Abstract
Background
Calcitonin gene-related peptide and its receptor, consisting of receptor activity-modifying protein 1 and calcitonin receptor-like receptor, are of considerable interest because of the role they play in migraine and recently developed migraine therapies.
Methods
To better understand the function of this neuropeptide, we used immunohistochemistry to determine a detailed distribution of calcitonin gene-related peptide, receptor activity-modifying protein 1 and calcitonin receptor-like receptor in the rat brain in a region of 0.5–1.5 mm lateral to the midline. We found calcitonin gene-related peptide immunoreactivity in most of the neurons of the cerebral cortex, hippocampus, cerebellum, thalamic nuclei, hypothalamic nuclei and brainstem nuclei. In contrast, receptor activity-modifying protein 1 and calcitonin receptor-like receptor immunoreactivity were found almost exclusively in the neuronal processes in the investigated regions.
Conclusion
Overall, the degree of expression of calcitonin gene-related peptide and calcitonin gene-related peptide receptor components in the central nervous system is astonishingly complex and suggestive of many different brain functions, including a possible role in migraine. However, currently, the presence of calcitonin gene-related peptide and the nature of its receptors throughout the brain is an enigma yet to be solved.
Introduction
Calcitonin gene-related peptide (CGRP) is the only neuronal messenger that has been shown consistently to be released in excess following an acute migraine attack. Upon successful treatment with a triptan, which acts presynaptically to inhibit CGRP release, or a postsynaptic CGRP receptor antagonist, migraine pain is reduced and the headache disappears (1). The origin of the released CGRP has been linked to the cranial trigeminovascular system, possibly from areas outside the blood–brain barrier (2). On the other hand, brain imaging during a migraine attack indicates the participation of numerous regions within the central nervous system (CNS) (3). The current focus on CGRP in acute migraine attacks has led to the search for the localisation of CGRP receptors in order to explain the possible site of action of the CGRP receptor antagonist, gepants (1). The CGRP receptor includes two components: a seven transmembrane-spanning G-protein coupled receptor, calcitonin receptor-like receptor (CLR) and a single transmembrane-spanning protein, the receptor activity-modifying protein 1 (RAMP1). Previous work has shown that CLR/RAMP1 can be found in intracranial vessels (4,5), in dura mater (4,6), in the trigeminal ganglion (7–9), in the brainstem (10), in the trigeminal nucleus caudalis (TNC) (11,12) and in the cerebellum (13,14). Although recent studies suggest the brainstem is a central component of migraine/other primary headache disorders, in-depth knowledge of CGRP and the CGRP receptor elements in the brainstem and in the CNS in general is lacking (15).
The present study was designed to comprehensively map the distribution of CGRP and its receptor elements CLR and RAMP1 throughout the rat brain in order to provide an overview of their localisation in fibres and cells. These findings are important additions to the discussion of migraine/headache pathogenesis.
Materials and methods
Ten Wistar male rats were euthanised by CO2 inhalation followed by decapitation. The brains were carefully dissected, cut sagittally in the midline and placed in 4% paraformaldehyde (PF) for 4 h, followed by incubation overnight in Sörensen’s phosphate buffer (pH 7.2) containing sucrose (10% and then 25%). Thereafter, the tissue was embedded in Yazulla embedding medium (30% egg albumin, 3% gelatin) and cryo-sectioned at 12 µm. The sections were stored at −20℃ until use.
The study followed the guidelines of the European Communities Council (86/609/ECC) and was approved by the Regional Ethical Committee on Animal Research, Malmö/Lund, Sweden (M43-07).
Haematoxylin and eosin (H&E)
Sagittal cryo-sections of the whole brain, including cerebellum, brainstem and C1 segment of spinal cord, were stained using H&E (H 4 min, E 1 min). The staining was done in order to examine the morphology and condition of the tissue, and to identify the distance of the section from the midline. The 6th edition of The Rat Brain in Stereotaxic Coordinates by Paxinos and Watson (16) and H&E staining of sagittal sections spanning 0.5–1.5 mm lateral to the midline were used to identify the different areas subjected to a detailed study of CGRP and RAMP1/CLR distribution (Figure 1).
