Abstract
The effect of migraine on the prognosis of epilepsy has not been reported. The aim of this prospective 5-10-year follow-up study was to examine some outcome measures and the cumulative probability of being seizure-free in epilepsy patients with migraine, and to compare their results with those of epilepsy patients without migraine. Fifty-nine patients (40 women; mean age 25 years) were diagnosed with both epilepsy and migraine (EM group). The control group consisted of 56 patients with epilepsy but without migraine (E group). Both groups were recruited and followed up over similar periods. We compared the outcome variables in the EM group with those in the E group. Kaplan-Meier methods were used to assess the seizure-free curves. The EM group had a significantly lower cumulative probability of being seizure-free over 10 years compared with the E group. The other epilepsy outcome measures at follow-up differed significantly between the groups, with the EM group having a longer duration of epilepsy, a lower early treatment response, and a higher incidence of intractable epilepsy and achieving remission with polytherapy, and more seizure control and medication problems for at least the last 2 years of follow-up. Comorbid migraine had a negative effect on the prognosis of epilepsy.
Introduction
Migraine and epilepsy are two distinct neurological conditions and, despite many reviews of the relationship between them, the nature of this association is still unresolved (1–5). The reported frequency of migraine in epileptic populations ranges from 8.4% to 23%(6–10).
There is a lack of information on the prognosis of epilepsy in a person with comorbid migraine. Specifically, it is not known whether the chances of achieving remission of seizures, intractability of seizures, and other outcome measures differ between epilepsy sufferers with and without comorbid migraine. Controlled prospective studies are necessary. Thus, the aim of this prospective 5–10-year follow-up study was to investigate the prognosis of epilepsy in epileptic patients with migraine and to determine how migraine affected it.
Methods
Part 1
Part 1 started in January 1993 and lasted 5 years. The study sample was assembled from a previously reported cohort of patients with epilepsy. Details of the methods used in that study are described elsewhere (11). In summary, 412 consecutive patients with epilepsy (200 women; ages 15–70 years) from our epilepsy out-patient clinic, the Neurology Department at Karadeniz Technical University Medical Centre, were prospectively interviewed to determine whether they had suffered from migraine during the 5-year period between 2 January 1993 and 15 December 1998. The purpose of that study was to determine not only the prevalence of migraine in our epileptic population but also the temporal association between migraine attacks and seizures and the number of patients with ‘migraine-induced epilepsy’. The seizures and epileptic syndromes were classified according to the systems proposed by the International League Against Epilepsy (ILAE) (12, 13). Epilepsy was the primary reason for admission. Both new-onset and pre-existing epilepsy subjects were included. The diagnosis of migraine was made according to the criteria of the International Headache Society (IHS) (14). All patients with suspected secondary headaches were excluded. The diagnosis of migraine was made only in the patients with epilepsy if migraine attacks also occurred independently of seizures. The migraine type was classified as with or without aura.
Fifty-nine patients (40 women, 19 men; mean age 25 years) had a diagnosis of both epilepsy and migraine (EM group) in this cohort. Twenty-three (39%) patients (six men, 17 women) fulfilled the diagnostic criteria for migraine with aura and 36 (61%) (13 men, 23 women) for migraine without aura. Seven (12%) of the 59 patients had migraine-induced epileptic seizures and all of them also had migraine attacks independent of seizures. Further details are available elsewhere (11).
Control subjects
The control group consisted of 56 epileptic patients without migraine (E group). The subjects were recruited and followed up over similar periods (1993–2003 for EM patients and 1991–2003 for controls). For each patient, one control matched for age, sex, and similar epilepsy syndrome was enrolled. These particular variables were very important in the construction of a prognostic profile. The controls were also recruited during part 1 of the study. Nevertheless, because five appropriate controls [one with cryptogenic temporal lobe epilepsy (cTLE), one with symptomatic temporal lobe epilepsy (sTLE), one with mesial temporal lobe epilepsy with hipocampal sclerosis (hsTLE), one with idiopathic epilepsy with generalized tonic-clonic seizures only (IGE), and one with symptomatic generalized epilepsy (SGE)] could not be contacted during this period, five control patients with epilepsy being followed up in our clinic were retrospectively selected. No controls matching the three patients with occipital lobe epilepsy (OLE) could be found.
To have equal sample sizes, the clinical data of these 59 patients with epilepsy and migraine were compared with those of the 51 patients with epilepsy recruited in part 1 of our study and five additional retrospectively (1991–1993) recruited patients with epilepsy treated in our epilepsy out-patient clinic (33 women and 23 men; mean age 25 years).
