Abstract
Much research on visual functions in migraine has pinpointed the existence of abnormal visual processing between attacks. However, it is not clear if this is due to cortical hyper- or hypoexcitability. We aimed to clarify this issue by comparing motion perception thresholds of subjects with migraine with (MA) or without aura (MoA) and control subjects. Two types of dot kinetograms were used: in the first experiment coherently moving dots were presented in an incoherent environment, while in the second only coherent motion was seen. Subjects with migraine displayed significantly impaired motion perception compared with controls when they had to detect the direction of the coherently moving dots in an incoherent environment, while they were slightly better in a direction discrimination task, where only coherent motion was presented. This pattern of results is comparable to those achieved by an external excitability enhancement of V5 induced in healthy human subjects in a former study of our group. According to this, a cortical excitability enhancement can result in an impaired focusing on a given signal against a noisy background, but improves perception of non-ambiguous stimuli. Thus we conclude that migraineurs display enhanced visual cortical excitability between attacks in V5.
Introduction
Various aspects of visual processing have been investigated in subjects with migraine due to the visual disturbances that may precede or accompany the headache and to the fact that visual stimuli can trigger migraine. The common aim of these studies was to identify differences between migraineurs and controls and relate the findings to the pathophysiology of the disease. Additionally, it was suggested by several studies using a wide range of tasks that migraine patients may have visual cortical anomalies between attacks. More intense illusions to grating patterns (1, 2), as well as greater visual masking effects (3), reduced perceptual suppression of simple targets (4, 5) and shortened motion after-effect durations (6) were observed by different studies. Differences in visual cortical function between migraineurs and controls have also been revealed using transcranial magnetic stimulation (TMS). Lower phosphene thresholds (PTs) (7–10) even after 1 Hz repetitive TMS (rTMS) were detected (11). By contrast, other studies have found lower prelevance of phosphenes and increased PTs in migraineurs (12, 13). Electrophysiological studies have also implied abnormal primary visual processing by measuring the amplitude of visual evoked potentials (14, 15). From these studies an altered interictal excitability of the cerebral cortex in migraine, most pronounced in the visual areas, was proposed.
It has been suggested that in migraine patients not only the primary visual cortex but also extrastriate areas, including the human motion sensitive area, called V5 or MT, display altered excitability (16). Between migraine attacks, lower moving PTs and subjectively more vivid phosphene perceptions were detected in migraineurs than in control subjects when V5 was stimulated, which could be caused by cortical hyperexcitability. However, the physiology of stationary and moving phosphenes is not well known in detail. Recent studies have reported that the perception of moving PTs could be changed by the modulation of V1 activity alone (17, 18) and thus the change in phosphene perception could be due to an altered excitability of V1.
A recent study using motion coherence perimetry has found a decreased ability to detect coherent motion in a random dot paradigm in most but not all of the migraine patients tested (19). The authors proposed that the level of neuronal noise increased in the cortex due to the interictal neuronal hyperexcitability in patients and this interferes with performance on this task. However, the performance impairment of the patients could also have been caused by a substantial general excitability decrement, which would weaken primary visual perception.