Sagittal cryo-sections stained with H&E. The 6th edition of The Rat Brain in Stereotaxic Coordinates by Paxinos and Watson (16) was used to identify the different areas. Upper panel outline the regions of the brain investigated (0.5–1.5 mm lateral to the midline). Lower panel shows the regions analysed in the study. All nuclei and cell groupings begin with capital letters and nerve bundles with lower case letters. 10N: dorsal motor nucleus of vagus; 12N: hypoglossal nucleus; 4n: trochlear nerve; 4V: 4th ventricle; 7n: facial nerve; 7N: facial nucleus; ac: anterior commissure; CA1: field CA1 of the hippocampus; CA2: field CA2 of the hippocampus; CA3: field CA3 of the hippocampus; Cb: cerebellar lobule; cc: corpus callosum; CM: central medial thalamic nucleus; f: fornix, Gi: gigantocellular reticular nucleus; Gr: gracile nucleus; IO: inferior olive; LC: locus coeruleus; LSI: septal nucleus; Me5: mesensephalic trigeminal nucleus; Med: medial cerebellar nucleus; opt: optic tract; Pa: paraventricular hypothalamic nucleus; Pn: pontine nucleus; PVA: paraventricular thalamic nucleus; py: pyramidal tract; R: red nucleus; Re: reuniens thalamic nucleus; RMg: raphe magnus nucleus; Sol: Nu solitary tract; Sp5: spinal trigeminal tract).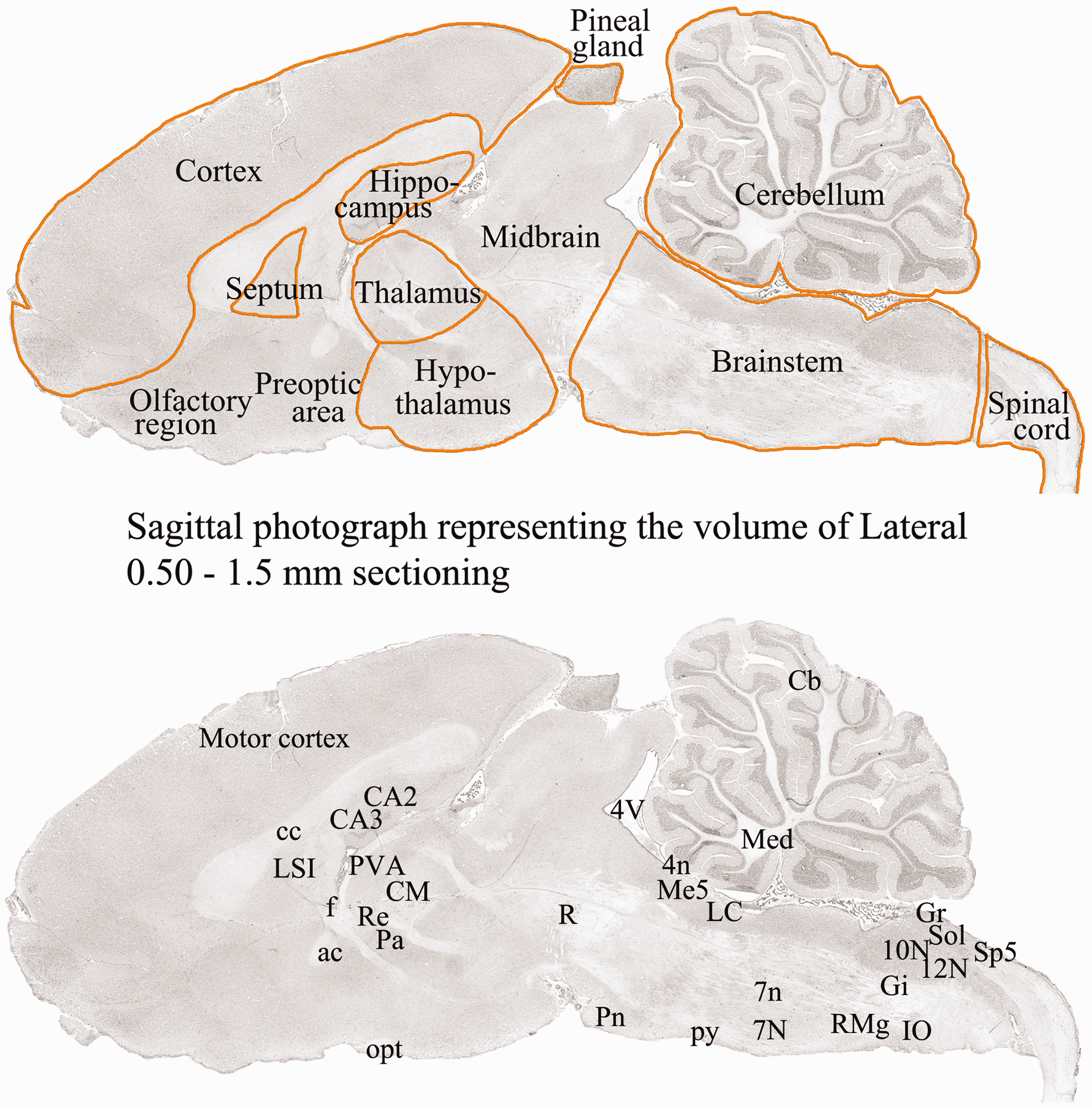
The abbreviations used in the photographs are taken from the English terms for these structures by Paxinos and Watson (16). The labels for all nuclei and cell groupings begin with capital letters, and nerve bundles are labelled using lower case letters (e.g. 7N = facial nucleus, 7n = facial nerve, LC = locus coeruleus, cc = corpus callosum).
Immunohistochemistry
Detailed description of primary and secondary antibodies.