Part 2
Enrolment continued for 5 years (1993–1998). The follow-up ended in November 2003. Outcome was assessed 5–10 years after enrolment. EM patients were followed for 5–10 years (mean 7.76 ± 1.68 years) and the E patients for 5–12 years (mean 7.98 ± 1.88 years). No patient was lost to follow-up. During the follow-up, we compared the seizure outcome variables in the EM group with those in the E group.
Both groups had been assembled by the same investigators, using the same diagnostic criteria. A minimum 1 year's follow-up was required for inclusion. The choice of therapy was left to the physician's judgement. Poor seizure control and/or antiepileptic drug (AED) toxicity were prerequisites for dose adjustments or subsequent AED changes. When treatment failed, an alternative monotherapy was suggested before the addition of a second AED. Besides considering the seizure type or epilepsy syndrome, while choosing AEDs we also took concomitant migraine disorder into account in the EM group. We preferred to institute a treatment with antimigraine AEDs because of their dual effects as an anticonvulsant and a migraine-preventive agent. None of the patients in the EM group received antimigraine drugs lowering the seizure threshold, such as tricylic antidepressants. Follow-up visits were encouraged at 3- and 6-month intervals and even at unscheduled times when specifically indicated. We used a seizure-free period of at least 2 years as the cut-off point for the discontinuation of treatment.
At entry, patients and controls were interviewed to collect the main demographic and clinical data that affect epilepsy prognosis to the greatest extent, such as age, sex, age at onset of seizures, disease duration at first admission, seizure types and frequency, epileptic syndrome, the number of seizures before the treatment (≤10 and > 10), time to onset of treatment from the first seizures, and follow-up duration (15–25). Risk factors included a family history of epilepsy and migraine, febrile seizures, head trauma, and others (e.g. prematurity). Duration of epilepsy at first admission was defined as the time from the onset of seizures to the patient's participation in the study. Seizure frequency was defined as the number of seizures during the year before the first admission (at the baseline) and was classified as 1 for one seizure, 2 for 2–10 seizures and 3 for > 10 seizures.
Table 1 shows the main demographic and clinical data of the patients at entry and follow-up. The groups did not differ in terms of their clinical characteristics, except for family history of migraine at the baseline. The family history of migraine in the EM group was significantly higher than that in the E group.
Demographic and clinical characteristics at entry, and follow-up time

IGE, Idiopathic epilepsy with generalized tonic-clonic seizures only; TLE, temporal lobe epilepsy; hsTLE, mesial temporal lobe epilepsy with hipocampal sclerosis; cTLE, cryptogenic TLE; sTLE, symptomatic TLE; OLE, occipital lobe epilepsy; LOLE, late onset childhood occipital epilepsy (gastaut type); cOLE, cryptogenic OLE; sOLE, symptomatic OLE; BECTS, benign childhood epilepsy with centrotemporal spikes; JME, juvenile myoclonic epilepsy; JAE, juvenile absence epilepsy; SGE, symptomatic generalized epilepsy; U, epilepsy undetermined whether focal or generalized.
χ2.
The number of seizures during the year before the first admission.
Table 2 summarizes the migraine features characterizing the severity of migraine attacks at the baseline. At follow-up, the severity of migraine of patients was categorized as good (an improvement) or poor migraine outcome (no improvement). Good migraine outcome was defined as the frequency and severity of migraine attacks reduced by at least 50% compared with the baseline. Poor migraine outcome was defined as a reduction of < 50%, no change or a worsening.
Baseline characteristics of migraine (n = 59)

The long-term outcome measures at follow-up
The following six clinical variables were recorded:
The duration of epilepsy: the number of days between the first and last seizure (26).
Early treatment response: a ≥ 75% seizure reduction or lack of seizures from the second month of effective AED therapy over 2 years (25).
The number of patients with refractor seizure disorder. Intractable epilepsy defined as (i) more than one seizure/month over 18 months and (ii) failure of more than two first-line AEDs (27). A drug was not considered to have failed unless it had been increased to maximum tolerable levels without adequate seizure control. Drugs stopped only because of side-effects were not considered therapeutic failures. In addition, lack of seizure control due to non-compliance was not considered to constitute drug failure.
Current seizure outcome. We analysed seizure status at the last follow-up visit (in November 2003), and patients were classified as those with seizure control (seizure-free with or without AEDs) or without seizure control (not seizure-free and taking medication) for at least the last 2 years of follow-up. We also recorded whether they were taking polytherapy or monotherapy.
