Abstract
Over the years the paradigm of treating early during the migraine attack has become well established in clinical practice. It is also recommended that the 5-HT1B/1D agonists be administered early during the migraine attack for efficacy. This is because it has been proposed that most migraineurs are less responsive to delayed treatment, owing to the development of central sensitization of the pain transmission. The main objective of this prospective, cross-over study at a specialist clinic was to evaluate if these recommendations should also apply to the subcutaneous formulation of sumatriptan. Results are based on 20 adult International Headache Society migraineurs. Two attacks (n = 40) were treated with 6 mg subcutaneous sumatriptan as early as possible after the onset of migraine headache and two attacks (n = 40) as late as the patients could bear. The median intra-individual difference between the two strategies in time from first occurrence of pain to injection was 5.7 h and the median intra-individual difference in pain intensity at the time of injection was 29 visual analogue units. No significant differences were found in time to freedom from pain, pain severity at 1 and 2 h, area under the curves from injection to pain free or in headache recurrence after injection. At the end of the study, most of the patients claimed that their medication was as effective when given early as when given late in the course of the attack. The discrepancy between our present findings and retrospective analyses of trials on oral triptans probably has more to do with the less disturbed pharmacokinetics early during the migraine attack than with central sensitization. Consequently, we recommend nonoral formulations of triptans, which do not necessarily have to be administered early during the migraine attack to provide efficacy. In conclusion, it is reassuring for migraineurs that it is worthwhile taking their medication in an appropriate formulation even if they have not been able to do so early in the course of the attack.
Keywords
Introduction
Migraine is an inherited, chronic, neurological disorder characterized by recurrent episodes of severe throbbing headaches associated with nausea, and increased sensitivity to sound and light but freedom from symptoms between the attacks (1–3). It is considered possible to identify five phases of a migraine attack: the prodromal phase, aura, the headache phase, resolution and postdromal phase (4). Of these, it is only the classic neurological aura which has a distinct onset and end. According to the international classification, a migraine attack lasts from 4 to 72 h (3). The aetiology is complex and incompletely known but, in addition to genetic predisposition, dilation and/or swelling of the intracranial extracerebral blood vessels seems to be a contributing factor, possibly in combination with sensitization of nerve fibres and/or neurogenic inflammation (5, 6). In short, migraine is considered to be a primary disorder of the central nervous system with secondary vascular effects, a neurovascular disorder (2).
The introduction of triptans (selective 5-HT1B/1D agonists) into clinical practice has expanded the therapeutic options for doctors treating migraine sufferers (2, 7, 8). For many migraineurs, however, oral therapy with triptan provides complete pain relief in only some of the attacks (8–11). There are at least two plausible explanations for this variation in consistency of response. The most likely is that gastrointestinal motility is inhibited and the gastric emptying time delayed during the migraine attack (11–15). In addition, there is always a risk that migraine-induced nausea will culminate in vomiting. Furthermore, in addition to their antimigraine properties, triptans per se appear to prolong the gastric emptying time by activating 5-HT1 receptors on gastric myenteric neurons (16–18). For these reasons, the absorption of any triptan taken orally during the migraine attack will be erratic and treatment effects inconsistent (11, 19). This is supported by the finding that the intra-individual consistency of response of the subcutaneous formulation of sumatriptan is much higher than that of the tablet (19). Despite these barriers to good efficacy and high reliability, the tablet is the most used formulation of triptans, since most patients favour this oral route of administration (11, 20).
The second explanation is that the increase in migraine pain intensity reflects the progress of the migraine attack and the cascade of pain mechanisms involved (21–23). If the migraine attack is allowed to continue without treatment for only a few hours, the migraineur runs a greater risk of sensitization of both second- and third-order neurons of the trigeminal nerve, leading to both cutaneous allodynia corresponding to the innervation area and poorer response to treatment with a triptan (21, 23–25). According to Burstein and coworkers, about 50% of migraineurs develop cutaneous allodynia within 1 h and about 75% within 4 h, and in the presence of cutaneous allodynia the triptans do not provide freedom from pain. This type of cutaneous allodynia is, thus, suggested to be driven by both attack duration and pain intensity (23). Whereas the throbbing characteristic of the pain and sensitivity to coughing (peripheral first-order trigeminal neuron sensitization) respond to triptan therapy, the cutaneous allodynia and expanded fields of perception (central second-order trigeminal neuron sensitization) may remain the same for hours/days. According to this, migraineurs are most responsive to medication within the initial 30–60 min of an attack before cutaneous allodynia develops, and the probability of a consistent pain-free outcome increases dramatically if triptan therapy is carefully timed to precede any signs of cutaneous allodynia (21–23). It is, however, still disputed whether this type of cutaneous allodynia can also be used clinically as a practical marker that predicts decreased responsiveness to acute treatment with a triptan (26).