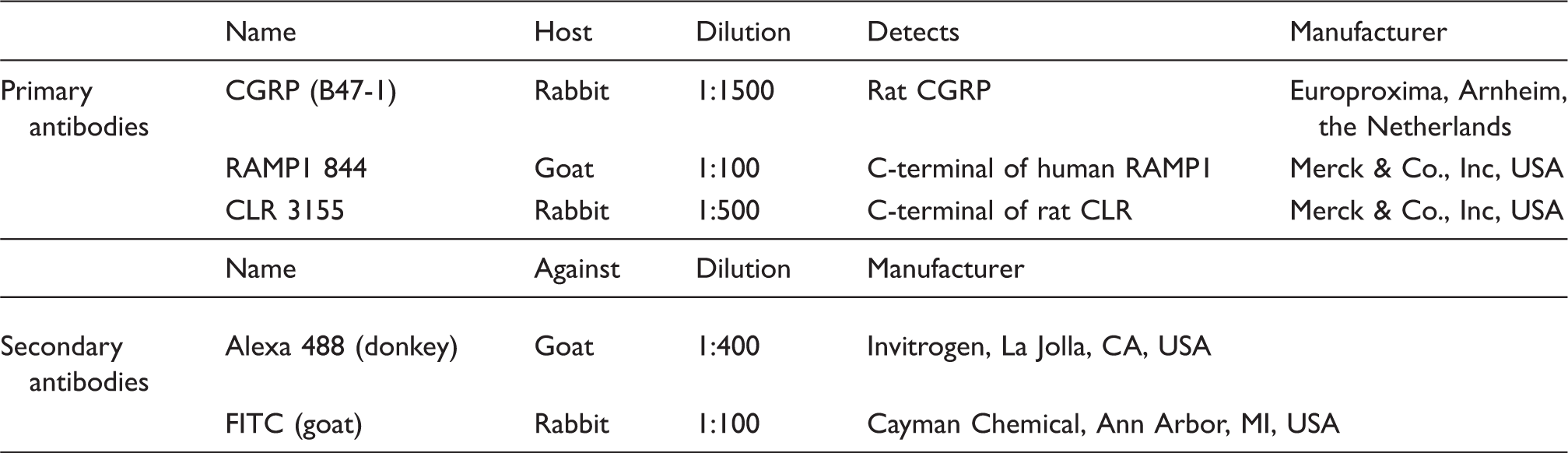
The development and specificity of the various CLR and RAMP1 antibodies have been demonstrated previously by our laboratory (7). The specificity of the antibodies was confirmed using HEK293 cells stably expressing the CGRP receptor and by western blot. Pre-absorption controls also have been performed with the CGRP, CLR and RAMP1 antibodies (7).
Omission of primary antibodies served as negative controls. The sections were examined in an epifluorescence microscope (Nikon 80i, Tokyo, Japan) equipped with a motor table, enabling us to get images of a whole section, and a Nikon DS-2MV camera. Finally, images were processed using Adobe Photoshop CS3 (v0.0 Adobe Systems, Mountain View, CA, USA).
Results
Cerebral cortex
The immunoreactive pattern was similar among the different regions of cortex, and therefore results for the cortex region as a whole are reported here. We primarily present findings that relate to the discussions on migraine pathophysiology.
CGRP
The density, size and morphology of CGRP immunoreactive cells, but also the shape and density of CGRP non-immunoreactive DAPI stained cells (most likely glial cells), indicate that all cortical neurons were positive for CGRP (Figure 2). The immunoreactivity was found mainly in the cell soma and visualised as positive grains in the cytoplasm (Figure 2, insert). The neuronal processes showed very limited or no immunoreactivity. Furthermore, the CGRP immunoreactivity was confined to cortical layers II–VI; consequently, no CGRP immunoreactivity was detected in cortical layer I, where there were few neuronal cell somas.
CGRP, RAMP1 and CLR distribution in cerebral cortex. The density, size and morphology of CGRP immunoreactive cells indicate that all cortical neurons were positive for CGRP (left panel). In addition, with the aid of DAPI staining of the nuclei, it was possible to distinguish neurons from glial cells or vessels according to the size and intensity of the DAPI staining. Immunoreactivity was mainly found in the cell soma and visualised as positive grains in the cytoplasm (insert). The neuronal processes showed very limited or no immunoreactivity. Furthermore, the CGRP immunoreactivity was confined to cortical layers II–VI. No neuronal CGRP immunoreactivity was found in cortical layer I (top). Slender RAMP1 immunoreactive fibres were found spanning through the entire cortex (middle panel) and also traversing through the cortical layers (insert). Positive horizontal fibres were most obvious in layer I (top) close to the brain surface and in layer III. CLR immunoreactivity was found in both neuronal cell somas and processes (right panel). In the cell soma, positivity was found in both nucleus and cytoplasm (insert). In addition, capillaries were often found positive for CLR.
RAMP1
Thin, slender RAMP1 immunoreactive fibres were found spanning across the entire cortex and also traversing through the cortical layers (Figure 2, insert). These horizontal fibres were most obvious in layer I, close to the brain surface, and in layer III. No neuronal cell somas were found to be positive for RAMP1.
CLR
Immunoreactivity was found in both neuronal cell somas and processes (Figure 2). In the cell soma, positivity was observed in both the nucleus and cytoplasm. In addition, capillaries were often found to be positive for CLR. Detailed analysis of the tissue revealed that the processes were often stubbier (Figure 2, insert). Overall the CLR staining gave an impression of high background-like staining compared to CGRP and RAMP1. This pattern contrasts with other structures of the brain (mainly nerves) where the CLR staining could be very well-defined.
