Abstract
Tanacetum parthenium (TP) is a member of the Asteracee family long used empirically as a herbal remedy for migraine. So far, however, clinical trials have failed to prove consistently the effectiveness of TP extracts in preventing migraine attacks, probably as a consequence of the uncertainty as regards the active principle. In this study, the biological effects of different TP extracts and purified parthenolide were tested in an animal model of migraine based on the quantification of neuronal activation induced by nitroglycerin. The extract enriched in parthenolide significantly reduced nitroglycerin-induced Fos expression in the nucleus trigeminalis caudalis. Purified parthenolide inhibited nitroglycerin-induced neuronal activation in additional brain nuclei and, significantly, the activity of nuclear factor-κB. These findings strongly suggest that parthenolide is the component responsible for the biological activity of TP as regards its antimigraine effect and provide important information for future controlled clinical trials.
Introduction
Tanacetum parthenium (TP), or feverfew, a member of the Asteracee family, is a phytomedicine that has become very popular since the 1980s in treating inflammatory conditions and migraine (1, 2).
Several clinical trials have tested the effectiveness of TP extracts in preventing migraine attacks (3–5), with controversial findings. This lack of consistency is probably related to two main factors: (i) uncertainty as regards the component responsible for its biological activity in the extracts, and (ii) qualitative and quantitative variability of components contained in the preparations used.
TP contains sesquiterpene lactones, flavonoid glycosides, pinenes and other compounds (6–8). Sesquiterpene lactones are chemical components with an α-methylenebutyrolactone unit as an integral part of their structure (Fig. 1). Parthenolide is the most predominant lactone, and it has been proposed as the component responsible for the biological activities of TP. Parthenolide inhibits platelet aggregation and serotonin release from platelet granules (9–12), lipopolysaccharide-induced nitric oxide formation and lipopolysaccharide-induced activation of the inducible isoform of nitric oxide synthase (iNOS) in rat microglial cells (13). Parthenolide has also proved effective in blocking the transcriptional activity of nuclear factor-κB (NF-κB) (14), a transcriptional factor involved in the mediation of pain and inflammation via the induction of proteins associated with the inflammatory response, such as adhesion molecules, cytokines, antioxidant enzymes and cyclooxygenase (15). While these findings suggest that parthenolide may be the relevant principle for the antimigraine activity of TP extracts, it is worth noting that several flavonoid glycosides contained in TP have shown vasodilating and anti-inflammatory activity (16, 17).

Effect of chronic treatment of vehicle upon Fos expression evoked by systemic nitroglycerin. Histograms represent the number of Fos-positive neurons counted in each nucleus (expressed as mean ± SD). ▪, Nitroglycerin alone intraperitoneally (i.p.) (group 1); □, nitroglycerin i.p. preceded by chronic vehicle administration (group 4);  , vehicle i.p. preceded by chronic vehicle administration (group 7). ∗P < 0.05 group 4 vs. group 1; ∗∗P < 0.05 group 7 vs. group 4. PVH, Paraventricular nucleus of the hypothalamus; SON, supraoptic nucleus of the hypothalamus; LC, locus coeruleus; PBN, parabrachial nucleus; NTS, nucleus tractus solitarius; NTC, nucleus trigeminalis caudalis.
, vehicle i.p. preceded by chronic vehicle administration (group 7). ∗P < 0.05 group 4 vs. group 1; ∗∗P < 0.05 group 7 vs. group 4. PVH, Paraventricular nucleus of the hypothalamus; SON, supraoptic nucleus of the hypothalamus; LC, locus coeruleus; PBN, parabrachial nucleus; NTS, nucleus tractus solitarius; NTC, nucleus trigeminalis caudalis.
In this study we therefore sought to evaluate whether parthenolide is actually the component responsible for the biological activity of TP as concerns its potential antimigraine activity. We tested extracts with a different content of parthenolide and purified parthenolide in a well-known animal model of migraine, based on the evaluation of nitroglycerin-induced changes in the rat brain (18–25). This model has been inspired by the observation that systemic administration of nitroglycerin induces migraine-like headaches in predisposed patients, but not in normal subjects (26–30). In previous experimental reports, we showed that administration of nitroglycerin activates a set of brain nuclei, which are involved in autonomic, neuroendocrine and nociceptive functions. This neuronal activation is maximal 4 h after the administration (18).
