Abstract
The aim was to investigate the association between temporomandibular disorders (TMD) and overall muscle tenderness, depressive symptoms, sleep difficulties, headache frequency and related symptoms in children with primary headache in comparison with controls. Based on an unselected population sample of 1135 Finnish schoolchildren classified according to the type of headache at age 12, altogether 297 children aged 13-14 from different headache groups and healthy controls were randomly selected for an interview and clinical examinations. Children with migraine had more TMD signs than children with nonmigrainous headaches or healthy controls. High TMD total scores were associated with palpation tenderness in other parts of the body and with frequent headache attacks. We conclude that children with overall headache, migraine in particular, and high total TMD scores showed an increased overall tenderness to muscle palpation and multiply manifested hypersensitivity pain.
Introduction
Children suffering from headache are more sensitive than other children to pain in general and to several other types of pain, such as neck, back and abdominal pain (1). It has also been reported that children with widespread musculoskeletal pain have higher scores on emotional and behavioural problem scales than control children (2). Especially children with migraine have more depression (3–5) compared with children with nonmigrainous headache or headache-free controls. Previous studies have indicated that they also have muscle tenderness in the pericranial and neck-shoulder region, earache and pains of psychosomatic origin (6, 7) In adults, depression and anxiety disorders are associated with migraine (8).
Temporomandibular disorders (TMD), also called craniomandibular disorders (CMD), represent a group of painful conditions involving muscles of mastication and temporomandibular joints. Among the symptoms of TMD, pain is the most commonly reported (9) and hope of pain relief is the most common reason for patients to seek treatment (10). Patients with TMD frequently suffer from pain in the neck and shoulder area. An association between TMD and subjective neck symptoms has been shown in typists Keyboard operators exposed to cervicobrachial stress (11). Adult patients with signs of TMD have impaired mobility of cervical spine and tenderness on palpation in the neck-shoulder muscles, as well as pain sites in other parts of the body (12–17). Patients with TMD have lower pain threshold and pain tolerance when compared to subjects without TMD (18). They also report more pain (19). Patients with TMD of myogenous origin report stress, depression, disability and dysfunctional illness (20).
Preschool-aged children with headache have been found to have more bruxism, tenderness in the occipital muscle insertion areas and tenderness in temporomandibular joint areas compared with headache-free children (21). There are only few reports about the association of TMD with overall pain and stress conditions in children (22–30).
In our previous report, children with migraine tended to show more signs of TMD compared to children with tension-type headache or healthy controls (31). Because signs and symptoms of TMD occurred in each headache group, we wanted to investigate other features associated with both headache and TMD. Our aim was to study the association of TMD with tenderness of neck, shoulder and other muscles, and with depressive symptoms and sleep difficulties in children with headache.
Subjects and methods
In 1998, a total of 1409 12–13-year-old school children in the sixth grade in all of the 35 Finnish-speaking primary schools in the city of Turku, South-Western Finland, were chosen as the original study population. Altogether 1135 (81%) pupils returned the questionnaire which they had willingly filled in with the help of their guardians.
Questions included the occurrence and features of headache during the preceding 6 months. Headache intensity was expressed by a trichotomic scale: mild, moderate or severe. The list of associated features included vomiting, nausea, loss of appetite, photophobia, phonophobia, pain aggravation due to physical activity, insomnia, hypersomnia, recurrent abdominal pain, and sensation of fainting. Sleep difficulties were asked about in more detail. The criteria of the Committee of the International Headache Society (IHS, 1988) were used for the headache classification (32).
In 1999, 70 children from each primary headache group (migraine; migraine-type not fulfilling the IHS criteria; tension-type headache; tension-type not fulfilling the IHS criteria) and from healthy controls were randomly selected. If the headache group included fewer than 70 children, as in the case of tension-type headache children not fulfilling the IHS criteria, the entire group was included in the study population. As a result, altogether 310 children aged 13–14 years (mean 13.4 years) participated in a face-to-face interview and in a clinical examination by a physician and a trained physiotherapist at the paediatric outpatient clinic in the city of Turku. A total of 297 children, 146 girls and 151 boys, came to the Institute of Dentistry, University of Turku, for a thorough stomatognathic examination.