Corpus callosum
Beneath the cortex lies the corpus callosum (cc), a white matter region containing fibres that connect the two hemispheres. No CGRP immunoreactivity was present in cc (Figure 3). On the other hand, RAMP1 and CLR immunoreactivity was abundant; fibres were intensely labelled with antibodies to both receptor proteins. At higher magnification, it was obvious that the RAMP1 fibres were thin, whereas most of the CLR positive fibres were thick with blurry surroundings (Figure 3). In addition, CLR staining was present in some of the capillaries.
CGRP, RAMP1 and CLR distribution in corpus callosum (cc). No CGRP immunoreactivity was present in the white matter of the corpus callosum (left panel). RAMP1 (middle panel) and CLR (right panel) immunoreactivity were intense with fibres labelled with both antibodies. In addition, CLR staining was present in some of the capillaries.
Hippocampus (CA1, CA2, CA3 and dentate gyrus)
The hippocampus is an enfolding of the cerebral cortex, having the shape in cross-section of a sea horse. It consists of grey matter covered with white matter and is divided into CA1, CA2, CA3 and the dentate gyrus, areas that are functionally and anatomically distinct. CA1, CA2 and the dentate gyrus showed similar staining patterns and are therefore grouped together. CA3 showed more intense staining.
CGRP
CGRP immunoreactivity in CA1, CA2 and the dentate gyrus was weak (Figure 4). The cytoplasm of the neurons contained few immunoreactive grains. In CA3, however, the cytoplasm of the pyramidal cell somas, displayed intense immunoreactivity (Figure 4, insert) in a similar way as was seen in the cerebral cortex.
CGRP, RAMP1 and CLR distribution in hippocampus. CGRP immunoreactivity in CA2 was weak (left, upper panel). The cytoplasm of the neurons contained few immunoreactive grains. In CA3 (left, lower panel), the cytoplasm of the pyramidal cells displayed intense immunoreactivity (insert). RAMP1 staining of CA2 displayed a few long slender processes and no cell soma staining (middle, upper panel). The extension of these processes indicated staining of the pyramidal cell processes. In the CA3 (middle, lower panel), the number of intense, immunoreactive fibres was abundant. The extension of the immunoreactive fibres was consistent with that of mossy fibres (originating from the dentate granule cells). CLR immunoreactivity (right, upper and lower panels) was confined to neuronal cytoplasm and nucleus, stubby fibres and capillaries with a similar pattern and in all areas investigated. Moreover, the extension of the positive fibres correlated with the extension of the pyramidal cells processes. CGRP, RAMP1 and CLR distribution in nerve structures. The images depict the following structures: fornix (f), anterior commissure (ac), optic tract (opt), trochlear nerve (4n), facial nerve (7n) and pyramidal tract (py). CGRP staining was not evident in the nerve structures. Both RAMP1 and CLR were expressed in the structures examined. Images of 4n and 7n illustrate the findings in nerve structures; there was no CGRP, but intense RAMP1 and CLR immunoreactive bundles of processes.

RAMP1
RAMP1 staining of CA1, CA2 and the dentate gyrus was found in a few long slender processes, but there was no staining of cell somas. The extension of these processes indicates these are pyramidal cell processes. In contrast, the CA3 region exhibited a higher number of immunoreactive fibres and also greater intensity of the RAMP1 staining (Figure 4). The extension of the immunoreactive fibres indicates that they are mossy fibres (originating from the dentate granule cells) and not the dendritic tree of the pyramidal cells.
CLR
In all four hippocampal regions, the immunoreactivity was confined to the neuronal cytoplasm and nucleus, stubby fibres and capillaries (Figure 4). Moreover, the extension of the immunoreactive fibres was consistent with the extension of pyramidal cell processes.
Nerve structures (fornix, anterior commissure, optic tract, trochlear nerve, facial nerve and pyramidal tract)
CGRP was not expressed in the nerve structures. In contrast, both RAMP1 and CLR were expressed in the structures examined. However, a difference existed between the general fibre structures and the nerves. In fibre structures, RAMP1 immunoreactivity was distinct whereas CLR staining gave a blurrier impression, as described for the corpus callosum above. When it came to the two nerves examined (trochlear nerve [4n] and facial nerve [7n]), the CLR staining was as well-defined as the RAMP1 staining; clear bundles of processes were seen with no diffuse background-like staining (Figure 5).
Cerebellum (cerebellar lobules and medial cerebellar nucleus)
CGRP
As we have shown earlier (13), CGRP immunoreactivity was mainly confined to the Purkinje cells in the cerebellar lobules (Figure 6). Similar to what was seen in cortex and hippocampus, the immunoreactivity was displayed as positive grains in the cell soma, often close to the nucleus. Fibres were not CGRP immunoreactive.