According to both these explanations, early treatment when the pain intensity is mild would have a better chance of providing good and consistent efficacy for a variety of reasons. Treating a migraine attack as soon as it begins has long been the common clinical strategy in order to anticipate the impaired absorption during the migraine attack. This has been so without knowing that one hereby also avoids development of cutaneous allodynia and poorer response to acute symptomatic treatment (27). This appears to be supported by analyses of data from clinical trials demonstrating improved outcome of most triptans given orally during mild pain as opposed to when pain is more severe (28–40). Counter-claiming this theoretical paradigm, patients in daily clinical practice commonly report subcutaneous sumatriptan as having high efficacy independently of the time or pain intensity when treatment is initiated (41). To investigate these issues further, Dahlöf and coworkers performed an early, large (n = 90), prospective, double-blind, randomized, placebo-controlled, cross-over study comparing early and late (4 h later) sumatriptan given as 6 mg subcutaneous injection to migraineurs characterized by attacks associated with frequent headache recurrences [GlaxoSmithKline (GSK), data on file]. All attacks were treated when pain was moderate/severe. In the GSK study, the primary efficacy parameter was a comparison of the number of headache recurrences after early and late treatment. Secondary efficacy parameters were the number of patients who obtained headache relief (grade 2/3 to grade 0/1) at 60 and 120 min after each dose and migraine symptoms (nausea, vomiting and/or photo-/phonophobia) at 60 and 120 min after each dose. Further comparisons were made of time to meaningful relief, clinical disability and pattern of sumatriptan use over the 72-h period of the open phase. No significant differences were found in any of the efficacy parameters. Headache relief 1 h after injection was reported by about 80% of subjects after both early and delayed treatment. The rate of recurrence was 71% after early and 74% after late injection (GSK, data on file). It was concluded that subcutaneous sumatriptan administered late in the course of the attack was as effective as the same treatment when given early. Accordingly, the therapeutic advantages of treating early appear to apply for oral therapy with triptans but not to be valid for parenteral administration of these drugs.
Subcutaneous injection is associated with the highest intra-individual response consistency among all formulations of sumatriptan. Within-patient consistency of response for three out of three attacks occurs in 73%, 35%, 32% and 41% of patients treated with sumatriptan injection, nasal spray, tablets, and suppository, respectively (7, 19). Thus, from the perspective of reliability of response, subcutaneous injection is the best choice. In order to obtain further data on subcutaneous sumatriptan given in different phases of the attack we performed an open cross-over study in clinical practice designed to receive the longest possible separation in time between early and late treatment. Furthermore, a new integrated time-intensity method for self-assessment of migraine symptoms was tested.
Methods
Design
This was an open, prospective, cross-over study conducted between November 2001 and January 2004 in an out-patient setting at the Gothenburg Migraine Clinic. The protocol was approved by the Ethics Committee of Göteborg University.
Subjects
Eligible patients were aged 18–65 years and had used sumatriptan regularly without any contraindications for at least 6 months in acute migraine, with or without aura according to the International Headache Society criteria (42), during the previous 12 months. Patients were excluded if they had known or probable drug-induced headache, or had any severe concurrent medical condition which affected data interpretation. The required number of participants to achieve adequate power could not be calculated beforehand. A sample size considered large enough to catch a clinically relevant finding was set at approximately 30 (30). A total of 58 patients were included into the study. A total of 30 (n = 30) participants dropped out. Explanations given for this were: ‘too difficult’ (n = 12), ‘lack of time’ (n = 4), ‘mental exhaustion’ (n = 3), ‘lack of motivation’ (n = 3), no need for injections (n = 1) and withdrawn consent for no specified reason (n = 7). An additional seven (n = 7) patients were excluded from the analysis owing to inadequate data (n = 3), other acute medication before injection of sumatriptan (n = 2), pregnancy (n = 1) and lack of curves from attacks treated early (n = 1).
