 4.9 ± 0.9 years (range 2–6), compared with 14.7 ± 1.4 years (range 12–18) for the patients in Group 2. The mean age of onset in Group 1 was 3.8 ± 1.0 years (range 1.0–5.8), and 13.1 ± 1.2 years (range 12–16.5) in Group 2. The gender prevalence was significantly (P < 0.01) different in the two groups: the percentage of males was higher in Group 1 (58%) than in Group 2 (39%).
Patient population

Headache pattern
Frequency of attacks: a subclassification of this parameter allowed a subdivision of the patients, characterized by: (i) less than one attack per week, (ii) one to three attacks per week, and (iii) more than four attacks per week. A comparison between Groups 1 and 2 regarding these subdivisions showed these values, respectively, without statistical significance: (i) 39% vs. 45%; (ii) 47% vs. 31%; (iii) 14% vs. 24%.
Duration of attacks: this value was different in the two groups (Table 3, Fig. 1) and significantly lower in Group 1 (P < 0.01). The attack lasted <1 h in 52% of subjects in Group 1 and in 15% of subjects in Group 2, 1–2 h in 20% and 24%, respectively, 2–5 h in 18% and 34%, respectively, and >5 h in 10% and 27%, respectively.
Duration of attacks

Duration of attacks in the two groups of patients.
Pain intensity was ‘severe’ in 33% vs. 44%, ‘moderate’ in 21% vs. 22% and ‘mild’ in 48% vs. 34% of the patients in Groups 1 and 2, respectively (P = NS).
Pain localization was unilateral in 24% of subjects in both groups, and bilateral in 76%, without significant differences.
Pain quality was pressing/tightening in 46% vs. 51%, pulsating in 42% vs. 45% and stabbing in 12% vs. 4% of patients in Groups 1 and 2, respectively (P = NS).
Pain exacerbation with physical activity was more frequent in Group 2 compared with Group 1 (64% vs. 39%) (P < 0.01).
Associated symptoms: at preschool age (Group 1) associated symptoms such as nausea (20% vs. 39%), photophobia (52% vs. 70%) and phonophobia (54% vs. 72%) were less frequent than at school age (Group 2) (P < 0.01), while vomiting was not significantly different between the two groups (18% vs. 19%).
IHS (1988)-based diagnoses
Concerning the first and second diagnostic levels of IHS classification (13), as shown in Table 4, it was possible to diagnose migraine in 36% vs. 58%, tension-type headache in 42% vs. 41%, and idiopathic stabbing headache in 10% vs. 1% of Groups 1 and Group 2, respectively. In Group 1, 12% of patients were not classifiable, while all subjects in Group 2 were properly classified.
Classification of headaches∗

MA, migraine with aura; MoA, migraine without aura; TTH, tension-type headache.
According to IHS (1988) criteria (13).
In the stratification of second level diagnoses there was a significant difference between the two groups (Table 5). Moreover, the diagnoses of ‘migraine disorder 1.7’ predominated in Group 1 (54%) in comparison with Group 2 (22%) (P < 0.01), and the diagnosis of ‘tension-type disorder 2.3’ prevailed in Group 1 (51%) vs. Group 2 (39%) (P < 0.05).
Diagnoses of migraine and tension-type headache (TTH)†

According to IHS (1988) criteria (13).
P < 0.01;
P < 0.05.
MoA, migraine without aura; MA, migraine with aura; TTH, tension-type headache.
Winner IHS-R(1997)-based diagnoses
When the re-classification by Winner et al. (24) of the criteria in use was taken into account (Table 1), the diagnoses of migraine without aura in juvenile patients increased from 16% to 35% and from 29% to 52% in Groups 1 and 2, respectively (Table 6). Moreover, more than half of the subjects previously classified (13) as 1.7 and, in particular, 56% in Group 1 and 60% in Group 2, were reclassified as migraine without aura.