CGRP, RAMP1 and CLR distribution in cerebellum. As described earlier by our group (13), CGRP immunoreactivity was displayed as positive grains in the Purkinje cell soma (left, upper panel), often close to the nucleus. Fibres were not CGRP immunoreactive. RAMP1 staining (middle, upper panel) was found on the surface of the Purkinje cells and in fibres parallel to the cerebellar surface, indicating RAMP1 positivity in the granule cell axons spreading within the molecular layer. Furthermore, slender positive processes were found within the granular cell layer and the white matter. Immunostaining with CLR antibodies (right, upper panel) disclosed immunoreactivity in the molecular layer; with high background-like staining, and the Purkinje cell somas displayed positivity. Middle and lower panels show immunoreactivity in medial cerebellar nucleus (Med). Large neurons showed CGRP immunoreactivity (left panels). The complete neuronal cell soma contained CGRP positive grains. In Med, a tight mass of thin slender RAMP1 immunoreactive processes were observed (middle panels). Stubby CLR-positive fibres and neuronal cell soma also were found (right panels).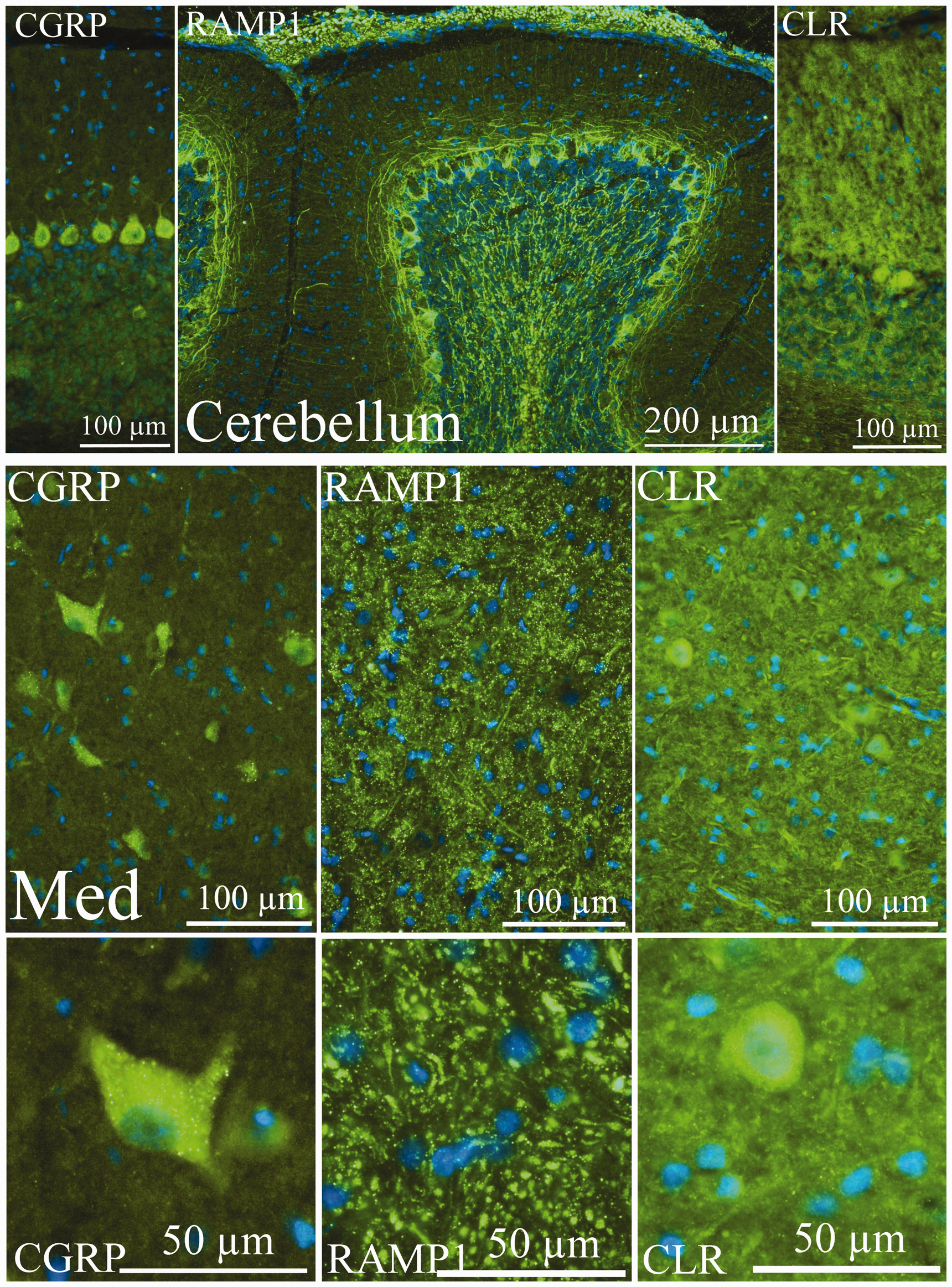
Large neurons in the medial cerebellar nucleus (Med) also showed CGRP immunoreactivity (Figure 6). The entire neuronal cell soma contained CGRP-positive grains. Occasional pearl-like CGRP-positive fibres were found.
RAMP1
RAMP1 staining was found on the surface of the Purkinje cells (similar to that described earlier (13) and in parallel fibres, indicating RAMP1 positivity in the granule cell axons spreading within the molecular layer (Figure 6). Furthermore, slender positive processes were found within the granular cell layer and the white matter.
In Med, a tight mass of thin slender RAMP1 immunoreactive processes was observed (Figure 6). No staining was found in the cell somas.
CLR
In a previous study by Edvinsson et al. (13), somewhat inconsistent results were obtained using the CLR rat antibody compared to what was observed with the CGRP and RAMP1 antibodies. Here we found that the molecular layer showed high background-like staining and that the Purkinje cell somas displayed positivity, in disagreement with our earlier findings (Figure 6). In the granular layer, however, stubby immunopositive fibres were found.