One woman (designated S:020), aged 60 with migraine without aura for 45 years, developed a status migrainosus during both of the attacks treated late and was therefore not eligible for statistical analysis according to the protocol. This patient was not using any migraine prophylaxis. She experiences scalp tenderness during her usual attacks as well as pin-prick-like paraesthesias in the skin after sumatriptan injection. Her data are reported separately (see Results).
Protocol
Two attacks were treated with 6 mg subcutaneous sumatriptan as early as possible after the onset of migraine headache and two attacks as late as the patients could bear with at least a 48-h sumatriptan-free period between the attacks. Patients took sumatriptan either early or late in a sequence of their own choosing. A maximum of five open label doses were taken per attack, and not more than two sumatriptan doses were permitted in any 24-h period. Rescue medication (excluding ergotamine-containing products) was permitted when an inadequate response to the study medication was observed within 6 h after the first dose. No open-label sumatriptan doses were permitted after use of rescue medication.
All characteristic symptoms were documented using 100-point visual analogue (VA) scales (Fig. 1) filled in hour-by-hour during the awake portion of 72 h from the start of attacks. The present report is based on headache intensity but not aura, nausea, vomiting, photophobia or phonophobia (which were used only to confirm that all treated headaches were in fact migraine). Data extracted from the scale are the exact time of injection from start of migraine headache, the pain intensity at time of injection, and the pain intensity 1 and 2 h after injection. Using an electronic digitizer (IS/One Input system™; Kurta Corp, Phoenix, AZ, USA), the area under the curves (AUC) from injection to pain free was measured. This parameter constitutes the (changing) pain intensity multiplied by suffered migraine time. Data are reported as AUC units (25 mm2) equal to 10 VA steps multiplied by 1 h. Finally, the number of recurrences (defined as return of migraine less than 24 h after pain free from injection) was documented.

The 100-point visual analogue (VA) scale filled in hour by hour during the awake portion of 72 h from the start of attacks. Information extracted in the present study was time from first occurrence of pain to injection (A), pain intensity at the time of injection (B), pain intensity at 1 and 2 h after injection (C, D), time from injection to freedom from pain (E), area under the curves from injection to pain free (F) and number of recurrences after injection (G).
Statistical analysis
Sign-test for paired samples was used to compare non-parametric data (VA scales). Paired samples t-test was used for parametric data. All analyses were performed using the Statistical Package for Social Sciences (SPSS™) program (version 11.0 for MacOSX; SPSS, Inc., Chicago, IL, USA). Significance levels were set at 0.05.
Results
The analysis is based on 20 patients (median age 47.0 years, range 25–59, 15 women) with migraine (12 migraine without aura only and an additional six with a combination of migraine without aura and migraine with aura). The mean number of years with migraine was 29.0 (SD 9.6, median 31.5).
The mean reported attack frequency was 4.1 per month, and the mean number of injections per attack was 1.2 (median 1 per attack). Accordingly, the mean use of sumatriptan was 5.0 (SD 4.1, median 3.5) injections per month. Additional medications used for acute migraine treatment were paracetamol (n = 1), pronaxen sodium (n = 1), ibuprofen (n = 1), acetylsalicylic acid +codeine (n = 2), naratriptan (n = 1), rizatriptan (n = 2) and eletriptan (n = 1). Three patients had been put on pharmacological prophylaxis (one on propranolol and two on amitriptylin) at least 6 months before the start of the study.
The mean time from first experience of pain to injection was 1 h 36 min (median 1.0 h) for early treatment and 7 h 16 min (median 6.1 h) for late treatment. The mean intra-individual difference in time between early and late treatment was 5.7 h (median 4.5, range 0.9–14.5 h). This difference between the two strategies in time to injection was highly significant (P < 0.0001, paired samples t-test). The mean intra-individual difference in pain intensity between the two strategies was 29 VA units (median 25, range −15 to +81 units), which was also significant (P = 0.031, sign test).
One hour after administration of the first injection, 42% (17/40) of attacks treated early had reached complete headache relief, compared with 28% (11/40) following late treatment. Two hours after administration of the first injection, 62% (25/40) of attacks treated early had reached complete headache relief, compared with 55% (22/40) following late treatment. No significant difference was found in pain severity either at 1 h (P = 0.24, sign test) or 2 h (P = 0.57, sign test) after injection. The data are shown in Fig. 2.
