Abstract
The introduction of the triptans brought advances in achieving complete and sustained pain resolution in migraine patients, compared with non-migraine-specific treatments. However, sustained pain-free rates for triptans recorded in many clinical trials are still relatively low. This may be due to study participants being treated late into the attack, when pain is already moderate or severe. Studies with almotriptan have shown that efficacy is enhanced when treatment is given early in a migraine attack while pain is still mild, compared with later administration when pain intensity is greater. Developments in our understanding of migraine pathophysiology provide a rationale for this phenomenon, with improved efficacy seen when abortive treatment is administered before central sensitization develops. A limited window of therapeutic opportunity exists early in an attack to improve the outcome of triptan treatment. Early intervention is recommended to avoid the significant pain and disability commonly associated with moderate or severe migraine.
Introduction
Migraine is a chronic, intermittent condition, which can be associated with significant pain and debilitation (1–3). Advances in our understanding of the pathophysiology of this disease have enabled significant progress in drug therapy and management strategies. The desirable goal for acute migraine treatment from a clinical and patient perspective is the rapid and sustained complete removal of pain and restoration of normal functionality when an attack occurs. The introduction of the triptans over a decade ago heralded a new era in migraine therapy, and research into their optimum use is continuing. Response to treatment taken late in the course of an attack, when pain and disability have become severe, is often reduced and knowledge of the progressive nature of migraine symptoms might explain this phenomenon. There appears to be a limited window of therapeutic opportunity early in the attack when treatment is particularly effective. This is corroborated by the results of recent clinical studies of early intervention with almotriptan in the acute treatment of migraine (4–6).
Efficacy of triptan therapy – an overview
The low level of patient satisfaction with migraine treatment revealed by the American Headache Study (7,8) suggests a need for improved efficacy in acute migraine treatment. Migraine patients desire a treatment that rapidly and completely relieves pain and prevents headache recurrence (7). Sustained pain-free is a composite endpoint that approximates these requirements. As discussed previously in this supplement, this outcome combines being pain free within 2 h with no use of rescue medication and no recurrence of moderate to severe headache within 24 h of medication. Sustained pain-free response is the most rigorous assessment of the efficacy of an acute migraine treatment, and is considered by the International Headache Society (IHS) to be the optimal final goal for development of such drugs (9). However, while sustained pain-free response is the most ideal efficacy endpoint, it is also the most difficult to achieve. Treatment tolerability is also an important issue for migraineurs. Recently, a survey revealed that concerns about adverse events cause 67% of patients to delay or avoid taking their prescription medications for migraine, causing additional pain and disability (10).
Beginning with the introduction of sumatriptan in 1991, the triptans clearly represent a significant advance in acute migraine therapy. These agents are indicated for the treatment of moderate to severe migraine and in patients whose mild to moderate migraine responds poorly to non-steroidal anti-inflammatory drugs (NSAIDS) or combination analgesics (11). Triptans show benefits over ergot derivatives by providing selective pharmacology, consistent pharmacokinetics and efficacy and a well-established safety record (12). Despite this advance, treatment is still not ideal, as many clinical trials have found sustained pain-free rates for the triptans to be relatively low. In the meta-analysis of the commercially available oral triptans (13), data from over 24 000 patients revealed that sustained pain-free rates for all the triptans and doses investigated ranged from 18% to 27%. Almotriptan 12.5 mg, eletriptan 80 mg and rizatriptan 10 mg were shown to have the highest sustained pain-free rates of all the oral triptans, being significantly more effective than sumatriptan 100 mg.
The reason for the relatively low rates of sustained pain-free response found in the literature may be due to the fact that most clinical trials designed to evaluate acute migraine therapies typically require patients to wait until the pain is moderate or severe in intensity before treating with study medication. This may serve to ensure that the headache being treated in the clinical study is, indeed, migrainous and establishes a situation where all patients have the possibility of a measurable improvement in headache severity (6). A potential breakthrough in the management of acute migraine involves the timing of treatment. Emerging data suggest that the pain-free efficacy of triptan therapy is enhanced when treating mild attacks compared with moderate to severe attacks (4,6,14,15). Early intervention when pain is mild may therefore provide a strategy for improving migraine treatment efficacy.