The number of the patients achieving remission. Remission was defined as a seizure-free period and was calculated for 1, 2, 3, 4, 5, 6, 7, 8, 9 and 10 years. In addition, the occurrence of remission with monotherapy or polytherapy and of more than one period of remission was recorded. When there was more than one remission period, the longest period was used as the basis for the remission period. Subjects with more than one period, and those who entered remission with polytherapy for at least one period were defined as achieving remission with polytherapy.
Cumulative probability of being seizure-free. This is explained below.
Statistical analysis
The end point for this analysis was a probability of being seizure-free with increasing follow-up time (5–10 years). Kaplan–Meier estimates were used to predict the remission and the events. Relapse was considered an event for Kaplan–Meier analysis. Log rank analysis was used to compare the seizure-free curves.
In the additional analyses, because epilepsy outcome in the EM group could be affected by the migraine severity, we investigated the cumulative seizure-free curves of the migraine outcome groups. Therefore, we compared the cumulative epilepsy remission curves of the poor migraine outcome group with those of the good migraine outcome group.
Continuous variables were analysed for significance by Student's t-test. Discrete variables were analysed by χ2 or Fisher's exact test. A P-value < 0.05 was considered statistically significant.
Results
The baseline characteristics of the EM patients were similar to those of the E patients with regard to age, sex, age at onset of seizures, disease duration at first admission, seizure types and frequency, epileptic syndrome, the number of seizures before treatment (≤10 and > 10), time to the onset of treatment from the first seizures, and follow-up time at the end of the study (Table 1). These baseline characteristics were the clinical data that affected the epilepsy prognosis the most. However, the long-term outcome measures of the patients differed during the follow-up period (Table 3). At follow-up, there were significant differences between the groups in terms of duration of epilepsy, early treatment response, incidence of intractable epilepsy and entering remission with polytherapy or monotherapy, and remission duration (Table 3). There were no differences between the groups in terms of number of subjects achieving remission at any point during the follow-up, and of those with more than one period of remission (Table 3). The EM patients manifested a longer duration of epilepsy (P = 0.02) and had intractable epilepsy more frequently (P = 0.01). In the E group, 30 (54%) patients had an early treatment response; this figure was significantly lower (22%) in the EM group (Table 3). The percentage of patients entering remission with polytherapy was significantly higher in the EM group than in the E group.
Epilepsy outcome measures at follow-up
For the first 2 years.
Seizure-free for at least 1 year.
Because some patients achieved remission at some point and then relapsed, the number of subjects with seizure control at the last follow-up visit is smaller than the number of subjects achieving remission.
Significant.
NS, Non-significant.