The aim of the present study was to specify if the performance deficit described by this previous study (19) was caused by a noise increment due to cortical hyperexcitability or by a general perceptual deficit which may be caused by cortical hypoexcitability in migraineurs. We have used two types of dot kinetograms, which are frequently applied to measure motion perception threshold (20–23). We compared motion perception with and without distractors: in the first task the coherent motion was presented in an incoherent environment (motion-perception task with distractors), while in the second task only an up or down motion was presented, without incoherently moving dots (motion-perception task without distractors). In a previous study using these stimuli we applied cathodal or anodal direct current stimulation (tDCS) in order to modulate the excitability of V5 during the performance of both paradigms (24). tDCS influences cortical excitability both during and after stimulation in a polarity dependent way: weak cathodal stimulation decreases cortical excitability by membrane hyperpolarization, while anodal stimulation enhances it by subthreshold membrane depolarization (18, 24–28). The duration of the induced after-effects depends on the stimulation duration. Using the above-mentioned dot kinetograms, an increment of cortical excitability of the left V5 by anodal stimulation improved the subject's performance in a distractor-free task, while perception was worsened by the excitability-diminishing cathodal stimulation. Interestingly, in the task with distractors, the effect of stimulation was partly reversed: a diminution of cortical excitability by cathodal stimulation improved the motion perception of the coherent motion if this was presented among distractors, while anodal stimulation had no significant effect. These different effects are probably caused by a ‘focusing effect’ of cathodal stimulation: in the task with distractors, not only the encoding pattern of correct motion direction, but also some suboptimum neuronal patterns encoding non-correct motion directions are activated – to a certain degree simultaneously (24). This process probably results in a kind of ‘fuzzy’ activation state of the encoding neuronal pattern in response to different motion directions. In this case, cathodal stimulation may have focused the correct perception of these parameters by decreasing the global excitation level and thus diminishing the amount of activation of concurrent patterns below threshold, while the originally highly activated neuronal pattern encoding the correct direction remains above threshold. By this kind of modulation, cathodal stimulation could improve the perception of the ‘correct’ motion.
We hypothesized that if the area V5 is hyperexcitable in migraineurs, their performance may be similar to the performance of healthy human subjects after an externally induced cortical excitability increment by anodal stimulation. Thus, their performance would be improved in a distractor-free condition, as was found after anodal stimulation; conversely, in the paradigm with distractors they should show no change in performance. Alternatively, if the excitability enhancement is sufficiently strong in migraineurs to de-focus activation in the task with distractors, we would expect worse performance compared with healthy controls. On the other hand, cortical hypoexcitability would decrease performance in a distractor-free task due to decreased activation of the respective motion-encoding cortical representation. For the task with distractors, moderate hypoexcitability could result in an improvement of performance, due to a focusing effect, like after cathodal stimulation, or impair it, if the hypoexcitability is prominent enough to result in a substantial activational deficit of all of the motion encoding patterns.
Methods
Subjects
The study involved 20 subjects with migraine (mean age 27.5 years, range 22–52 years, SD 7.2; four men, 11 with aura) and 20 control subjects (mean age 28.2 years, range 20–56 years, SD: 9.5; six men). All of the subjects had a visual acuity score better than 0.9 measured by Schnellen charts and gave their written informed consent according to the Declaration of Helsinki. The study was approved by the Ethics Committee of the University of Göttingen.
Migraine was diagnosed by expert neurologists according to the International Headache Society criteria (in Cephalalgia, 2nd edn, 24:2004). Subjects with migraine were not on prophylactic medication. Nine patients suffered from migraine only of the right side, six of the left and five of both sides. The experiments were conducted in headache-free intervals. The last migraine attack occurred on an average 61.8 days before the experiment (range 1–90 days). None of the subjects had a migraine attack within the following 3 days after the experiments. The frequency of the migraine attacks was between 4 and 50/year (mean 20.7). There were various sorts of medication that were preferred by the subjects in case of a migraine attack: rizatriptan, four subjects; sumatriptan, five subjects; acetylsalicylic acid, two subjects; paracetamol, two subjects; ibuprofen, two subjects; acetylsalicylic acid, paracetamol and caffeine, one subject. The experimenter was blinded to the the diagnosis of the subject at the time of testing.