Migraine without aura according to IHS (1988) and IHS-R (1997) criteria in two groups of patients
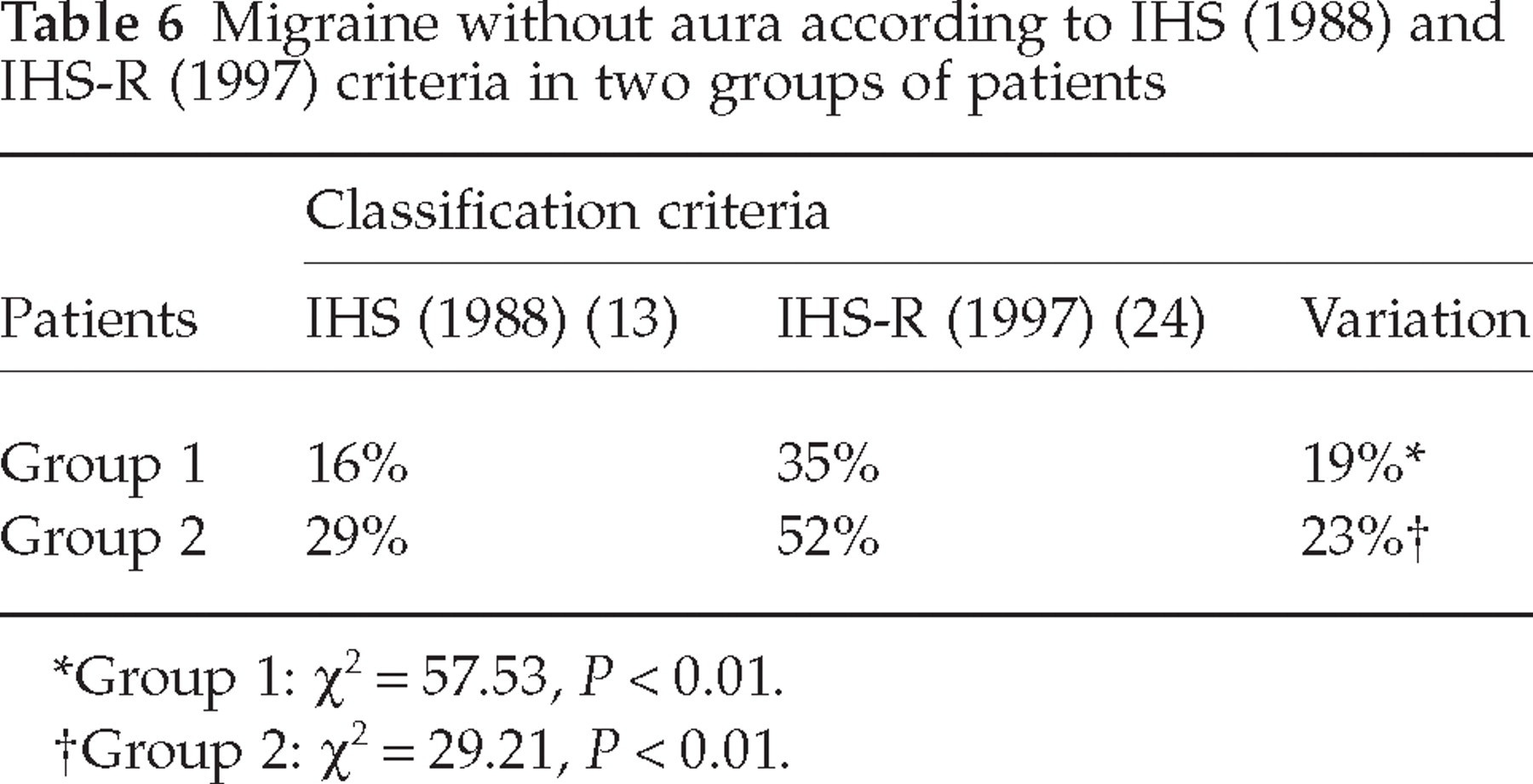
Group 1: χ2 = 57.53, P < 0.01.
Group 2: χ2 = 29.21, P < 0.01.
Family history of headache
This term is defined as the presence of at least one first-degree relative affected by migraine or tension-type headache. In 78% of subjects in Group 1 and in 59% in Group 2 there was a positive family history (P < 0.05). The mean age of headache onset in the parents in Group 1 (16 years; SD = 7.2; SEM = 0.9) was significantly less than that in Group 2 (26 years; SD = 10.3; SEM = 2) (P < 0.01; 95% confidence interval −14.04, −6.46).