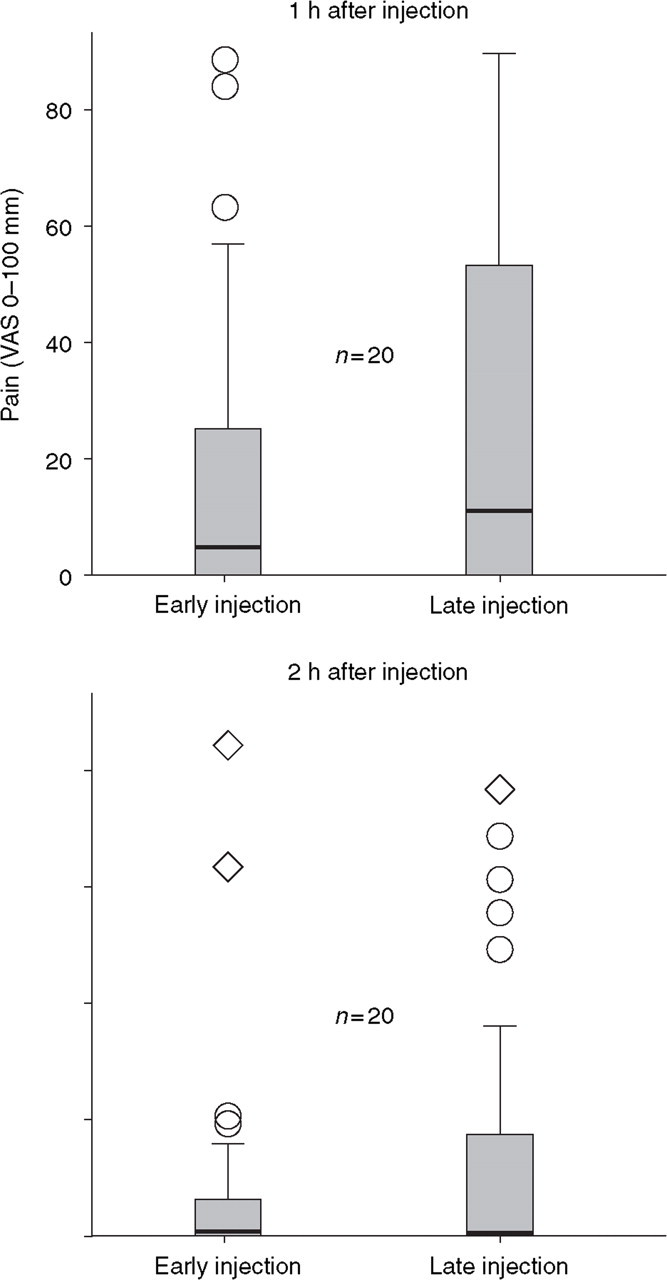
Pain severity at 1 and 2 h after injection in 40 attacks treated early vs. 40 attacks treated late (n = 20). Plots give the interquartile range (boxes), median (line within boxes), the highest and lowest values excluding outliers and extreme values (whiskers), outliers (open circles) and extreme values (rhombi).
As seen in Fig. 3, the mean time from injection to pain free was 1.5 h (median 1.2, range 0.2–5.2 h) after early treatment with sumatriptan, compared with 1.78 h (median 1.5, range 0.2–6.0 h) following late treatment. No significant difference was found in this respect between the early and delayed sumatriptan sequences (P = 0.22, paired samples t-test). Forty per cent (8/20) of patients did not have a shorter mean time to pain free in the attacks treated early compared with those treated late.

Mean intra-individual time from injection to freedom from pain in 40 attacks treated early vs. 40 attacks treated late (n = 20). Error bars show 95% confidence intervals of means.
In the majority (65%, n = 14) of patients the pain intensity at injection divided by time from injection to pain free after first injection (a theoretical approximation of the mean velocity of improvement) was superior in attacks treated late (median 63 VA steps/h) compared with early (median 45 VA steps/h). This difference was not statistically significant (P = 0.26, sign test).
There was no significant difference (P = 0.50, sign test) in the mean intra-individual AUC from first injection to pain free between attacks treated early and late. In eight patients (40%), the mean AUC from first injection to pain free was lower in the attacks treated late. The median AUC from first injection to pain free was 3.0 AUC units (mean 4.9, SD 5.9, range 0.5–26.0 units) after early treatment and 5.8 AUC units (mean 8.0, SD 6.3, range 1.5–21.9 units) after late treatment. The data are shown in Fig. 4.