Protocol
Subjects were comfortably seated in a semidarkened room at a distance of 75 cm from a monitor. Random dot kinetograms were generated using a standard VisionWorks system (VisionWorks; Vision Research Graphics, Durham, NC, USA) and presented on a colour high-resolution monitor. The steady-state luminance of a stationary dot was 10 cd/m2, while the background luminance was 2 cd/m2. In the first experiment, a single-interval, forced-choice, motion-direction discrimination task was used. Subjects had to report the direction (up or down) of coherent motion in a 10° × 10° random dot stimulus by pushing the suitable button on a computer mouse. The middle of the stimulus was placed 10° away from the fixation point on the left side of the screen, to stimulate the left hemifield more. In the first experiment, the presentation time was 72 ms and the stimuli contained 300 white square dots, the dot speed was 5°/s. The diameter of one dot was 3 pixels. The direction of the coherent motion was randomly varied between up and down. At the beginning of the first experiment, 40% of the dots were moving coherently, the remaining 60% randomly in different spatial directions (no noise dots were moving in exactly the same direction as the signal dots). If the dots moved outside of the 10° × 10° window, they were randomly re-plotted within the window for the next frame. After two consecutive correct or incorrect responses the percentage of coherently moving dots was decreased or increased by 4%, respectively. After two staircase reversals, the step size decreased to 1%. The trial was terminated after six staircase reversals. The use of six staircase reversals conforms to standard practice in psychophysical experimentation. In this way, the lowest percentage of coherently moving dots needed to identify a direction (i.e. motion perception threshold) was determined. The trial was repeated three times, and the average duration of the experiment was about 3 × 5 min.
In the second experiment, the method of constant stimuli was used. The presentation time was 48 ms, which was used in a previous experiment which included healthy subjects (24). The stimuli contained 200 dots, all moving coherently. In each block, 70 stimuli were presented and the dot speed was 5°/s. The subject's task was to identify the direction (up or down) by pushing the appropriate mouse button. In this task the number of correctly identified directions was counted. The average reaction time (RT) was also determined. The trial was repeated three times, and the average duration of the experiment was about 3 × 4 min.
Statistical analysis
The percentages of coherently moving dots at threshold or the number of correctly identified directions were entered into a between-subject factor
Results
Coherently moving dots with distractors
The mean motion perception threshold was 37.0% (SD 9.86) in controls, 63.65% (SD 11.82) in MA and 59.0% (SD 7.2) in MoA. The
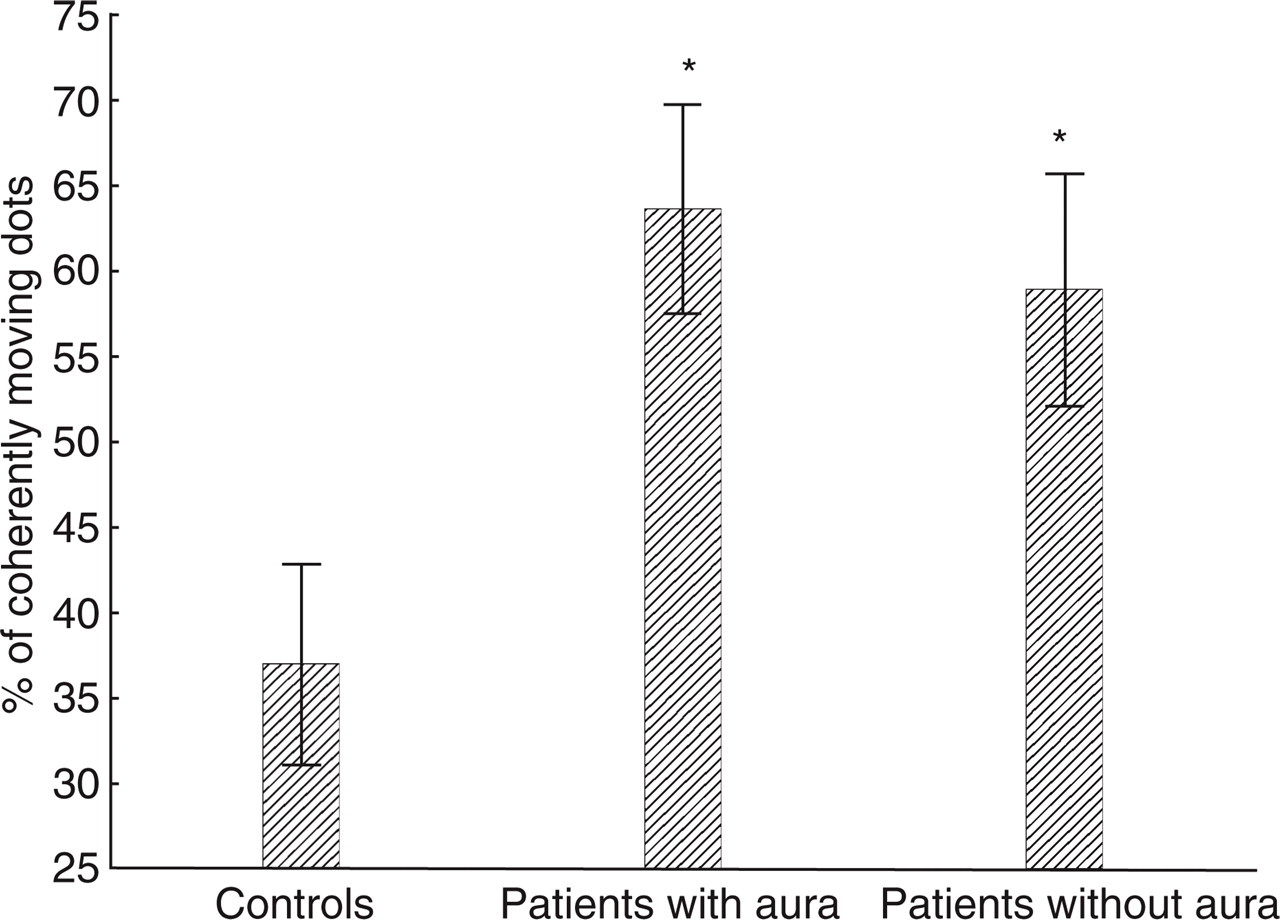
Motion perception thresholds (the lowest percentage of coherently moving dots needed to identify a direction) of control subjects and migraine patients when subjects were asked to detect the direction of coherent movement among randomly moving dots. Error bars represent SDs. Asterisks show significant effects.