Longitudinal study
The questionnaire sent to all the subjects belonging to Group 1 (n = 163) was returned filled by 71% (n = 115) of families. The mean interval between the first medical evaluation and the return of the questionnaire was 3.5 ± 2.7 years (range 0.5–10), with the mean age of patients being 8.3 ± 2.8 years.
Strength of the diagnoses
At the end of the follow-up (Table 7), the initial first-level diagnoses remained the same for 61% of the subjects with migraine, for 46% with tension-type headache, and for only 20% with idiopathic stabbing headache. In addition, within the group of patients initially with unclassified headache, 25% remained unclassified, 25% evolved into migraine without aura, 38% evolved into episodic tension-type headache, and the remaining 12% showed remission of headaches.
Follow-up: evolution of headache diagnoses of preschool age children (Group 1)
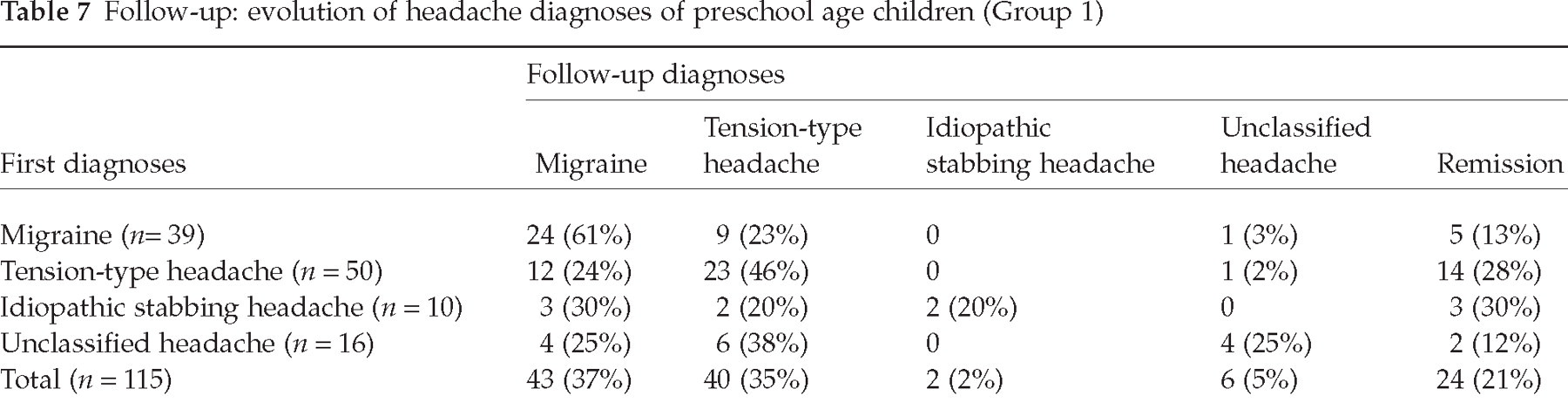
As regards the second level of diagnoses (13), since the first visit, none of the episodic tension-type headaches had become chronic, while 50% of chronic tension-type headaches had become episodic. Moreover, 14% of migraine without aura had evolved into episodic tension-type headache, while none of the latter had become migraine without aura.
Prognosis
During follow-up, remission in 21%, improvement in 46%, worsening in 15% and stability in 18% of cases were seen. Thus, the overall evolution of headache was favourable in 67% and unfavourable in 33% of patients in Group 1 (Table 8).
Follow-up: clinical headache evolution of preschool age children (Group 1)

Predictive prognostic factors
Two predictive and prognostic factors were identified: (i) patients with tension-type and idiopathic stabbing headache had a favourable evolution in 80% of cases, compared with 49% of patients with migraine (P < 0.05), in terms of remission (28% vs. 13%) and improvement (52% vs. 36%); and (ii) in the migraine group patients without vomiting associated with the headache attack at the initial visit had favourable outcomes in 74% of cases, compared with 33% of patients that had vomiting. In particular, at follow-up, 25% of patients without vomiting were still in remission compared with none of the patient with this symptom.
Discussion
This study demonstrates that, in juvenile headaches, there is a different gender distribution in the two groups, with higher prevalence of males at preschool age, as reported in the literature on the general population (6, 14). Nevertheless, other studies have not found any gender differences in early development (11, 25), while a higher female prevalence of headache during puberty has always been reported (26–28).
In the male population, headaches present not only a greater precocity in the onset of attacks (29), but also a higher remission rate in adult age (28).