Materials and methods
Experiments were performed in male Sprague–Dawley rats (180–250 g body weight; Charles River, Calco, Italy), which were housed three to four per cage at 22 °C on 12-h light/dark cycle with food and water ad libitum for 6 days before starting the treatment.
The study was divided into two parts: part I was devoted to the comparison of the effect of two different extracts prepared by Indena S.p.A. (Milan, Italy) upon nitroglycerin-induced neuronal activation. The first extract was depleted of parthenolide (TPD, HPLC content 0.08%); the second one was enriched in parthenolide (TPE, HPLC content 26%); part II consisted of the evaluation of the effect of purified parthenolide, extracted from TP (Indena S.p.A) (HLPC content 98%) in the same experimental model.
A total of 78 rats were evaluated, subdivided into groups of six to eight animals each.
The experimental protocol was approved by the Ethics Committee at the IRCCS Neurological Institute C. Mondino Foundation.
Preparation of extracts
Fresh TP were extracted with methanol. The methanolic phase was then submitted to further purification for the preparation of TPE and purified parthenolide. The hexanic, dichlorometanic and etheroacetic phases were pooled to constitute TPD. Extracts were stored at room temperature. Preparations were accurately divided into small pieces, dissolved in methylcellulose 0.5% (used as vehicle), and administered to the animals as intraperitoneal suspensions. Suspensions were freshly prepared every day.
Experimental groups in part I
The different groups of rats underwent one of the following treatments (see Table 1 for details):
Synopsis of experimental groups and treatments (see text for details of doses and route of administration)

Group 1: nitroglycerin at a dose of 10 mg/kg intraperitoneally (i.p.).
Group 2: TPD extract at the dose of 100 mg/kg i.p. for six consecutive days.
Group 3: TPE extract at a dose of 100 mg/kg i.p. for six consecutive days.
Group 4: equivalent volume of vehicle i.p. for six consecutive days.
Group 5: TPD extract at a dose of 100 mg/kg i.p. for six consecutive days.
Group 6: TPE extract at the dose of 100 mg/kg i.p. for 6 consecutive days;
Group 7: equivalent volume of vehicle i.p. for six consecutive days.
Rats in group 1 were perfused on day 1, 4 h after being injected with nitroglycerin.
On day 7, following the 6-day treatment period, the animals of groups 2, 3 and 4 were injected with nitroglycerin (10 mg/kg i.p.), while the rats of groups 5, 6 and 7 were injected with an equivalent volume of vehicle; 4 h after the acute administration of nitroglycerin or vehicle they were perfused.
The dose of nitroglycerin was chosen according to previous reports (18–22), while the dose of extract was defined on the basis of toxicological data and the results of pilot investigations conducted on TPE, which showed that 100 mg corresponded to 1/5 of the LD50 (50% mortality) (Indena S.p.A., data on file).
Experimental groups in part II
Groups of six to eight rats each underwent one of the following treatments for six consecutive days:
Group 8: vehicle i.p.
Group 9: purified parthenolide at the dose of 15 mg/kg i.p.
On day 7, following the 6-day treatment, animals of both groups were injected with nitroglycerin (10 mg/kg i.p), and perfused 4 h later.
The dose of purified parthenolide was chosen according to the data on the HPLC content of parthenolide into TPE extract (26%) and based on toxicological data and the results of pilot investigations (Indena S.p.A., data on file).
Sheehan et al. (31) have recently reported that parthenolide may exert a haemodynamic effect, therefore two additional groups of rats (groups 10 and 11) underwent monitoring of peripheral arterial pressure following the same treatment schedule as groups 8 and 9. Recordings of peripheral blood pressure were obtained by tail-cuff plethysmography (NIBP 23934; IITC Life Science, Woodland Hills, CA, USA): rats were placed in a restrainer and a cuff was placed on the tail. After a 15-min acclimatization, five measurements of blood pressure were taken at intervals of 1 min for baseline. Recordings were subsequently obtained at 20-min intervals for 2 h after the administration of nitroglycerin or vehicle.