Coherently moving dots without distractors
The mean percentage of correct responses was 81.1% (SD 12.2) in controls, 93.65 (SD 6.6) in MA and 89.76 (SD 7.0) in MoA from the total of 3 × 70 trials. The

Percentage of correct responses of the subjects when only coherent motion was presented. Error bars represent SDs.
Reaction times
The mean RT in the distractor-free task was 550 ms (SD 114.7) in controls, 563.5 ms (SD 159) in MA and 534.9 ms (SD 108.6) in MoA. There were no significant differences between the three groups (Fig. 3).

Reaction times of the subjects. Error bars represent SDs.
Relationships between performance and clinical parameters
There was no correlation between the performance of the subjects and the duration (task with distractors: r =−0.03; task without distractors: r = 0.11) or frequency (task with distractors: r = 0.13; task without distractors: r =−0.02) of the migraine attacks or the time between the last migraine attack and the date of the testing (task with distractors: r = 0.11; task without distractors: r =−0.54).
Discussion
In this study the performance of migraineurs differed significantly from the control group with regard to motion perception: first, migraineurs showed better performance in the task where only coherent motion was present. These results are identical to a previous study in which an externally induced cortical excitability enhancement improved motion perception in an elementary motion direction identification task when the motion was uniform (24). Here, increased excitability should elevate the activation of the respective motion-encoding cortical representation and thus improve perception. In the case of hypoexcitability in migraineurs, the performance should have been worsened because of the impaired primary visual processing caused by decreased activity of the respective motion-encoding representation. Second, with regard to the task in which the direction of coherent motion needed to be identified in an incoherent environment, the motion perception thresholds in subjects with migraine were higher compared with control subjects, showing that a higher percentage of dots was necessary for them to see a pattern as coherent. The latter results are probably caused by a ‘de-focusing’ effect of the enhanced excitability: when more than one neuronal motion-encoding pattern is active and their activities are above threshold, a generally higher cortical excitability could impair the signal-to-noise ratio and therefore worsen motion-discrimination performance in a task in which the target is presented with distractors. In contrast, moderate hypoexcitability would have resulted in an improvement of performance due to a focusing effect, as shown in the cathodal stimulation condition of a previous study (24). Alternatively, if the hypoexcitability was prominent enough to result in a substantial activational deficit of all of the motion encoding patterns, it could also have impaired performance. However, according to the pattern of results with regard to both tasks and previous studies (19), it is highly probable that migraine patients differ in performance from healthy subjects due to cortical hyperexcitability.