Our data suggest that preschool age headaches have different clinical characteristics from those of successive ages. In particular, the attacks are shorter: the duration of headache is <1 h in 52% of preschool subjects, while this short duration is reported only in 15% of subjects aged 12–18 years.
The higher prevalence of migraine in Group 2 can explain the greater prevalence in this group of associated symptoms such as nausea, photo- and phonophobia in comparison with Group 1. Vomiting was present in about 20% of subjects in both groups.
These data show that the 1988 IHS criteria (13) may not describe fully the diagnosis of headache during juvenile age, especially in the preschool age. In fact, at the end of our study, 12% of these subjects remained unclassified. Furthermore, Aromaa et al. have used the current criteria (13) to evaluate a sample of patients aged 6 years, and recently found that no more than the 90% of subjects were classified (30).
Nevertheless, the sensitivity of IHS criteria (13) increases with age of the patients. In a multicentre study, 96.9% of patients aged 11.3 ± 3.0 years (range 2–18 years) were classified (17), with a higher percentage diagnosed in the adolescent period (98.6%) (22) and even more in adulthood (99.7%) (31). Our study demonstrates that the sensitivity of IHS criteria increases with advancing age, presumably because this classification resulted more specific than sensible for migrainous patients (13). Moreover, some of the parameters, i.e. duration of attacks, appeared too restrictive, so that in the last version the duration of attacks has been reduced (23).
The percentage of headaches classified as 1.7 and 2.3 (13), which are more frequent during the paediatric age (18, 21), decreases with increasing age. In fact, in our study it was possible to perform an accurate diagnosis in 47% of cases and a ‘probable’ diagnosis (1.7 and 2.3) in 41% of subjects in Group 1, vs. 71% and 29% in Group 2, respectively. Moreover, when Winner's revised criteria (24) were applied, a noticeable increase in the percentage of migraine without aura diagnoses was found, and this supports the hypothesis of the good specificity and poor sensitivity of the 1988 IHS criteria (13) for classification of paediatric migraine.
This study took place retrospectively from 1992 to 2001 and consequently data were analysed according to the 1988 IHS classification (13). Recently the 2nd edition of the International Classification of Headache Disorders has been published (23). This has not changed the major principles of the classification and diagnosis of primary headache disorders and the hierarchical system has remained unchanged. Like the first, the 2nd edition classifies patients according to the phenomenology of their headache (i.e. symptom based). In this new classification the attack of migraine without aura in children may last 1–72 h, i.e. it is shorter than in the previous classification (13).
Furthemore, new identities were introduced as ‘probable migraine without aura’ (code 1.6.1) and ‘probable tension-type headache’ (code 2.4) groups, which recall the same criteria included in the 1988 IHS classification for subgroups 1.7 and 2.3 respectively (13). Similar, the ‘primary stabbing headaches’ subgroup (code 4.1) overlaps the previous classification (13).
In our study, idiopathic stabbing headache is clearly more frequent in Group 1 than in Group 2, as reported in a previous study (32). The prevalence of this kind of headache in children ranges from 3.3% (32) to 3.6% (33) to 5.1% (34), with onset during the first 10 years of age and, unlike in the adult, rarely associated with other kinds of primary headache (32–34).
It is also relevant to note that the positive family history of primary headache represents an additional risk factor, especially for young patients. Similarly, in accordance with other data in the literature (35–37), a recent study demonstrated a higher frequency of positive family history of migraine in a group of subjects with headaches starting before 6 years of age compared with a group of subjects with onset after 10 years of age (12). In addition, an early onset of primary headache in the same family was also reported for migraine (12).
Although not included in the current diagnostic criteria for migraine (13), the significance of a positive family history of recurrent headaches was always well appreciated in the past systems of classification of juvenile headache (38, 39). It is now well established that a correlation exists between a positive family history and headache: this ranges, in different studies, from 46.5% to 88% (3, 12, 40). Furthermore, a positive family history is more important for young patients affected by migraine in comparison with those affected by tension-type headache (41), and a maternal family history appears especially important (3, 12).
In a 40-year follow-up study of school children with headache, Bille et al. (28) demonstrated that about one-third of patients with a positive family history presented typical attacks of migraine, thus confirming the importance of hereditary factors in migraine.