Evidence to support early intervention
One of the first studies to support early intervention in a migraine attack was the Spectrum Study (14). Cady and colleagues found that early triptan treatment, when pain was mild, produced a greater pain-free response than when administered later in the attack, once the pain had become moderate to severe. The study was a posthoc analysis performed in 26 patients with disabling headache who treated a total of 46 mild and 166 moderate or severe headaches with oral sumatriptan 50 mg or placebo. Headaches treated while pain was mild were analysed separately and compared with treatment of moderate or severe pain to examine the effect of early intervention, and statistical comparisons with placebo were carried out. In general, more patients with mild pain reached a pain-free status with triptan treatment than those with moderate to severe pain. Across all headaches treated, pain-free response at 2 h was 50% with sumatriptan vs 0% for placebo when pain was mild, compared with 27 vs 6% when pain was moderate or severe. At 4 h, pain-free rates were 85% with sumatriptan vs 17% for placebo when treatment was administered while pain was mild, compared with 48% for sumatriptan vs 19% for placebo when pain was moderate or severe. Significance tests vs placebo were performed for the first-treated attacks. When pain was mild on treatment administration, 2-h pain-free rates were 52% for sumatriptan vs 0% for placebo. Conversely, when pain was moderate to severe, pain-free rates were somewhat lower (29% for sumatriptan vs 8% for placebo). A similar pattern was seen at 4 h postdose: treatment when pain was mild produced a pain-free response rate of 78% with sumatriptan, which was significantly higher than placebo (0%, P = 0.02), compared with a lower rate of 50% for sumatriptan vs 17% for placebo when pain was moderate to severe. A trend towards lower headache recurrence in attacks treated while pain was mild was also observed compared with when pain was moderate or severe (13 vs 18%). These observations led the authors to conclude that patients with disabling migraine may benefit from early intervention with a triptan while pain is mild.
Recent developments in our understanding of migraine pathophysiology have provided a rationale for early intervention. Cutaneous allodynia, whereby non-painful stimuli are perceived as painful, develops in 80% of migraine sufferers in the later stages of an attack (16). Research has shown that migraine pain is progressive within an attack, with initial peripheral activation and sensitization affecting the intracranial blood vessels and the meninges. Later, peripheral pain fibres become hypersensitive mediating the throbbing pain of migraine, which is worsened by movement. When additional central sensitization of the trigeminal nucleus caudalis occurs, this is the phase when cutaneous allodynia develops (16,17). This interpretation of the pathophysiological mechanisms of migraine supports the early use of antimigraine drugs that target peripheral nociceptors, before the development of central sensitization. Theoretically, early treatment before pain progresses to moderate or severe intensity could avoid or reduce sensitization and, consequently, improve treatment response.
Preliminary observations from Burstein and colleagues in migraine patients suggest that the presence of cutaneous allodynia influences the response to triptan treatment and that early treatment can prevent these distressing symptoms (15). In 30 migraine attacks studied, cutaneous allodynia had not developed in 10 of the attacks by the time of triptan administration. In these attacks, triptan therapy completely relieved the headache and prevented the development of allodynia. However, in 18 of the 20 attacks where cutaneous allodynia had already established, triptan therapy provided little or no relief from the headache and was ineffective in suppressing the ongoing allodynia. Nevertheless, late triptan therapy in patients with allodynia did abolish the throbbing pain associated with migraine, suggesting that the triptan was acting at the peripheral neuronal sites. These findings suggest that central sensitization plays a key role in maintaining the headache and that triptan therapy abolishes the pain prior to, but not after, the establishment of central sensitization. Such observations serve to establish the clinical basis for early treatment with a triptan during a migraine attack.
Regarding the timing of intervention, as central sensitization and the associated cutaneous allodynia develop during the first hour of an attack in most migraine patients (16), there appears to be a limited window of therapeutic opportunity. In the race against allodynia it is important therefore for the therapeutic level of a triptan to be reached in the first hour of a migraine attack in order to offer the maximum potential of enhanced efficacy (Fig. 1).

The therapeutic window of opportunity for early triptan therapy. Reproduced from (16) with permission. © 2000 Oxford University Press.