Immunohistochemistry
Rats were anaesthetized and perfused through the ascending aorta with 300 ml of cold saline followed by 300 ml of ice-cold 4% paraformaldheyde, pH 8. Brains were removed and postfixed in the same fixative, then placed in increasing concentrations of sucrose (10, 20 and 30%) in KPBS for 72 h. Brains were then sectioned at 50 µm thickness on a freezing microtome into six wells containing antifreeze or KPBS solutions and processed for immunohistochemistry, as free-floating sections.
In order to avoid variability due to the staining procedure, each animal and its control(s) were processed at the same time. Sections were processed for immunohystochemical detection of Fos using a rabbit polyclonal antiserum diluted 1 : 10 000 (Oncogene Research Products, San Diego, CA, USA). Following several rinses in KPBS, sections were exposed to solution of normal goat serum (NGS 4%) for 30 min at room temperature. Sections were then incubated for 48 h at 4 °C in primary antibody in KPBS containing 4% NGS and 0.4% Triton X-100 (TX). After several rinses in TX 0.04%, sections were incubated with a secondary biotinylated antibody (Vector Laboratories, Burlingame, CA, USA) for 90 min and processed after rinses in TX 0.04% with avidin–biotin complex (Vectastain; Vector Laboratories) for 90 min. Before staining, sections were rinsed with acetate imidazole (AI) buffer 0.1
Tolerability
As an indirect measure of treatment tolerability, body weight was monitored daily for the entire experimental period (day 1 through day 7) (32–34).
Data analysis
Animals were analysed groupwise, each group with its control(s), separately for experimental groups in part I and II. Cell counts of individual nuclei were made from every sixth section throughout their rostrocaudal extent for each rat and its control. In order to avoid differences related to the asymmetrical sectioning of the brain, Fos-positive cells were counted bilaterally (three sections for each nucleus) (Scion Image Analysis; Scion Corporation, Frederick, MD, USA) and the mean value obtained from the two sides was used for statistical analysis. Student's t-test for unpaired data was used to compare differences in the mean number of Fos-immunoreactive nuclei per cell group between controls and treatment groups.
Mean blood pressure values were used for statistical analysis at baseline and at consecutive 20-min intervals. Effects of treatments were analysed using a two-way analysis of variance (
Results
Effect of chronic administration of vehicle upon nitroglycerin-induced Fos expression
In agreement with our previous findings, acute nitroglycerin administration induced significant Fos expression in the paraventricular and supraoptic nuclei of the hypothalamus, the central nucleus of the amygdala, the ventrolateral column of periaqueductal grey, locus coeruleus, parabrachial nucleus, nucleus trigeminalis caudalis and nucleus tractus solitarius (18–21).
Chronic i.p. administration of vehicle significantly reduced nitroglycerin-induced Fos expression in some nuclei, namely paraventricular and supraoptic nuclei of the hypothalamus, locus coeruleus, nucleus tractus solitarius and nucleus trigeminalis caudalis (Fig. 1). However, nitroglycerin administration following chronic vehicle administration still induced significant levels of Fos expression in selected brain areas, including the paraventricular and supraoptic nuclei of the hypothalamus, parabrachial nucleus, nucleus tractus solitarius and nucleus trigeminalis caudalis.
Effect of TPD and TPE on Fos expression
With the exception of the parabrachial nucleus, no significant Fos expression was observed in the brain nuclei of interest when comparing animals chronically treated with TPD or TPE with animals chronically treated with vehicle (Fig. 2).

Effect upon Fos expression induced by chronic treatment with extracts of Tanacetum parthenium depleted of (TPD) and enriched in (TPE) parthenolide. Histograms represent the number of Fos-positive neurons counted in each nucleus (expressed as mean ± SD). ▪, TPD for 6 days (group 5); □, TPE for 6 days (group 6);  , vehicle for 6 days (group 7). ∗P < 0.05 vs. vehicle. PVH, Paraventricular nucleus of the hypothalamus; SON, supraoptic nucleus of the hypothalamus; LC, locus coeruleus; PBN, parabrachial nucleus; NTS, nucleus tractus solitarius; NTC, nucleus trigeminalis caudalis.