Table 3 also shows the proportion of patients having had seizure control and no control at the latest follow-up check according to group (seizure outcome at the last follow-up visit). Of the patients with ‘epilepsy only’, 46% were both free of seizures and had not received medication for at least the last 2 years of the follow-up, compared with only 12% of the EM patients (P = 0.000). Furthermore, 29% of the E patients were still having seizures and receiving medication, compared with 56% of the EM patients (P = 0.003). There was no difference regarding ‘taking AEDs and being free of seizures’ between the groups (Table 3).
Analysis for the cumulative probability of being seizure-free
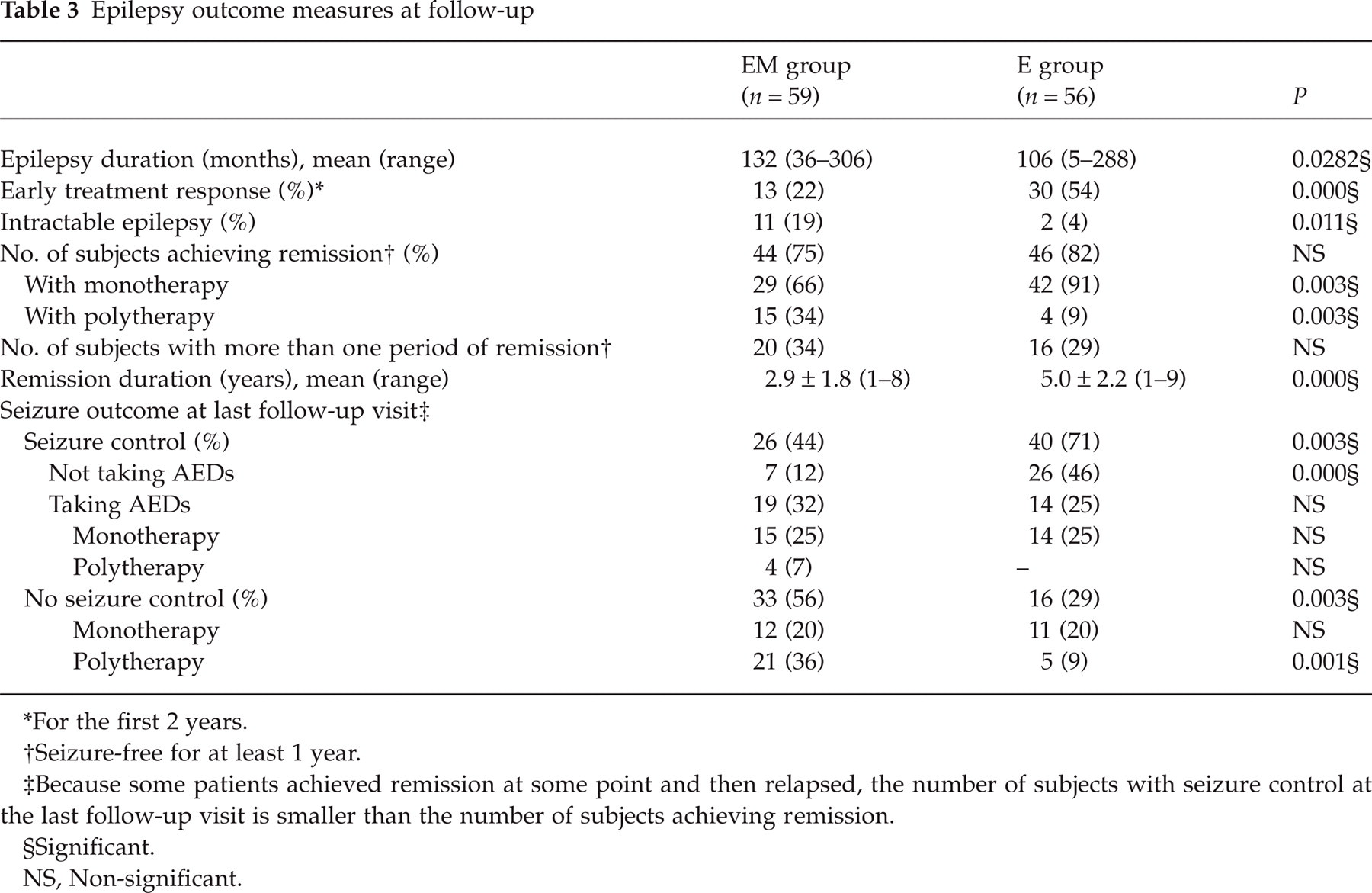
Figure 1 shows ‘seizure-free’ curves for the length of follow-up in the EM and E groups. There was a significant difference in the cumulative probability of being seizure-free between the two groups. At the 10-year follow-up, the proportion of subjects remaining seizure-free for the whole follow-up period was zero. That is, none of the subjects in the study had experienced a seizure-free period of 10 or more years. Some patients achieved remission at some point and then relapsed. The association between migraine and higher relapse risk approached significance. The epilepsy–migraine patients had significantly lower cumulative seizure-free curves over 10 years compared with the epilepsy patients (log rank test = 18.33; P < 0.001) (Fig. 1).

Cumulative seizure-free curves during the 10 years of follow-up estimated by Kaplan–Meier analysis for epilepsy patients with and without migraine. (log rank test = 18.33; P < 0.001). –––, Epilepsy–migraine group; - - - -, epilepsy group.
In addition, we estimated the migraine severity during follow-up in EM group. In this group, migraine remitted or improved (good migraine outcome) in 83% (49/59) of patients and 17% (10/59) of patients had a poor migraine outcome. The ‘seizure-free’ curves of patients with EM, who were stratified by migraine outcome, are shown in Fig. 2. Within the EM group, the cumulative probability of being seizure-free did not differ between the poor and good migraine outcome groups (log rank test = 1.34; P = 0.24).
In the epilepsy–migraine (EM) group, cumulative seizure-free curves during the 10 years of follow-up estimated by Kaplan–Meier analysis for patients with poor and good migraine outcome (log rank = 1.34; P = 0.2477). –––, Poor outcome; - - - -, good outcome.
Discussion
No previous report found that migraine is associated with a poorer outcome for epilepsy. Our findings show that patients with epilepsy and migraine had a lower chance of becoming seizure-free than patients with only epilepsy. We also found that epilepsy–migraine patients had a higher incidence of intractable seizures, a longer duration of the disease, a better chance of achieving remission with polytherapy and a lower early treatment response than the epilepsy controls. The system of active follow-up allowed us good estimates of the current seizure status for at least the last 2 years of follow-up. In comparison with the epilepsy control patients, the EM patients had significant seizure control and medication such as seizure control with polytherapy problems for at least the last 2 years of follow-up. These findings indicate that, as a group, epilepsy in patients with migraine was likely to have a poor prognosis.