Early treatment with almotriptan
A number of recent studies have assessed the efficacy of almotriptan when given early in an attack. A posthoc analysis of an open-label European study has also addressed the potential benefits of intervention with a triptan during the mild phase of a headache attack (6). In this study, a total of 762 migraineurs used oral almotriptan 12.5 mg for headache attacks of any severity. The design of the trial allowed direct efficacy comparisons between treatment of mild pain and treatment of moderate or severe pain. Specifically, the posthoc analysis evaluated pain-free rates at 1 and 2 h, use of rescue medication, incidence of recurrence and adverse events in those patients who had treated at least three mild attacks and three moderate or severe attacks. A total of 118 migraineurs and 708 attacks were included in the analysis, with equal numbers of attacks involving treatment when pain was mild and when pain was moderate or severe. A pattern similar to that of the Spectrum Study was found. At 1 h following almotriptan treatment, pain-free status was achieved in significantly more mild attacks compared with moderate or severe attacks (47 vs 14%; P < 0.001) (Fig. 2). At 2 h, 84% of mild attacks vs 53% of moderate or severe attacks treated with almotriptan were pain free (P < 0.001). The incidence of headache recurrence was significantly lower for mild attacks compared with moderate or severe attacks (28 vs 33%; P < 0.01). Rescue medication was used significantly less often in mild attacks compared with in moderate or severe attacks (8 vs 13%; P < 0.01). The incidence of adverse events with almotriptan remained low irrespective of when treatment was administered (6% for mild vs 7% for moderate to severe attacks). The results of this study confirmed that treating early with almotriptan during a migraine attack when the pain was mild (i.e. before it became moderate or severe) significantly improved patient outcomes.
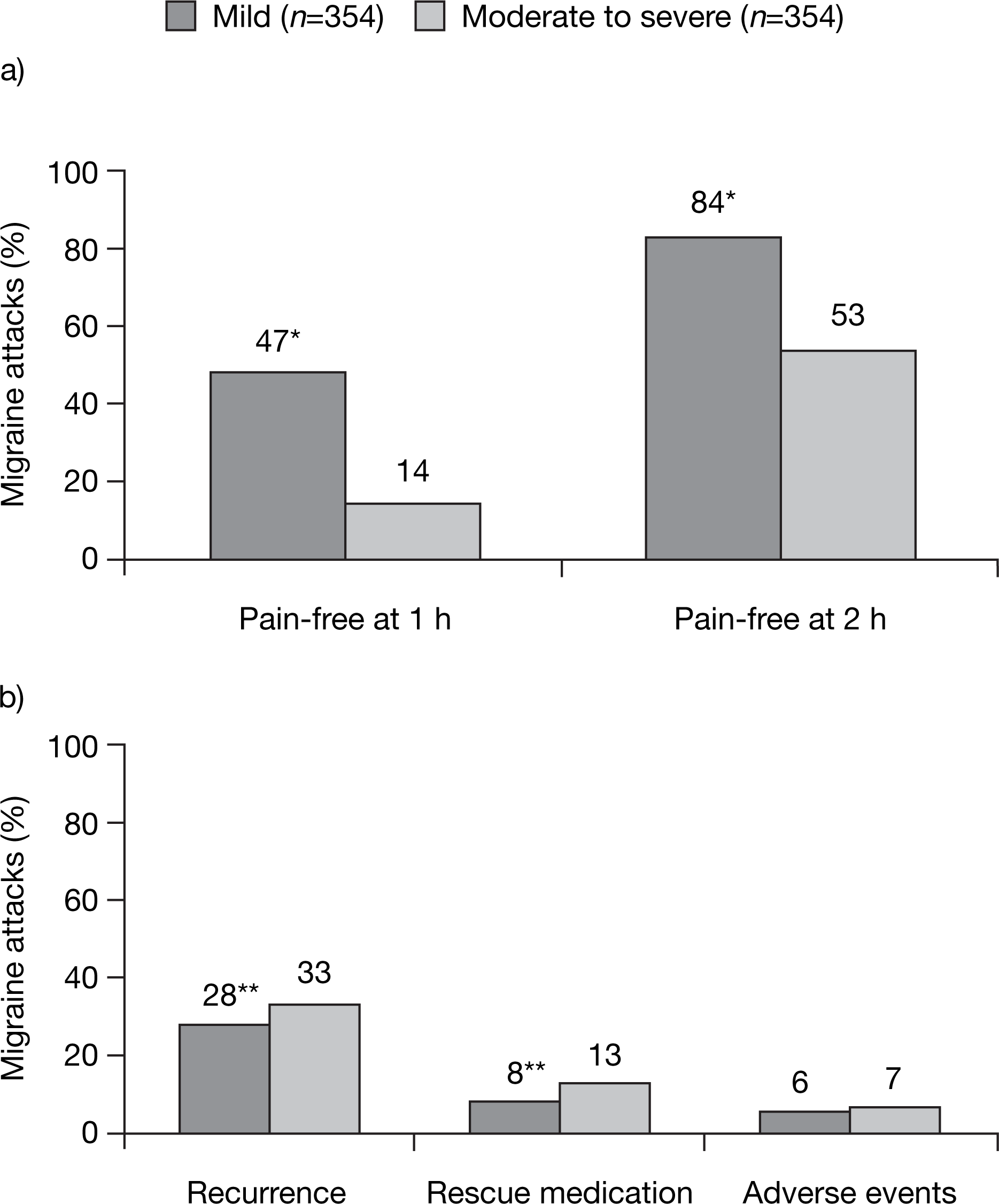
Proportion of headaches with: (a) pain-free response; (b) headache recurrence, use of medication and adverse event rates associated with early versus late treatment with almotriptan. ∗P < 0.001, ∗∗P < 0.01; versus moderate to severe pain. Reproduced from (6) with permission. © 2002 Blackwell Publishing Ltd.