, vehicle for 6 days (group 7). ∗P < 0.05 vs. vehicle. PVH, Paraventricular nucleus of the hypothalamus; SON, supraoptic nucleus of the hypothalamus; LC, locus coeruleus; PBN, parabrachial nucleus; NTS, nucleus tractus solitarius; NTC, nucleus trigeminalis caudalis.
Effect of TPD and TPE upon nitroglycerin-induced Fos expression
Chronic administration of TPE significantly reduced Fos expression induced by the i.p. administration of nitroglycerin in the nucleus trigeminalis caudalis (Fig. 3), while TPD proved devoid of any significant effect on the investigated model.

Effect upon nitroglycerin-induced Fos expression of chronic treatment with the extracts of Tanacetum parthenium depleted of (TPD) and enriched in (TPE) parthenolide. Histograms represent the number of Fos-positive neurons counted in each nucleus (expressed as mean ± SD). ▪, Vehicle (group 4); □, TPD (group 2);  , TPE (group 3). ∗P < 0.05 vs. group 4; †P < 0.05 vs. group 2. PVH, Paraventricular nucleus of the hypothalamus; SON, supraoptic nucleus of the hypothalamus; LC, locus coeruleus; PBN, parabrachial nucleus; NTS, nucleus tractus solitarius; NTC, nucleus trigeminalis caudalis.
, TPE (group 3). ∗P < 0.05 vs. group 4; †P < 0.05 vs. group 2. PVH, Paraventricular nucleus of the hypothalamus; SON, supraoptic nucleus of the hypothalamus; LC, locus coeruleus; PBN, parabrachial nucleus; NTS, nucleus tractus solitarius; NTC, nucleus trigeminalis caudalis.
Tolerability of TPE and TPD