In Med, immunoreactivity was found in stubby fibres and neuronal cell somas (Figure 6).
Thalamus and hypothalamus (paraventricular thalamic nucleus, central medial thalamic nucleus, reuniens thalamic nucleus and paraventricular hypothalamic nucleus)
The thalamus is one of the most complicated structures in the brain and is involved in nearly all of its activities. In addition, much work has indicated that the thalamus and hypothalamus are possible sites of origin for the migraine attack (17). Here we demonstrate that the thalamic nuclei investigated showed similar CGRP immunoreactivity, which is intense cytoplasmic staining of the neurons (Figure 7). In the hypothalamic nuclei, the staining was less intense. The RAMP1 immunohistochemistry showed a similar pattern for all areas with a tight mass of positive slender processes. Positive CLR immunostaining was found in both blood vessels and neurons (Figure 7).
CGRP, RAMP1 and CLR distribution in thalamus and hypothalamus. The images depict paraventricular thalamic nucleus (PVA), central medial thalamic nucleus (CM), reuniens thalamic nucleus (Re) and paraventricular hypothalamic nucleus (Pa). The thalamic nuclei investigated showed similar CGRP immunoreactivity: intense cytoplasmic staining of the neurons (left column). In the hypothalamic nucleus, the staining was less intense (Pa, left column). The RAMP1 immunohistochemistry (middle column) showed a similar pattern for all nuclei with a tight mass of positive slender processes. CLR immunohistochemistry (right column) displayed positive blood vessels (asterisks) and neurons.
Mesencephalic trigeminal nucleus, red nucleus, locus coeruleus, raphe magnus nucleus, pontine nucleus, facial nucleus and inferior olive
In this group of nuclei, CGRP immunoreactivity was present in the neuronal cell somas, but not in the fibres (Figure 8). RAMP1 staining was found in slender fibres and, in addition, in the neuronal cell somas to a varying degree. CLR immunoreactivity was found in stubby fibres, cell somas and in blood vessels.
CGRP, RAMP1 and CLR distribution in brainstem. The images depict mesencephalic trigeminal nucleus (Me5), red nucleus (R), locus coeruleus (LC), raphe magnus nucleus (RMg), pontine nucleus (Pn) and facial nucleus (7N). In all the nuclei, CGRP immunoreactivity was present in the neuronal cell somas, but not in the fibres. RAMP1 staining was found in slender fibres and, in addition, in the neuronal cell somas to a varying degree. CLR immunoreactivity was found in stubby fibres, cell somas and in blood vessels (asterisks).
Septal nucleus
While CGRP immunoreactive fibres have been consistently observed in the TG, SPG and spinal cord, we found very few areas in the brain that contained CGRP immunoreactive fibres. However, the exception to this was the septal nucleus (Figure 9). This nucleus displayed long, slender, pearl-like CGRP-positive fibres together with cell soma staining.
CGRP distribution in septal nucleus. Very few areas in the brain contained CGRP immunoreactive fibres. The exception was the septal nucleus. The nucleus displayed long, slender, pearl-like CGRP-positive fibres together with cell soma staining.
Gigantocellular reticular nucleus (Gi), hypoglossal nucleus (12N), dorsal motor nucleus of the vagus (10N), solitary tract nucleus (Sol), gracile nucleus (Gr) and spinal trigeminal tract (Sp5)
Consecutive sections containing Gi, 12N, 10N, Sol and Gr stained with antibodies against CGRP or RAMP1 underscored the specificity of these antibodies (Figure 10). The CGRP-stained sections show intensely stained neurons in 12N and rather weakly stained, slender processes in the RAMP1 immuno-stained section. The Sol contained CGRP-positive neurons, but most of all, CGRP immuno-intense fibres. In the RAMP1 immuno-stained sections, there was hardly any immunoreactivity at all. In Gi, 10N and Gr, varying degrees of CGRP immunoreactivity were found in the neurons. In the RAMP1-stained sections, immunoreactive fibres were observed.
CGRP, RAMP1 and CLR distribution in gigantocellular reticular nucleus (Gi), hypoglossal nucleus (12N), dorsal motor nucleus of the vagus (10N), solitary tract nucleus (Sol) and gracile nucleus (Gr). Consecutive sections containing Gi, 12N, 10N, Sol and Gr stained with antibodies against CGRP or RAMP1 revealed the specificity of these antibodies. The CGRP stained section (upper image) showed intensely stained neurons in 12N and rather weakly stained, slender processes in the RAMP1 immuno-stained section (lower image). The CGRP-stained Sol region showed positive neurons, but most of all CGRP immuno-intense fibres. In the RAMP1 immuno-stained section, there was hardly any immunoreactivity. In Gi, 10N and Gr, varying degrees of CGRP immunoreactive neurons were found. In the RAMP1-stained section, immunoreactive fibres were observed.