Although this study represents the largest case–control study of an epileptic patient population with migraine, it was limited by the relatively small number of participants, because it was difficult to find an age-, sex-, and epileptic syndrome-matched control for each case for different types of epilepsy (subgroups of epilepsy syndromes). Although the differences we found were highly significant, the small subsamples of different epileptic syndromes in this study make the findings tentative. Whether the prognosis is similar across the various subtypes of epileptic syndromes or there is a specific association with one subtype is not clear. The cumulative seizure-free curves for the subtypes of epilepsy were not analysed because of the small number of subgroups. Larger studies are needed to determine whether any prognosis differences exist between the different types of epilepsy.
Many factors affect the prognosis of epilepsy, but it is probably largely determined by the underlying cause of the seizures, i.e. the particular epilepsy syndrome (15–25). Because remission of seizures was the most obvious measure of the course of the disease, we mostly studied variations in achieving seizure-free status rather than the other outcome variables.
The decision to select control patients with the same epilepsy syndrome without migraine at entry was due to the fact that we wanted the groups to have similar factors of known or suspected prognostic significance (such as the underlying cause of the seizures, the particular epilepsy syndrome, age, age at onset of seizure, the time to onset of treatment, the number of seizures before treatment, disease duration of admission, and follow-up time). A difference in the selection of treatments could not have accounted for the difference between the two groups of patients, because appropriate antiepileptic drugs for specific seizure types or syndrome have been used, first and foremost, in both groups. When an antimigraine AED was appropriate for the seizure type or syndrome, it was used in the EM group. As a result, we may be closer to measuring the effect of migraine itself, rather than the combined effects of other factors that may affect the prognosis of epilepsy. The present findings that the groups were similar with regard to epilepsy syndrome and other prognostic factors suggest that the risk of a poor prognosis in EM patients was probably linked with the migraine.
Because a poor migraine outcome would affect the prognosis of epilepsy in the EM group, we performed a subanalysis. A negative influence from the migraine severity on the outcome of epilepsy seemed unlikely. The subanalysis showed that the cumulative probability of being seizure-free for patients with EM was similar, whether migraine outcome was poor or not. Therefore, our data are reassuring, dismissing the possibility that migraine severity could contribute to the poor outcome of epilepsy.
The present results showed that comorbid migraine affected the prognosis of epilepsy: (i) longer duration of epilepsy, an intractable seizure disorder, and late treatment response were related to comorbid migraine; (ii) the chance of being seizure-free during the 10-year follow-up was lower in a person with epilepsy and comorbid migraine. That is, patients with migraine and epilepsy were more likely to relapse than those with only epilepsy.
The poor prognosis in EM patients raises the question about the relation between the poor prognosis and comorbid migraine. What does a poor prognosis mean? Why do patients with epilepsy and migraine fare worse than those with only epilepsy? We do not know the cause or causes of a poor prognosis in these cases. However, two approaches might help to explain the relation between the poor prognosis and migraine in epilepsy patients.
First, the concurrence of the two disorders suggested the hypothesis that both are caused by a genetic disorder (9, 10). These diseases have prominent genetic components. The difficulty in understanding these diseases arises from the complexity of the clinical phenotypes as well as from the genetic heterogeneity that almost certainly exists. The similarities between these disorders, including their episodic nature, precipitating factors, and therapeutic response, are striking. As a result, a shared genetic susceptibility to both disorders may be responsible for an underlying brain state that is common to both, and might lead to the poor prognosis of epilepsy. Genetic studies may provide more definitive answers. Second, neuronal hyperexcitability in migraine may be associated with poor seizure control. Perhaps an altered brain state characterized by neuronal excitability increases the risk of both migraine and epilepsy and accounts for their comorbidity (6, 8, 9, 28). The double effects of both disorders may increase neuronal excitability or decrease the threshold for epileptic attacks and may be responsible for the poor prognosis of epilepsy.
Regarding the number of epilepsy patients also affected by migraine, prospective studies comparing epilepsy patients with and without migraine are necessary. Our study has explored our understanding of the pathophysiology of these disorders and can provide some clues for further studies.
Footnotes
Acknowledgements
We gratefully acknowledge Dr Gamze Çan and Dr Murat Topbaş (Public Health Department, Karadeniz Technical University Medical Faculty) for their significant contribution to the statistical analyses; and Dr Sezer Ş. Komsuğlu (Neurology Department, Kocaeli University Medical Faculty), Dr Serap Saygı (Neurology Department, Hacettepe University Medical Faculty), and Dr Betül Baykan (Neurology Department, Istanbul University Medical Faculty) for their helpful criticisms and suggestions.