Animals treated with TPD and vehicle showed a physiological increase in body weight (+42.0 ± 12.6 g and + 56.3 ± 8.1 g, respectively) (Table 2). Conversely, physiological increase in body weight was abolished in animals treated with TPE (+7.2 ± 15.2 g; P < 0.03 vs. TPD and vehicle groups).
Tolerability of the extracts of Tanacetum parthenium depleted of (TPD) and enriched in (TPE) parthenolide: body weight increase (in g) during the 6-day treatment period
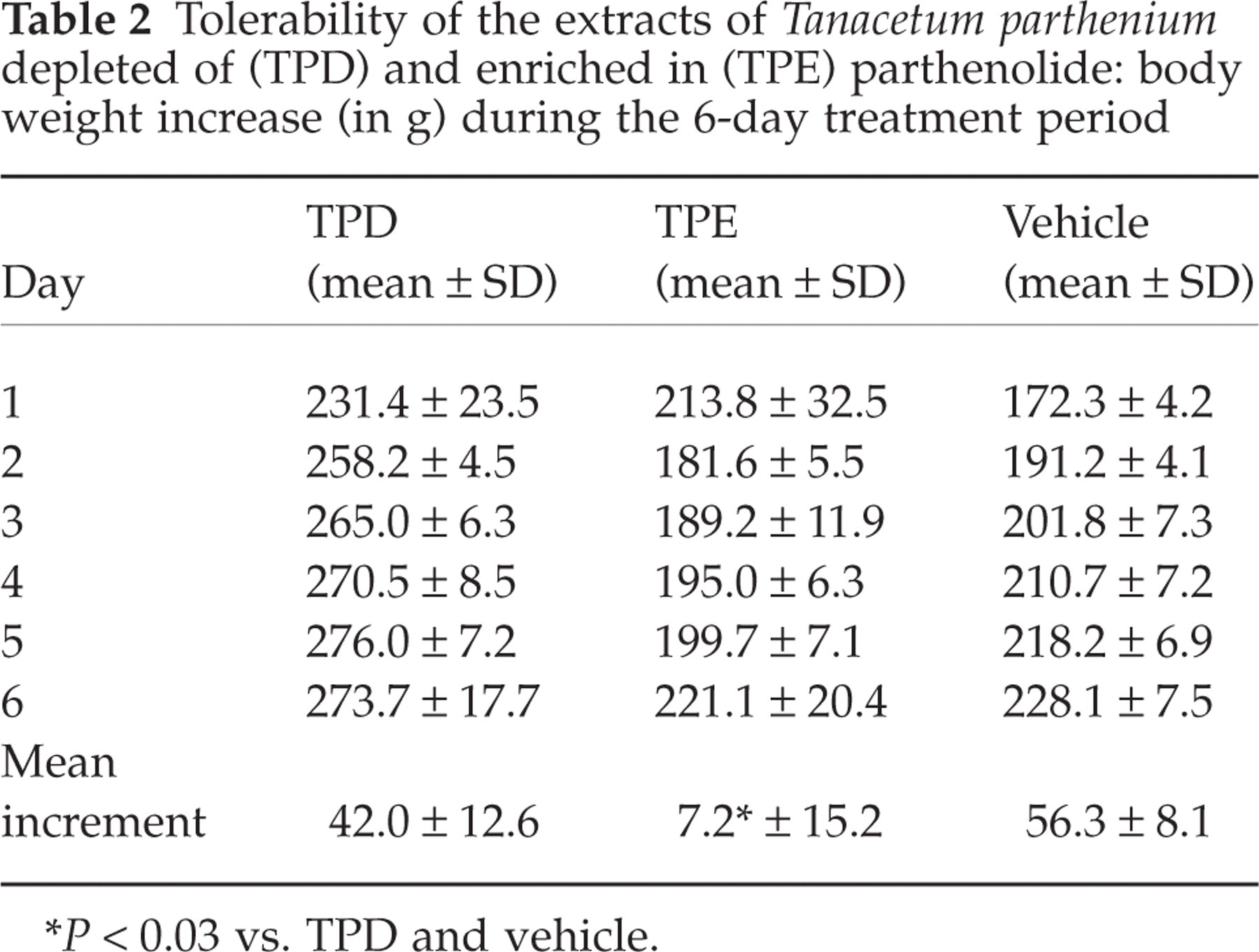
P < 0.03 vs. TPD and vehicle.
Effect of parthenolide purified from TP extracts upon nitroglycerin-induced Fos-expression
Chronic treatment with purified parthenolide significantly reduced nitroglycerin-induced neuronal activation in the following brain nuclei: paraventricular nucleus of the hypothalamus, parabrachial nucleus, nucleus tractus solitarius and nucleus trigeminalis caudalis (Figs 4 and 5).

Effect of purified parthenolide upon nitroglycerin-induced Fos expression. Histograms represent the number of Fos-positive neurons counted in each nucleus (expressed as mean ± SD). ▪, Vehicle (group 8); □, purified parthenolide (group 9). ∗P < 0.05 and ∗∗P < 0.03 group 9 vs. group 8. PVH, Paraventricular nucleus of the hypothalamus; SON, supraoptic nucleus of the hypothalamus; LC, locus coeruleus; PBN, parabrachial nucleus; NTS, nucleus tractus solitarius; NTC, nucleus trigeminalis caudalis.
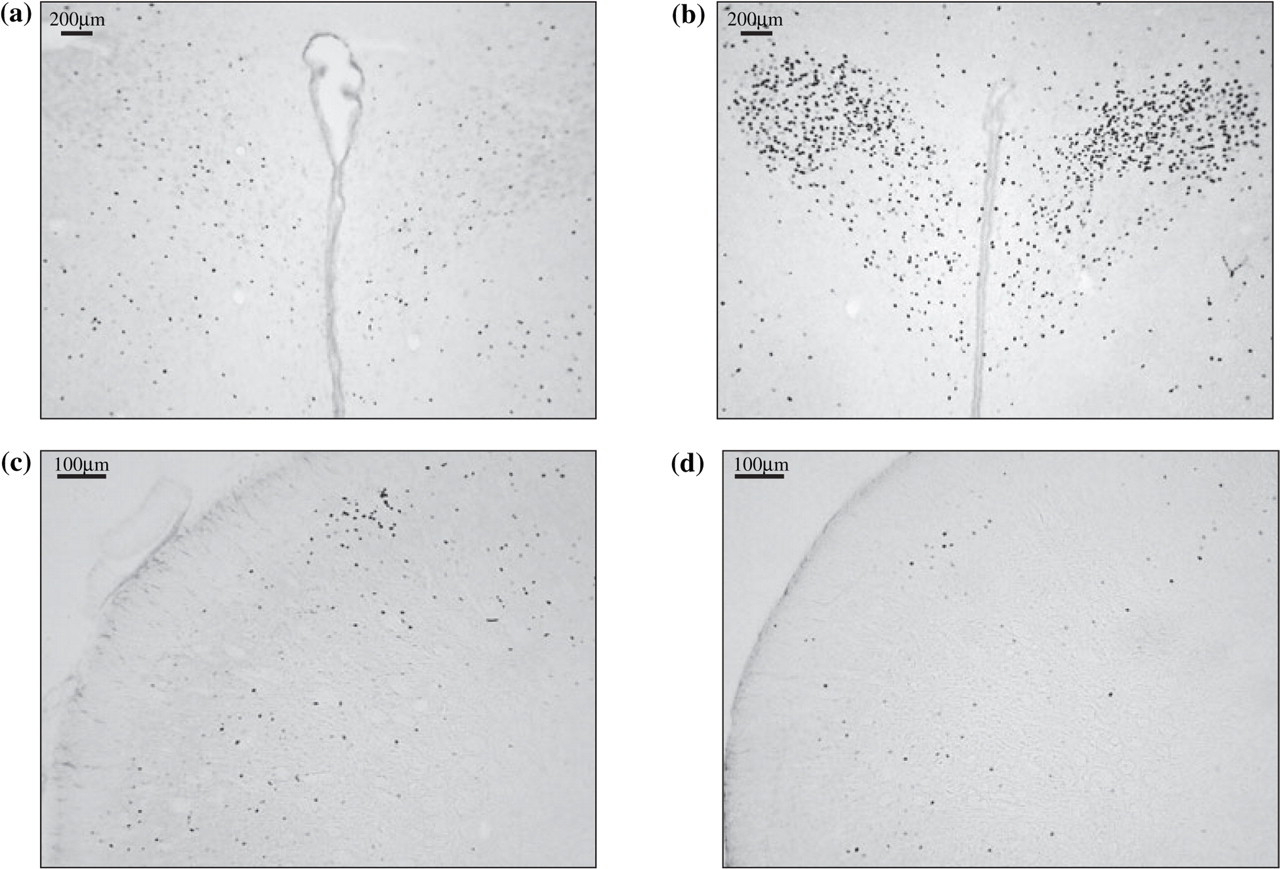
Micrographs of representative sections of paraventricular nucleus of the hypothalamus (top) and nucleus trigeminalis caudalis (bottom) of animals chronically treated with purified parthenolide (a,c) or vehicle (b,d) before receiving systemic nitroglycerin.