Children were asked to complete the Children's Depression Inventory (CDI), a validated instrument for detecting depression (33). The final headache diagnoses were based on a face-to-face interview, which in some cases affected the classification.
Clinical examination
The examination by a named physiotherapist included manual palpation of muscle tenderness in the neck-shoulder area on seven bilateral pericranial and neck-shoulder tender points (the frontal and temporal muscles, insertion of the suboccipital muscle, anterior aspect of C5-7, midpoint of the upper trapezius muscle, origin of the supraspinatus muscle, insertion of the levator scapulae muscle). A modified version of the Total Tenderness Score System (34) was applied. Reaction to the pain was classified into four categories: 0 = no pain, 1 = no visible reaction but reports mild pain, 2 = reports pain, distorting the face, 3 = reports considerable pain and withdraws. The total score was used in the final analyses. The Fisher dolorimeter (35) was used to measure the pressure pain threshold of five bilateral points (the frontal and temporal muscles, insertion of the suboccipital muscle, midpoint of the upper trapezius muscle, insertion of the levator scapulae muscle). The supraspinatus muscle and anterior aspect of C5–7 were excluded because of technical difficulties in measurement. The rubber tip of the instrument had a surface of 1 cm2, and pain was measured in kg/cm2 units. The range was 0–10 kg. The dolorimeter was placed on a selected point and pressure was increased at a rate of 1 kg/s. The child indicated verbally when he or she sensed a change from pressure to pain. Results were expressed as a mean dolorimetric score of 5 bilateral points for the final analyses. Both manual and dolorimetric recordings were also made on three bilateral fibromyalgic points: elbow 2 cm distal to the lateral epicondyle, medial fat pad of the knee proximal to the joint line and upper outer quadrant of buttock in anterior fold of the muscle. The examinations were conducted between autumn 1999 and spring 2000. A two-week interval was kept between the medical and stomatognathic examinations.
Stomatognathic examination
The children were interviewed, examined clinically and scored for TMD signs as described previously (31). Before the study began, the examiner was trained by experienced clinicians at the Institute of Dentistry. The stomatognathic examiner was blinded to the results of the earlier examinations and the interview. In the interview, questions about joint sounds, jaw mobility, pain while chewing, sensation of fatigue in the jaw, pain or sensation in the ear or throat, and parafunctional habits, such as bruxism and nail biting were asked about. The stomatognathic examination included measurements of mandibular movements and recordings of joint sounds using a stethoscope. Pain, locking or luxation of the joints during movements were examined. The temporomandibular joints were palpated laterally and posteriorly. The following masticatory muscles were palpated bilaterally: the anterior and posterior portions and the insertion into the coronoid process of the temporal muscle, the deep and superficial portion of the masseter muscle, the posterior portion of the digasticus muscle, and the medial and lateral pterygoid muscles. If the subject did not react to palpation, no tenderness was recorded. Mild tenderness was present if the subject verbally reported any pain during palpation. Moderate tenderness gave rise to blink reflex and severe tenderness to withdrawal. The clinical signs were scored from 0 to 2 for every muscle, depending on the degree of tenderness. Joint tenderness was graded depending on whether it was lateral, posterior or both. One point was given to each joint if any sound was registered. Difficulties in guiding the mandible into the centric relation (CR) or pain recorded during this manipulation scored one point. When these points were added together (maximum 35) the child was given a score for the severity of TMD signs (31).
The overlaps in the clinical examinations of the physiotherapist and dentist regarding the frontal and temporal muscle were carefully excluded before the analyses. Results of the palpation of the lateral pterygoid muscles were excluded from the analyses due to difficulties in obtaining reliable observations. The one subject with chronic tension-type headache was excluded from the analysis. Nausea and/or vomiting were asked about as a criterion for migraine in the classification of different headache types. However, in another analysis it was combined with fainting sensation since they both reflect dysfunction of the autonomic nervous system.