Discussion
This is the first comprehensive study of the distribution of CGRP and RAMP1/CLR in the rat brain. The more detailed understanding of the location of CGRP and its receptor components can provide direction for future studies of the putative roles of this neuropeptide in relation to the CNS. Mapping of CGRP and RAMP1/CLR in the 1-mm-thick brain slice gave the impression that almost all neurons are CGRP-positive and almost all fibres are RAMP1/CLR-positive: (i) in cerebral cortex, cerebellum and hippocampus; (ii) in nerve fibre structures and nerves; and (iii) in the nuclei of thalamus, hypothalamus and brainstem, which implies that the function(s) of this transmitter/mediator molecule and its receptor components may involve the entire brain.
In the many areas, the overall impression of the intense CLR staining was that it was different from CGRP and RAMP1 in that it was background-like and exhibited unique fibre morphology. The CLR-positive fibres had a thicker, stubbier profile which could indicate that the slender fibres positive for RAMP1 were not completely and fully stained. We have recently shown in rat retina that CLR expression dominates vastly over RAMP1 using flow cytometry (18). This might explain why CLR staining appears to be a result of high background rather than a specific staining of functional RAMP1/CLR receptors.
Cortical structures
The density, size and morphology of CGRP immunoreactive cells indicate that all neuronal cell somas were positive for CGRP, as was earlier described (19). The neuronal processes showed very limited or no CGRP immunoreactivity. In contrast, staining for the receptor components showed a different pattern. Thus, slender RAMP1 immunoreactive fibres were found spanning through the entire cortex, but also tangentially through the cortex lending to the conclusion that RAMP1-positive fibres originate from pyramidal and/or stellate cells. It is interesting to note that the pyramidal cells are the only cells in the cerebral cortex with efferent projections targeting other areas in the brain where we found CGRP and RAMP1/CLR expression (Figure 11a).
H&E and neuronal circuits within cerebral cortex (a), hippocampus (b) and cerebellum (c).
Differences in the local concentrations of neurotransmitter receptor binding sites have been reported for several receptor types (reviewed by Eickhoff et al. (20)). It is suggested that mean concentration and laminar distribution pattern need to be considered in the assessment of neurochemical differences between cortical areas (20). With the method used in the present study, we were unable to quantitatively determine concentrations of CGRP and RAMP1/CLR, or to examine laminar resolution. However, our more qualitative results, which show the presence of CGRP and RAMP1/CLR within the entire cortex, ought to be of interest in understanding the structural basis of normal and pathological brain function. One aspect of interest is whether the cortical CGRP system has a role in the phenomena of spreading depression (21).
Hippocampus
In the present study, we found a high level of staining in projections and cell soma in region CA3. CGRP was found mainly in the pyramidal cells of CA3, whereas RAMP1 appears in the mossy fibres. This distribution could indicate that RAMP1 immunoreactive mossy fibres from the dentate gyrus synapse with the CGRP-positive pyramidal cells in CA3, since granule cells within the granule cell layer generate mossy fibres that extend for the full transverse extent of CA3 (Figure 11b) (22).
Stress is a ‘necessary evil’ for survival; however, severe and long-term stress disrupt brain structure and function. It is well-known that stress is a trigger of migraine headaches (reviewed by Akerman et al. (23). Recently, Liu et al. investigated the structural changes of hippocampus and amygdala and their relationships with migraine frequency and prognosis in migraine patients (24). The hippocampal mean adjusted volume varied as a function of headache frequency and peaked (increased volume) in patients with attacks 5–7 days/month and was negative (decreased volume) in patients with 15–30 attacks/month. In addition, the hippocampus volume was positively associated with a good two-year outcome of migraine. Since the CA3 area is a region especially vulnerable to stress and seizure-induced damage (25), it is reasonable to suggest a relationship of stress-related hippocampal volume change to headache frequency (24).
The neuro-immunohistochemical features presented here are unique from a transmitter/receptor distribution point of view, and possibly also from a functional point of view. The hippocampal system is said to be dedicated to the organisation of experiences in memory (26) and that CA3 is essential for encoding and storing episodic memories (27). In addition, spatial information is said to be represented in the hippocampus, and CA1 and CA3 provide essential information for navigation (reviewed by van Strien et al. (27)). It has been suggested that the increase of hippocampus volume in patients with 3–7 headache days/month implies hippocampus adaptation to the increased physical and emotional demands in this frequency range, and can be viewed as a learning process of adaptation and adjustment to ‘migraine experiences’ (28). On the other hand, a decrease in hippocampus volume with increasing headache frequency suggests that frequent attacks of migraine (beyond a threshold) exceed the coping ability of the neural system and lead to a maladaptive volume reduction (29). Nevertheless, the hippocampus displays structural plasticity linked to both headache frequency and clinical outcome of migraine, and this could suggest a role of CGRP and RAMP1/CLR in hippocampal neural plasticity in migraineurs.
Cerebellum
The distribution of CGRP and its receptor components in the cerebellar lobules has previous been published by our group (13). We concluded in 2011, as confirmed by the present results, that there exists a rich expression of CGRP in the Purkinje cells with receptor elements present in the fibres clearly pointing at a functional role of CGRP in the cerebellum. Clinical PET studies have shown that cerebellar regions are activated during migraine attacks (30,31). More recently, activation of cerebellum was demonstrated during the premonitory phases of migraine (32).