Mean intra-individual ‘pain–time area under the curve’ (AUC) from injection to pain free in 40 attacks treated early vs. 40 attacks treated late (n = 20). One AUC unit is equal to 10 visual analogue scale steps multiplied by 1 h. Plots give the interquartile range (boxes), median (line within boxes), the highest and lowest values excluding outliers and extreme values (whiskers), outliers (open circles) and extreme values (rhombi).
Fifteen patients (75%) experienced at least one headache recurrence during the four attacks. The mean number of recurrences in attacks treated early was 0.9 (SD 1.2, range 0–5), compared with 1.1 (SD 1.0, range 0–5) in attacks treated late. Data are shown in Fig. 5. Only 35% (n = 7) had a higher incidence of recurrence in the attacks treated late. No significant difference was found in the number of recurrences in attacks treated early compared with late (P = 0.60, paired samples t-test).

Mean intra-individual number of recurrences after pain free in 40 attacks treated early vs. 40 attacks treated late (n = 20). Error bars shoe 95% confidence intervals of means.
More than one injection of sumatriptan was used in 15 (38%) of the attacks treated early and in 17 (42%) of the attacks treated late. Additional rescue medication was used in six (15%) of the attacks treated early and in seven (18%) of the attacks treated late, with an equal distribution of analgesics and triptans between the two treatment strategies.
At the end of the study, the majority of the patients claimed that their medication was equally effective when given early as late in the course of the attack (data shown in Fig. 6).

Distribution of participants concerning which treatment strategy was perceived as most effective.
One patient (designated S:020, demographic data described above) developed a status migrainosus as a result of both the attacks treated late (second and third in order). In the two attacks treated early, the times from first experience of migraine headache to first injection were 2.2 h and 3.0 h, respectively. In the two attacks treated late, the times from first experience of migraine headache to first injection were 48.3 h and 19.2 h (both developing into status migrainosus). The pain intensities (VA steps) in the four attacks were at the time of injection/1 h later/and 2 h later: 89/60/37 (first attack treated early), 80/65/50 (second attack treated early), 90/86/80 (first attack treated late) and 82/78/73 (second attack treated late). The registered times to pain free from injection were 41.8 h and 66.0 h, in the two attacks treated early. The times from injection to pain free (excluding time from first experience of migraine headache to injection) were retrospectively reported as 5 days and nights in both the attacks treated late. No attack was treated with further injections of sumatriptan or additional rescue medication. In the first (but not the second) of the attacks treated early, one recurrence was documented.
Discussion
The principal goal when designing the protocol was to investigate the consequences of the longest possible delay (mean 6 h) to treatment in a setting with high external validity (applicability to real-world clinical care). In addition, this eventually led to significant and large differences in pain intensity at the time of injection. In earlier clinical trials, treatment at mild intensity has been used as an indication for early treatment without compelling support (28–33). But because there is no simple relationship between the point of time and pain intensity in the natural course of still untreated migraine attacks (M.L. and C.D., data on file), it is dangerous to draw conclusions from such analyses in line with the hypothesized superiority of early treatment (30, 43). In order to improve data validity, a high-resolution 100-point VA scale was developed and used for assessment of migraine attack symptoms in the present study.
The presently studied group is comparable to the source population in terms of additional drug therapy, since 15% (n = 3) were using prophylaxis. Concomitant therapy is undesirable in early studies of new drugs for acute migraine treatment, but since one can not simply apply efficacy data from such unusually homogeneous research settings to common method of practice, it is logical to confirm and compare the expected advantages of different strategies of acute treatment in a naturalistic and representative population. Due to the design of the protocol, it was also hard to recruit the necessary number of patients. For these two reasons we decided not to limit the population by excluding patients using prophylaxis. Registrations of efficacy were performed before additional acute medication. The results of a second injection (if any) are therefore not reported. The observed results for pain-free at 2 h (55% and 62%) are higher than those reported for randomized controlled trials (RCTs) (44). This is expected, since the study was performed on selected responders.