A posthoc analysis has been made of an open-label, multicentre, long-term trial in the USA which evaluated whether treatment of migraine with almotriptan when pain intensity was mild improved 1- and 2-h pain-free and sustained pain-free rates compared with treatment when pain intensity was moderate or severe (5). Patients were instructed to take a single dose of almotriptan 12.5 mg at the onset of a migraine attack of any severity. This protocol was unlike most clinical trials which typically limit the administration of the study drug to attacks of moderate or severe pain. A second dose of almotriptan could be taken if headache recurred within 24 h of the initial dose. Use of rescue medication and recurrence of pain (i.e. return of moderate or severe pain within 2–24 h of taking the study medication) were also recorded. Sustained pain-free rates, defined as being pain free at 2 h with no recurrence or use of rescue medication up to 24 h postdose were calculated. Evaluations for the posthoc analysis were derived from 582 patients treating 10 645 attacks with at least one dose of almotriptan. The results of the study showed that a higher proportion of migraine attacks treated when pain intensity was mild achieved a pain-free status at 1 h (35.3%) compared with attacks treated when pain intensity was moderate or severe (7.5%; P < 0.001) (Fig. 3a). Pain-free rates at 2 h were also significantly higher when pain intensity at dose administration was mild (76.9%) compared with when intensity was moderate or severe (43.9%; P < 0.001). Recurrence of headache and use of rescue medication were also reduced when attacks of mild intensity were treated. Headache recurred in 12.9% of those with mild pain vs 25.0% in the moderate or severe pain group (P < 0.001). Rescue medication was used in 9.4% of mild attacks vs 17.2% for moderate or severe attacks (P < 0.001) (Fig. 3b). Sustained pain-free rates were nearly twice as high when attacks were treated during mild pain (66.6%) than when attacks were treated during moderate or severe pain (36.6%, P < 0.001) (Fig. 3c). These findings of improved pain responses when almotriptan treatment is administered when attacks are mild in intensity support the use of early intervention with triptans in the acute management of migraine.

Comparison of: (a) pain-free rates at 1 and 2 h; (b) recurrence and use of rescue medication; and (c) sustained pain-free rates in the treatment of mild versus moderate to severe migraine pain. ∗P < 0.001 vs moderate to severe pain intensity. Data from (5).
An analysis of a double-blind study investigated the effect of time to triptan treatment on the clinical response in migraine patients (4). In the original study, 668 patients with a history of migraine for at least 1 year who met IHS criteria for migraine with or without aura were randomized to a single oral dose of almotriptan 12.5 mg, almotriptan 25 mg, sumatriptan 100 mg or placebo (18). Patients were instructed to take their medication at the onset of a migraine attack of moderate or severe intensity. The primary efficacy endpoint was pain relief at 2 h (defined as improvement from moderate or severe pain to mild or no pain 2 h after dose administration). Secondary endpoints included pain relief at 1 h, pain free at 1 and 2 h, headache recurrence and use of rescue medication within 24 h of dose administration. A posthoc analysis was carried out for patients receiving either almotriptan 12.5 mg, sumatriptan 100 mg or placebo who took the study medication within 1 h of migraine onset (i.e. excluding patients taking almotriptan 25 mg). The analysis focused on the efficacy parameters of pain-free at 2 h and sustained pain-free response (defined as being pain free at 2 h with no rescue medication and no recurrence of moderate or severe headache within 24 h of medication). Of the 477 patients reviewed, 253 (53.3%) treated their attack within 1 h of onset so were included in the analysis. Compared with placebo, treatment within 1 h of attack onset achieved significantly higher 2-h pain-free rates for almotriptan (37.9%vs 18.9%; P = 0.016) and for sumatriptan (35.7 vs 18.9%; P = 0.028) (Fig. 4). Initiation of treatment within the first hour after acute migraine onset resulted in a significantly higher sustained pain-free response for almotriptan compared with placebo (34.7 vs 17.0%; P = 0.022). Sustained pain-free rates with sumatriptan were greater than with placebo although not significantly so (29.6 vs 17.0%) (Fig. 4). This analysis supports the early intervention with almotriptan to improve outcomes in acute migraine attacks.