Statistical methods
The TMD sum score was divided into three categories before the analysis due to high skewness in the distribution. The children were scored as healthy regarding TMD if the sum score was zero, having very mild TMD when the score was between 1 and 4, and mild or moderate TMD when the score was five or more (31). The analysis of associations between the TMD classification and other variables was carried out using cumulative logistic regression analysis, which takes into account the ordinal scale in a polytochomous response variable (36). In addition to the P-value, the results of the analysis were quantified by cumulative odds ratios (COR) and 95% confidence intervals (95%CI). Gender and headache-adjusted analyses were carried out separately for each studied variable. Two multivariate analyses were done: the first one with variables recorded from all subjects, and the second one with variables recorded only from headache subjects. The statistical computations were performed with the SAS System for Windows, release 8.2/2001.
Results
Girls had significantly more signs of TMD compared to boys (P = 0.024, COR = 1.7, 95% CI = 1.07–2.66) (Table 1). After adjusting for gender, the overall difference between the headache groups and TMD signs was not statistically significant (P = 0.115). However, when the headache groups were compared separately with healthy controls, children with migraine and migraine-type headache had significantly more signs of TMD (COR = 2.1, 95% CI = 1.1–4.2 for migraine and COR = 2.2, 95% CI = 1.1–4.2 for migraine-type headache).
Gender, characteristics of headache and associated factors, palpation and dolorimetric values, depression and sleep difficulties in different temporomandibular disorders (TMD) groups
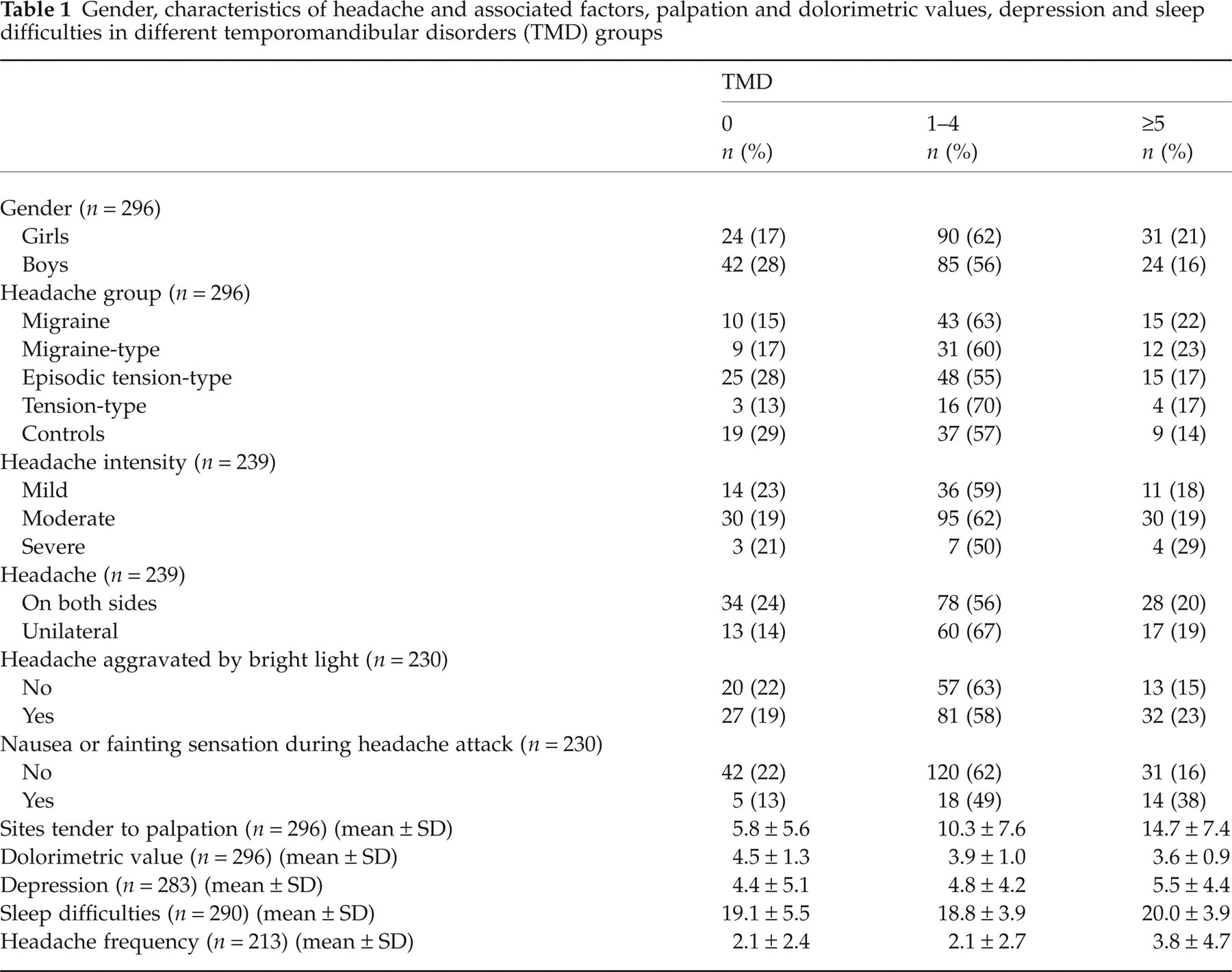
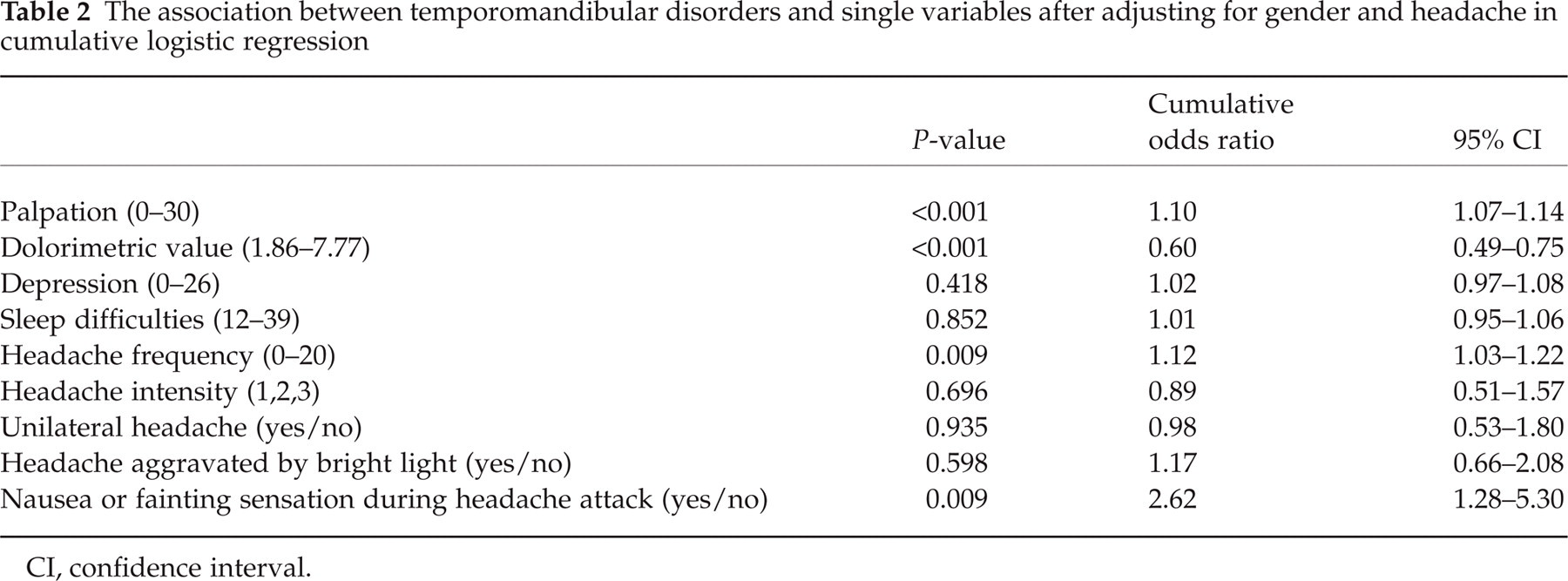
After adjusting for both gender and headache, TMD signs were significantly associated with palpation tenderness, dolorimetric values in the neck and shoulder area, as well as in the whole body, headache frequency and nausea or fainting sensation during headache attacks (Table 2).