The main finding of this study is that a non-oral formulation of a triptan does not necessarily have to be administered early in the migraine attack to be effective. Although a smaller change in pain was usually required to reach pain free when attacks were treated early compared with late, 40% of the patients did not document a superiority of early injections in this principal parameter. In the same percentage of participants, the mean AUC from first injection to pain free was lower in the attacks treated late, although the mean pain intensity at injection was higher. This and the approximation of the mean velocity (VA steps per hour) of improvement should, however, be interpreted with caution, since the meaning of absolute reductions in symptom scale scores depends upon baseline pain intensity (45). It cannot be concluded that early treatment has an equivalent effect to delayed treatment, but it is the authors’ opinion that if there were a clinically relevant difference, it would have been revealed. Most importantly, only one-third of the patients rated their overall impression of early injection as more effective.
It is likely that the discrepancy between our present findings and the analyses of trials on oral triptans is mainly a pharmacokinetic phenomenon attributable to the increasingly disturbed gastric emptying during attacks of migraine, leading to a compromised uptake of oral triptans when pain is severe. There is a risk of erratic absorption of any drug taken orally during a migraine attack, and it appears as if triptans per se can delay gastric emptying even further by activating 5-HT1 receptors on gastric myenteric neurons. Sumatriptan has, for example, been demonstrated to cause a relaxation of the fundus of the ventricle, reflected by an intragastric volume increase (7, 46–48).
Independently of the effect of early treatment, it is interesting that the rate of pain free after late treatment was 62%. At that point in time, a majority of patients can be expected to have developed allodynia (23). In this study, allodynia was not assessed using proper techniques (quantitative sensory testing, QST). Accordingly, it is not known if the lack of a significant difference in effect between early and late treatment is an expected finding because patients were non-allodynic. The study should be repeated on allodynic patients only, continually tested in the clinic for absence or presence of allodynia during different phases of migraine attacks and treatment, a project for which we do not have the necessary resources. As described in Results, a comparatively old patient with a long time to response in the two attacks treated early developed a status migrainosus in both the attacks treated late. She experiences allodynia in the scalp during her usual attacks as well as paraesthesias after injection of sumatriptan. This exceptional outlier supports the notion that in certain patients neuronal sensitization is a cause of a decreased responsiveness to triptans taken late during migraine attacks, and that cutaneous allodynia during the usual attacks may in some cases be used as an indicator of this potential problem (23). It has also been demonstrated that triptans per se may induce cutaneous allodynia (49). It is interesting to speculate that the rate of recurrence would be higher due to neuronal sensitization if the initial migraine headache were not treated early (32). But since delaying the time of subcutaneous treatment had no significant influence on the incidence of migraine recurrence in the present study, this remains a hypothesis.
In a large (n = 912) study of zolmitriptan (5 mg) administered as a nasal spray, patients were allowed to treat a single migraine attack at any intensity and at any time. It was found that the active drug was superior to placebo at all time points. In severe migraine the 2-h headache response was 59% for zolmitriptan vs. 12% for placebo (50). The favourable characteristics of late compared with early subcutaneous triptan injection may thus also, although to a lesser extent, apply to nasal spray formulations, a hypothesis that is being investigated in our clinic.
Delaying the time of treatment of a headache with subcutaneous sumatriptan by 4 h had no influence on efficacy or on incidence or timing of headache recurrence in the earlier and larger RCT (GSK, data on file). A further delay of about 2 h in the present study did not change the outcome between early and late treatment with subcutaneous sumatriptan. Data from studies with methodological differences can not easily be compared, but the point is that no significant difference was found in any efficacy parameter in either of the two studies. Along the same lines, it was recently reported that subcutaneous sumatriptan 6 mg is an effective migraine treatment in patients who historically fail to respond to oral triptans, and that the association between the presence of allodynia and lack of pain-free response may be overcome by the use of subcutaneous sumatriptan (51, 52). It should be mentioned that the presence of allodynia in the latter study was not confirmed by QST. In general, however, it can thus be concluded that a nonoral formulation of a triptan does not have to be administered early during the migraine attack to provide efficacy, and that most migraineurs are not unresponsive to delayed treatment with a triptan as a result of central sensitization. It is reassuring for patients to be informed of the fact that it is worthwhile taking their medication even if they have not managed to do so early in the course of an attack.
Footnotes
Acknowledgements
We thank Martin Gellerstedt for valuable statistical comments during the preparation of the manuscript. The study was financed by the Gothenburg Migraine Clinic with no external monetary support. In March 2004, the study was presented in Madrid and subsequently received the Migraine Innovators Award (sponsored by Astrazeneca) on behalf of the faculty.