Percentage of patients pain-free at 2 h and with sustained pain-free response when treated within 1 h of migraine attack onset. ∗P = 0.016, †P = 0.028, ‡P = 0.022; versus placebo. Reproduced with permission from (4). © 2004 Blackwell Publishing Ltd.
Two large Spanish studies, known as Tolerability and Efficacy of Almogran® (TEA 2000 and TEA 2001), have investigated the impact of early treatment in the context of routine clinical practice (19–21). These observational studies were carried out by 317 neurologists (recruiting 1643 patients diagnosed with migraine) and by 640 primary care physicians (including 2074 migraine patients), respectively, in a study of efficacy and tolerability of almotriptan 12.5 mg. Each study had the same protocol design. Patients with migraine, as defined by IHS standards, were instructed to report data on their migraine attacks in a diary for 3 months, including their experience of the efficacy and tolerability of almotriptan 12.5 mg used to treat these attacks. Data from 4253 and 4183 attacks were generated by the two studies. Patients taking almotriptan at the beginning of the attacks when pain was still mild showed the best results (Fig. 5). In the neurologist study, when treatment was taken when pain was mild 59.5% of patients were pain-free at 2 h, compared with 23.2% when treatment was administered once pain had escalated to moderate or severe intensity. A similar pattern was seen in the patients recruited by primary care physicians, with pain-free rates of 80.5% when pain was mild and 48.5% when pain was moderate or severe. Sustained pain-free rates were also enhanced when treatment was taken early in the attack. In the neurologist-led study, the rates were 43.9% with early almotriptan treatment vs 16.1% when treatment was administered once pain had become moderate to severe. In the physician-led study, sustained pain-free rates were 70.9% for mild vs 43.0% for moderate to severe pain intensity on treatment. Headache recurrence was low in both studies (21.2% in the neurologist study and 10.7% in the physician study). The incidence of adverse events was also low in both studies (3.9% in the neurologist and 1.1% in the physician study), with a very low incidence of chest pain (0.2% in the neurologist study and 0.1% in the primary care physician study) and cardiovascular symptoms (0.4% in the neurologist study and 0.1% in the physician study). Therefore, these studies show that the key features of almotriptan revealed in clinical trials – i.e. placebo-like tolerability and rapid and sustained efficacy, particularly when administered early in an attack – are also apparent in clinical practice.
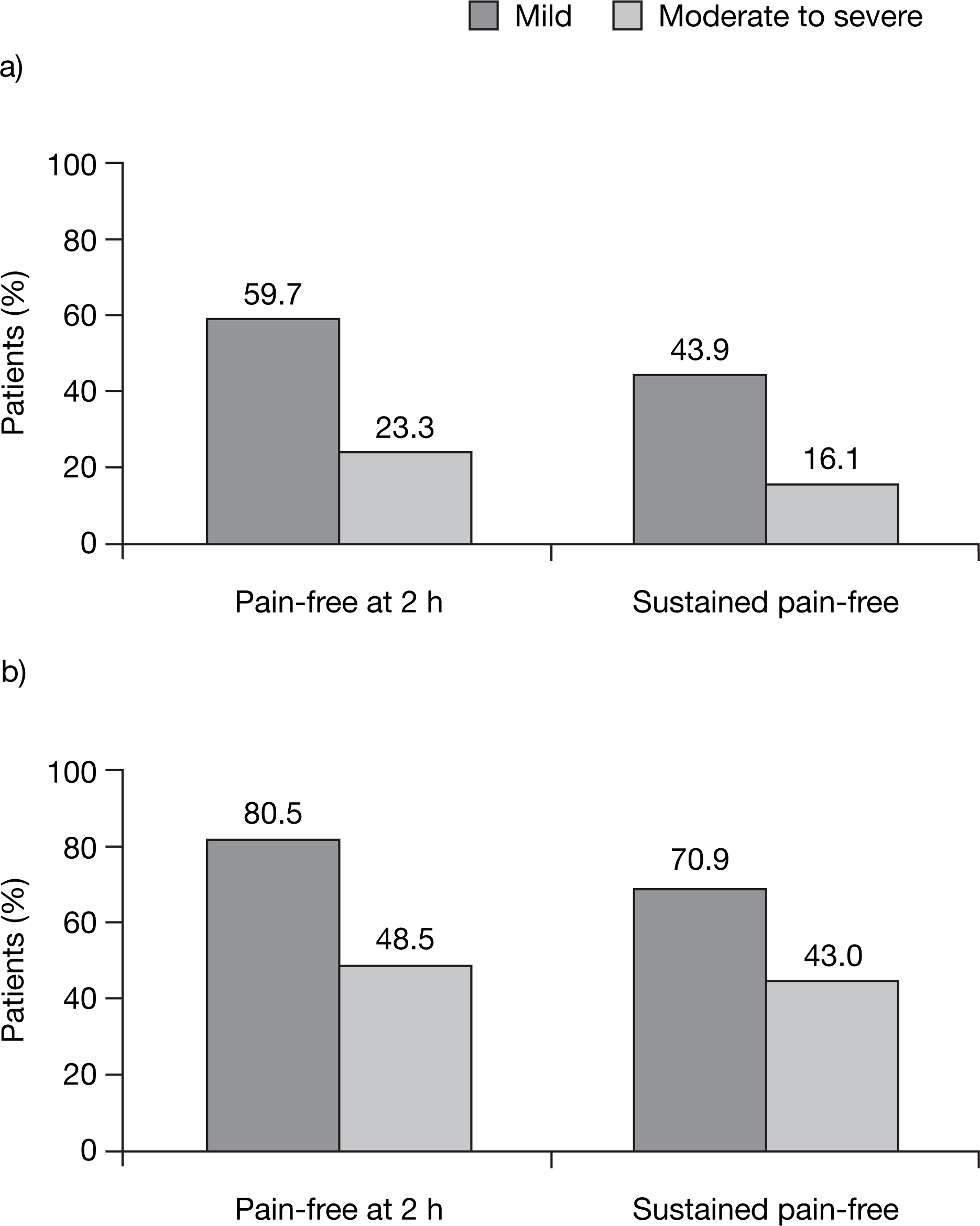
Pain-free at 2 h and sustained pain-free rates with almotriptan in clinical experience studies led by: (a) neurologists; and (b) primary care physicians. Data from (21).
Early intervention strategies