The association between temporomandibular disorders and single variables after adjusting for gender and headache in cumulative logistic regression
CI, confidence interval.
When the variables were analysed together after adjusting for gender and headache, the strongest association was seen between TMD signs and tenderness to palpation. Dolorimetric values were also associated with TMD. The association was nonsignificant with depressive symptoms, sleep difficulties, headache intensity, unilateral headache, headache aggravated by bright light, nausea or fainting sensation during headache attack, and headache frequency (Table 3).
The associations between temporomandibular disorders and variables in multivariate cumulative logistic regression analysis. The analysis is adjusted for gender and headache

COR, cumulative odds ratio CI, confidence interval.
Discussion
Our results showed that children with TMD had other painful body areas, which is in accordance with earlier studies on children (29) and studies on adult TMD patients (12–17). A correlation was observed between tender sites to palpation and dolorimetric values in our study, indicating that the more sites tender to palpation the child had, the lower the dolorimetric value and pain threshold. As stated by Davidoff (37), trigger points are essential in myofascial pain which is often found in headaches, fibromyalgia, TMD and occipital neuralgia. Hyperalgesic trigger points may be involved as an exclusive source of pain when central changes in nociceptive stimuli from muscles may interact with peripheral myofascial nociceptive mechanisms (38, 39).
In children, earlier studies have reported an association between overall headache and TMD (40, 41). The overall differences between the headache groups in our study were not significant but the results were still in line with the previous studies. Contrary to adults (42–45), migraine seemed to have stronger association with TMD signs than had tension-type headache. This is in line with the hypothesis that acute migraine attacks can specifically facilitate trigeminal nociceptive system (46). One factor that can influence comparison with earlier studies is the different headache criteria used. In our study, the earlier IHS criteria from 1988 were used during the whole study period. Our results also showed that headache frequency was associated with both TMD and palpation tenderness and dolorimetric values, which represent overall pain sensitivity. In the multivariate cumulative logistic regression analysis, these variables overran the effect of headache frequency.
The observed gender difference in children both for TMD and headache have been controvresial in earlier studies (28, 40, 41, 47–49). Our previous analysis included a tendency towards gender difference (31). Earlier studies in adults have shown that women have more TMD signs and treatment need than men (50, 51). The same is true for headache (52).
The association between TMD and depression has been shown in many earlier studies in adult TMD patients (53–55) and in children (29), but in the present study, no significant association between depression and TMD was observed. There are also studies showing no difference between patients with TMD, other pain and controls regarding personality type, response to illness, attitudes towards health care or ways of coping with stress (56). There is an ongoing debate about the cause-effect relationship of pain and behavioural or emotional changes.
Conclusions
Children with temporomandibular dysfunction and overall headache, especially migraine, show a widespread distribution of pain, probably indicating an increased susceptibility to pain in general and hyperactivity to pain with multiple symptoms. Clinicians treating children with headache should also take temporomandibular dysfunction into consideration. TMD should always be considered when headache is associated with earache, difficulties in opening the mouth, fatigue, or stiffness of jaw and tenderness of masticatory muscles.