Chronic administration of purified parthenolide did not prevent nitroglycerin-induced reduction in blood pressure.
Tolerability of purified parthenolide
Animals treated with purified parthenolide showed a physiological increase in body weight (+51.3 ± 12.5 g), which was comparable to that observed in rats treated with vehicle (+45.6 ± 8.2 g; parthenolide vs. vehicle: ns) (Table 3).
Tolerability of purified parthenolide: body weight increase (in g) during the 6-day treatment period

Purified parthenolide vs. vehicle: ns.
Discussion
We have previously shown that exposure to nitroglycerin induces the activation of several brain nuclei that play a role in pain transmission (nucleus trigeminalis caudalis, periaqueductal grey) and autonomic control (locus coeruleus, parabrachial nucleus, nucleus tractus solitarius and paraventricular nucleus of the hypothalamus) (18–21). Nitroglycerin administration causes the prototypical induction of migraine headache in migraineurs, but not in controls (26–30). The cellular and molecular features of this response in rodent meninges resemble what has been found in other experimental migraine models: sterile inflammation and cytokine release (35–37). In addition, nitroglycerin has been shown to increase the discharge rate of neurons located in the nucleus trigeminalis caudalis (25). Compelling evidence points to an important role for the nucleus trigeminalis caudalis in the pathogenesis of migraine attacks (38–41). Human studies have also demonstrated a pivotal role for other nuclei, e.g. periaqueductal grey and locus coeruleus (41–43), all of which were activated by nitroglycerin administration.