Some controversies remain regarding the initiation of intervention during the mild phase of a migraine episode. For example, the early treatment approach may not be suitable for all migraine sufferers, as attacks are highly variable and tension-type headache commonly co-occurs. However, early intervention can improve treatment outcomes, increase patient satisfaction and may therefore help reduce the burden of migraine on society and on the individual. As discussed above, early treatment might also prevent central sensitization and prevent attack progression. Moreover, it may prevent disease progression in susceptible people, although further investigation is required to confirm this hypothesis. From the available evidence, early intervention with triptans in patients with a high level of headache-related disability or headache recurrence is recommended. Data from population studies suggest that this may represent a large subgroup of migraineurs. A major US study has shown that more than 80% of migraine patients suffer from severe or extremely severe headache, and 62% of respondents experienced one or more severe headache per month (2,3). Furthermore, 91% of migraineurs report functional impairment with their headaches, with more than half of the total (53%) reporting severe impairment in activity or the requirement for bed rest with severe headaches.
Conclusions
Advances in our understanding of the pathophysiological mechanisms of migraine have led to improvements in drug therapy and management strategies, and will continue to do so. Clinical trials of triptans, which typically delay treatment until headache has reached moderate to severe intensity, have found sustained pain-free rates ranging from 18% to 27% for the oral triptans. However, recent clinical evidence clearly demonstrates that treatment efficacy is enhanced when taken early in the attack when pain is mild. This premise is based on efficacy parameters, such as headache response, pain-free responses and recurrence rates, as well as utilization of rescue medications and the composite endpoint of sustained pain-free rates. The development of cutaneous allodynia resulting from peripheral and central sensitization serves as the rational basis for early treatment. A limited window of therapeutic opportunity of approximately 60 min appears to exist in most migraine patients (Fig. 1). Additionally, early intervention may help patients to avoid the significant pain and disability associated with moderate or severe migraine.
The excellent tolerability and favourable efficacy profile of almotriptan has been demonstrated both in clinical trials and in clinical practice, and a meta-analysis of oral triptans identified almotriptan to be among the three agents most likely to achieve consistent treatment success. Furthermore, emerging data are revealing enhanced efficacy with almotriptan when treatment is administered early in a migraine attack, supporting its use in early intervention strategies.