In our follow-up, the headache with preschool age onset seems to have a relatively benign prognosis as to possible spontaneous remission (21%) or improvement (46%) in the short interval considered. The relatively favourable prognosis of our patients (67%) confirms the results of other recent studies regarding either patients with headaches (79%) (42) or with migraines (from 46% to 80%) (28, 43). In particular, the percentage of remission described here (21%) is in agreement with recent data in the literature, in which the remission ranged from 27% (44) to 34% (42). According to other authors, patients with headache onset during preschool age show a high probability of persistence into adulthood (8) and a recent study demonstrated a four-fold risk of unfavourable evolution in patients with preschool onset compared with subjects with later headache appearance (12).
A more favourable prognosis was reported for tension-type headache compared with migraine. The tension-type headache with preschool onset presents a decrease of attack frequency in time and more stability of diagnosis without transformation into migraine, which instead can potentially evolve into an episodic tension-type headache.
The better prognosis for tension-type headache vs. migraine (with improvement in 52% vs. 36% and remission in 28% vs. 13%, respectively) is in agreement with current literature (42, 44–46).
In conclusion, this study confirms some of the typical features of headache pattern in juvenile patients, such as the shorter duration of attacks, the lower increase in pain during physical activity, or the association of nausea, photophobia and phonophobia.
Tension-type headaches appear well represented during preschool age, exceeding the percentage of migraine and of unusual headaches, such as idiopathic stabbing headaches, which are relatively frequent during the first decade of life (32–34). Moreover, the past criteria of headache classification (13) appear to be less suitable for the preschool age, since it is not possible to reach the high percentage of diagnosis typical of pubertal age (14, 16–19, 45, 47, 48), and also because there are more subjects with a diagnosis of probability (type 1.7 and 2.3) (45). Nevertheless, when the revised criteria proposed by Winner et al. (24) were applied, the percentage of patients classified as affected by migraine increased in both groups of our study. A family history of primary headaches is well represented in preschool age patients with a significant precocity of onset.
In contrast to the excellent prognosis of stabbing headaches (32), migraine presents a less favourable evolution, especially in cases with associated vomiting. Finally, the follow-up of preschool age subjects showed a progressively bigger percentage of properly classified headaches, with a migraine diagnosis over the course of time.
Footnotes
Appendix
QUESTIONNAIRE
NAME AND SURNAME:
DATE OF COMPILATION:
THE QUESTIONNAIRE IS COMPILED BY:
Please, answer the following questions. If necessary, you can choose more than one answer.
(1) Have you had any headache in the last 6 months?
Yes
No
If not, when was the last time you had a headaches?
Less than 1 year ago
1–2 years ago
More than 2 years ago
Otherwise, if you have had a headache in the last 6 months, answer the following questions as well.
(2) Does your headache occur only during intercurrent illnesses (such as influenza, fever …)
Yes
No
(3) Which is the frequency of attacks?
Less than 1 attack/month
1–3 attacks/month
1 attack/week
2–3 attacks/week (less than 15 days/month)
4–6 attacks/week (more than 15 days/month)
(4) Usually how long does the pain last?
Less than 30 min
30 min−1 h
1–2 h
3–5 h
6–12 h
13–24 h
2–3 days
The pain is continuous
(5) What is the intensity of the pain?
Severe (it doesn't allow normal activities)
Moderate
Mild (it allows normal activities)
(6) Where is the pain location?
Unilateral
Bilateral
(7) The pain quality is:
Pressing/tightening (as a tight helmet)
Pulsating (as a beating hammer)
Stabbing (as a pin)
(8) Is the pain exacerbated by everyday physical activities?
Yes
No
(9) At what time of the day does the headache usually occur?
Upon waking
In the morning
In the afternoon/evening
At night
Variable
(10) Are one or more of the following symptoms present during the attack?
Nausea Blush
Vomiting Pallor
Photophobia Abdominal pain
Phonophobia Paraesthesia
Irritability Speech disorders
Perspiration Anorexia
Dimming eyes Giddiness
Tearing
(11) Do you take any medicine for headache?
Yes
No
If yes, specify name and posology: ............................................................................................
(12) Have you ever used or are you actually using a preventive therapy for your headache?
Yes
No
If yes, specify kind and length of the therapy: ............................................................................................