Using a similar but not identical random dot paradigm, a higher motion perception threshold was observed in MA subjects (19, 29). Besides motion threshold measurements, several studies have investigated low- and high-level visual tasks in subjects with migraine (30–32). Migraineurs were better in low-level (orientation detection, temporal order judgement) but not in higher level visual processing (visual priming), suggesting a hypersensitivity of the primary visual but not of the extrastriate visual areas (30). However, a later study using the same type of tasks has failed to replicate these results (32). Additionally, a recent study has observed that 1 Hz rTMS, which has an inhibitory effect in healthy subjects, speeded up the reaction times on illusory contour perception in migraineurs (31), supporting the view of a failure of inhibitory circuits in extrastriate areas. These partly contradictory results can be explained by the different difficulty levels of the applied tasks: in the first two studies, the error rates were high across the studies, indicating that subjects were responding at or near a chance level of accuracy. This suggests that subjects did not perform the task appropriately. Therefore, the reaction times were probably not the best reflections of the visuo-cognitive demands of the task. Besides these, the different results could also be due to the characteristics of the different types of tasks used by various studies: it is possible that using a ‘noisy’ task in which several neuronal encoding patterns in a given cortical area are activated results in impaired performance of migraineurs, while a distractor-free task improves performance in the same patient group.
The mechanisms that underlie the cortical hyperexcitability in interictal periods of migraineurs remain largely unknown and are probably multifactorial. Excessive excitation due to abnormal release of excitatory neurotransmitters is one possibility that is supported by the higher plasma level of glutamate in migraineurs (33). It was also observed that repeated episodes of cortical spreading depression can cause permanent selective suppression and/or damage of GABAergic inhibitory function (34). In accordance with this, a central GABA-agonist, sodium-valproate, is successfully used in migraine as a prophylactic treatment (35). Although the literature on cortical excitability in migraineurs is still controversial, these data suggest that the migraneous visual cortex is hyper- and not hypoexcitable, at least between attacks. Additionally, the hyperexcitability hypothesis could better explain the vulnerability of migraineurs to cortical spreading depression (36, 37), which is thought to be responsible for the migraineur's aura.
In our study we have found a modulation of motion perception, not only in MA subjects but also in MoA subjects. MA subjects did not differ significantly from MoA subjects concerning motion perception performances. This is in accordance with some other studies, which report no differences among migraine subgroups (6, 10, 13, 16, 38). In contrast, a few studies have found differences between migraine subgroups (5, 7, 12). It was suggested that in subjects with aura the cumulative physiological effects of many aura episodes and the occurrence of spreading depression (36, 37) may cause stronger ultrastructural changes in the visual system (4, 39) and therefore the visual changes observed in MA subjects are more pronounced that in MoA subjects. The group studied here consisted of younger subjects; it is possible that this is the reason for the absent group difference. However, it is very difficult to estimate total auras retrospectively, because migraine frequency and aura appearance fluctuate dramatically across years as a result of hormonal factors, life stressors and many other, as yet unknown factors. Finally, we did not find any correlation between the psychophysical performances of the subjects and the clinical parameters, including the duration of the disease, or the frequency and duration of the migraine attacks. Further investigations using a larger patient population would be necessary to examine these possible correlations.
In summary, we found altered motion perception in subjects with migraine between attacks compared with healthy subjects. The pattern of results supports the concept of interictal cortical hyperexcitability; however, considering the fact that cortical excitability undergoes dynamic changes in different subgroups of migraineurs, further studies are necessary to better define the precise nature of CNS dysfunction in this disease.
Footnotes
Acknowledgements
We thank Chris Crozier for the English corrections. This study was supported by the German Ministry of Research and Education (FKZ: 01EM0117).