In the present study, we demonstrated CGRP and RAMP1/CLR expression in the medial cerebellar nucleus and, together with the other CGRP and RAMP1/CLR positive circuitries presented here, one might further speculate upon the role of CGRP in these areas (Figure 11c). However, searching for clues to the function of CGRP and RAMP1/CLR in the cerebellum has to include, on one hand, a definition of the fundamental operations that the cerebellum performs and, on the other hand, a detailed investigation of the cellular properties (33).
Thalamic and hypothalamic nuclei
The body’s cycles, senses, thoughts and emotions are just a small sample of functions that involve the thalamus (www.upright-health.com). We depicted CGRP in the neuronal somas and RAMP1/CLR expression in the neuronal processes in a number of thalamic and hypothalamic nuclei. Noseda et al., using a combination of in vivo single-unit recording, juxtacellular labelling with tetramethylrhodamine dextran and in vitro immunohistochemistry, found no evidence for the presence of CGRP-positive axons in any thalamic nucleus containing trigeminovascular neurons (34). The absence of CGRP-positive fibres raised the possibility that CGRP release is not localised within the thalamus but rather may be released at a distant location of the receptors (34). This reasoning might explain our findings of CGRP immunoreactivity in cell somas only and not the neuronal processes.
It is said that the thalamic nuclei constitute a nodal area within a circuit that includes, for example, brainstem nuclei, hypothalamus and cortex (35). The nuclei receive very strong inputs from several systems that are defined by their specific neurotransmitters (35). The neurochemical pathways that connect to thalamic trigeminovascular neurons and the likelihood that many of them modulate neuronal activity define a sophisticated neuroanatomical network that may help us hypothesise how sensory, physiological, cognitive and affective conditions trigger, worsen or improve migraine headache (34).
Septal nucleus
The nucleus is a structure inferior to the corpus callosum and it has a wide array of reciprocal projections with many brain structures (36) and it plays a role in reward and reinforcement (37). It is suggested in pain research that the septal nucleus may primarily mediate affective-motivational components rather than sensory-discriminative components of pain (38).
In the present study, we showed that the septal nucleus displayed long, slender, pearl-like CGRP-positive fibres together with cell soma staining. In contrast to results in trigeminal ganglion, sphenopalatine ganglion (see Edvinsson and Warfvinge, this issue) and the spinal cord, CGRP immunoreactive processes in the CNS are a rare finding. Whether the current findings reflect CGRP involvement in the reinforcement/reward system or the ability to withstand pain could only be speculated upon. However, the robust septal nucleus projection to the hippocampus, a brain structure said to be involved in migraine (24), might indicate that the nucleus plays a role in ‘reward and reinforcement’ for patients suffering from migraine.
Brainstem nuclei
The brainstem plays a role in conduction of all information transmitted from the body to the cerebrum and cerebellum and vice versa. In addition, the brainstem has integrative functions in cardiovascular control, respiratory control, pain sensitivity control, alertness, awareness and consciousness. Thus, brainstem damage is a very serious and often life-threatening problem. With respect to migraine pathology, dysfunction of brainstem structures and networks could not only account for the somatosensory component of migraine (headache), but also for the auditory, olfactory and visual aspects. Moreover, dysfunction of the locus coeruleus could explain the distractibility and anxiety (39), which is often observed in migraineurs (reviewed by Sprenger and Goadsby (40)).
In the present study, we examined the mesencephalic trigeminal nucleus, red nucleus, locus coerules, raphe magnus nucleus, pontine nucleus, facial nucleus and inferior olive. We found CGRP immunoreactivity in all the nuclei. Bearing in mind the functional importance of the brainstem and its conduction of information within the different regions of the brain, our results indicate that CGRP is present not only within the brainstem but the entire brain.
Medulla oblongata
The medulla oblongata is the lowermost portion of the vertebrate brain, continuous with the spinal cord. Motor nuclei of the medulla oblongata include, for example, the Gi, the 12N, the 10N, and the sensory nuclei include, for example, Sol, Gr and Sp5. This region controls the reflex actions of the pharynx, larynx and tongue, which are related to deglutition, mastication and speech as well as the visceral reflexes of coughing, sneezing, sucking, vomiting, salivating and other secretory functions.
In the present study, we found CGRP and RAMP1/CLR immunoreactivity in the nuclei investigated, except from the Sol region where CGRP staining showed positive neurons and fibres, but no RAMP1/CLR immunoreactivity. It has been proposed that the solitary tract nucleus has a role in memory processing, in particular in activities with a high motivational component (41).
Conclusion
The present study shows that CGRP is present in almost all neuronal cell somas and receptor components almost exclusively in the neuronal processes in the brain. The degree of expression of CGRP and CGRP receptor components in the CNS is astonishingly complex and suggestive of many different brain functions, including a possible role in migraine. However, currently the presence of CGRP and the nature of its receptors throughout the brain is an enigma yet to be solved.
Key findings
CGRP and its receptor are important hot topics in migraine. The present study shows that CGRP is present in almost all neuronal cell somas in the brain. The receptor was almost exclusively found in the neuronal processes in the investigated regions. The expression of CGRP and its receptor components is astonishingly complex and suggestive of many different brain functions.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this research is supported by grants from the Swedish Research Council (no. 5958) and the Swedish Heart and Lung Foundation.