In this study we report that chronic administration of a TP extract depleted of parthenolide failed to interfere with nitroglycerin-induced neuronal activation, while an extract enriched in parthenolide inhibited nitroglycerin-induced activation of neurons located in the nucleus trigeminalis caudalis. Furthermore, parthenolide purified from TP inhibited nitroglycerin-induced activation of neurons located not only in the nucleus trigeminalis caudalis but also in the parabrachial nucleus, nucleus tractus solitarius and paraventricular nucleus of the hypothalamus. It is noteworthy that the extract enriched in parthenolide showed a low tolerability, while purified parthenolide was very well tolerated by the animals.
The present data, showing the inhibitory activity of parthenolide upon a subgroup of brain nuclei in response to nitroglycerin administration, strongly suggest that parthenolide is the component responsible for the biological activity of TP as regards its antimigraine effect. Previous reports have indicated parthenolide as the most active chemical constituent in TP extracts (44). This is partly based on the structural characteristics of the molecule, which possesses an α-methylenebutyrolactone unit that interacts with sulphydryl groups (45). In addition, parthenolide appears to work through several different mechanisms that may be relevant for migraine attacks. Parthenolide inhibits granular release from human platelets and neutrophils of proinflammatory compounds such as arachidonic acid and serotonin (9, 18, 45, 46). This lactone possesses a low affinity for 5HT2A receptors and has been shown to inhibit serotonin-mediated smooth muscle contraction (47, 48) and the responses of rabbit aortic rings to phenylephrine, serotonin, tromboxane mimetic U46619 and angiotensin II (49–51).
The originality and soundness of the present findings reside in the direct comparison of the performance of two different extracts with a differential and precise content in parthenolide, freshly prepared every day, in an animal model of migraine and in the subsequent evaluation of parthenolide, purified from TP, in the same model.
The possible mechanisms underlying the inhibitory effect of the extract enriched in parthenolide and that of purified parthenolide upon nitroglycerin-induced Fos expression in brain nuclei are at present elusive. It has been shown that parthenolide dramatically reduces the number of acid-soluble sulphydryl groups in platelets (52). On the other hand, it has been suggested that sulphydryl groups are necessary for nitroglycerin to form nitric oxide (20, 53). Therefore, parthenolide may alter the formation of nitric oxide from nitroglycerin oxide via the neutralization of sulphydryl groups. Further studies on the tissue distribution of parthenolide, its possible bioactivities and molecular signalling pathways involved in the brain compartment are needed for a better definition of the neurochemical determinants underlying the present findings. Another possible mechanism of action for parthenolide in the central nervous system may be related to its inhibitory effect upon NF-κB activity. NF-κB is ubiquitously expressed in the brain and, under basal conditions, is sequestered within the cytoplasm by the IκB family of inhibitory proteins. Reuter et al. recently demonstrated that nitroglycerin increases the expression of iNOS within rat meninges by triggering transcriptional events, such as phosphorylation and degradation of IκBα (14). In Reuter's paradigm (dura mater), parthenolide administration attenuated IκBα degradation, NF-κB activation and iNOS expression, but preliminary data from our laboratory (Tassorelli et al., personal observations) show that NK-κB is also expressed in the nucleus trigeminalis caudalis following nitroglycerin administration.
Several clinical trials have been performed in the attempt to evaluate the effectiveness of TP in migraine prophylaxis (2, 3, 5, 54, 55). These studies, however, were performed with different types of extract, rarely with a standardized content of parthenolide, and with no control of the stability of the extract constituents during the several-week trials. This may be one of the main reasons for the inconsistency of results. Indeed, a meta-analysis published in 1998 concluded that although the majority of studies seem to support the effectiveness of TP in preventing migraines, larger studies with longer follow-up are needed to ascertain the efficacy of TP (4). More recently, Pittler and Ernst came to the conclusion that there is insufficient evidence from randomized, double-blind trials to suggest an effect of feverfew over placebo for preventing migraine (56).
In conclusion, in the present study, fixed (freshly prepared) amounts of the extract enriched in parthenolide as well as purified parthenolide proved able to interfere with the activation of a key area in migraine pathogenesis, nucleus trigeminalis caudalis. The extract enriched in parthenolide, however, was associated with a poor tolerability, while purified parthenolide was well tolerated. Although caution is recommended when extrapolating animal tolerability to humans, the present data provide convincing evidence that would justify devising future clinical trials based on the use of purified parthenolide.
Acknowledegment
This work was supported by a grant from Indena S.p.A. (Italy).
