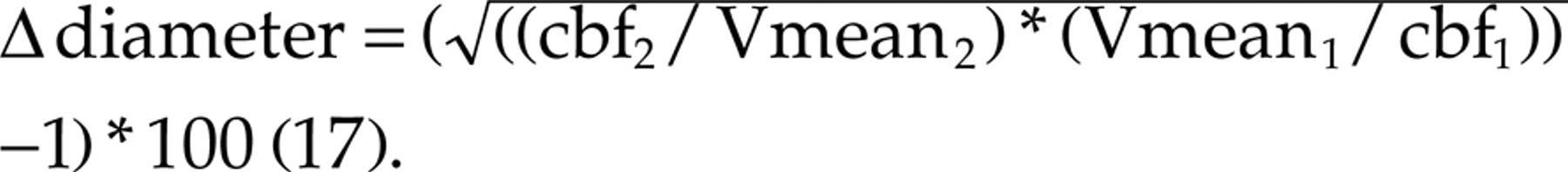Abstract
The aim of this study was to estimate the effect of Nitric Oxide synthase (NOS)-inhibition (L-NMMA) on the diameter of the middle cerebral artery (MCA) and on regional cerebral blood flow (rCBF). Furthermore, to assess the effect of L-NMMA on acetazolamide induced increases in MCA blood velocity (Vmean) and rCBF. In an open crossover design 12 healthy subjects attended the laboratory twice. The first day 6 mg/kg L-LNMMA i.v. over 15 min preceded 1 g acetazolamide iv over 5 min. Eight days later only acetazolamide was given. Vmean in MCA was determined with transcranial Doppler (TCD) and rCBF with Xe-133 inhalation SPECT at baseline, after L-NMMA and 25 and 55 min after acetazolamide infusion. After L-NMMA the decrease in rCBFMCA was 6.8% (± 7.4) (P < 0.019, n = 12), whereas Vmean was not affected (P = 0.83, n = 8). The change in MCA diameter was estimated to -1.3% (P = 0.44, n = 8). L-NMMA did not affect acetazolamide increases in Vmean (P = 0.67, n = 8) nor rCBF (P = 0.29, n = 12). The percentage increase of Vmean was 1.5 times that of rCBF (n = 8). Our data suggest that the basal tone of human cerebral arterioles but not of conduit arteries is NO-dependent. The action of acetazolamide in man is not NO-dependent.
Keywords
Introduction
Nitric oxide (NO) is an almost ubiquitous messenger molecule with many different functions in the human body (1). It also plays a role in several diseases of the brain including migraine and stroke (2–4). Nitric oxide synthases (NOS), the enzymes that catalyse the production of NO, can be inhibited by several substances (5). NG monomethyl
From animal experiments it is evident that NO participates in the maintenance of resting cerebral blood flow (CBF), although with species differences (6). Only few human studies deal with the effect of NOS inhibition on resting CBF or on the diameter of cerebral and cephalic arteries (7, 8, 9, 10).
NO is an important molecule in migraine pain (11) and a pilot study has indicated that L-NMMA is effective in the treatment of acute migraine attacks without aura (2).
The aura that can precede migraine headache is attributed to cortical spreading depression and is accompanied by markedly reduced CBF (12, 13). If L-NMMA or other nonspecific NOS- inhibitors markedly decrease CBF and/or constricts cerebral arteries, that would cause concern about their safety as drugs for migraine. The effect of L-NMMA on MCA diameter and CBF is therefore of clinical importance.
In the present study we estimate the effect of L-NMMA on the diameter of MCA and on global and regional CBF in man using Transcranial Doppler ultrasonography (TCD) and single-photon emission computed tomography (SPECT). Furthermore, we assess the effect of L-NMMA on acetazolamide (Diamox ®) induced increases in MCA blood velocity (Vmean) and rCBF.
Material
Twelve nonsmoking healthy subjects (4 females, 8 male; mean age 26 years, range 20–38 years; mean weight 74 kg, range 57–95 kg) were included. No daily medication except contraceptives was allowed. Before inclusion a physical examination including ECG had to be within normal limits. The subjects were not allowed to take medication, coffee, tea or alcohol for 12 h before the study.
Exclusion criteria were: Use of any kind of daily medication, except oral contraceptives; pregnancy or breastfeeding; excessive use of analgesics or alcohol; serious somatic or psychiatric disorders; ischaemic heart disease; a supine resting diastolic blood pressure more than 90 mmHg or a heart rate less than 50 bpm at entry of study. Volunteers were informed that they were free to withdraw at any time and all volunteers gave written consent. The study was approved by the local ethics committees of Copenhagen and Copenhagen County (KA 95090 gs) and complied with the Declaration of Helsinki.
Design and procedure
The study used a controlled crossover but open design. The subjects attended the laboratory on 2 different study days separated by at least 1 week. On day 1 L-NMMA(546C88, GSK, UK) 6 mg/kg was given i.v. over 15 min preceding infusion of acetazolamide (diamox®, Lederle, Germany) 1 g iv over 5 min. On day 8 only acetazolamide (1 g acetazolamide i.v. over 5 min) was given. L-NMMA was infused intravenously by a volumetric pump (Braun® Perfuser) and acetazolamide was infused intravenously by hand.
On both study days a cannula (Viggo Venflon® 1.4 mm) was placed in the right cubital vein for L-NMMA/acetazolamide infusion. Baseline values of mean maximal blood velocity (Vmean) in the Middle Cerebral Artery (MCA), regional cerebral blood flow (rCBF), blood pressure, heart rate and pCO2 were recorded after 30 min of rest in the supine position in a quiet room. The subjects had to stay supine throughout the study, and were asked to close their eyes and speaking was not allowed during the measurements.
On day 1 rCBF was recorded four times; at baseline, immediately after end of L-NMMA infusion, 25–30 min, respectively, 55–60 min after start of acetazolamide infusion.
On day 8 rCBF was only recorded three times: at baseline, 25–30 and 55–60 min after start of acetazolamide infusion.
On both days of study Vmean in the MCA was measured every 10 min until the last CBF measurement. Blood pressure and heart rate were recorded every 5 min and end tidal pCO2 was recorded simultaneously with Vmean and CBF throughout the study. The experimental time schedule is shown in Fig. 1.

The experimental time schedule.
Methods rCBF was measured with a highly sensitive, brain-dedicated, fast-rotating, single photon emission computerized tomograph (SPECT) (Tomomatic 232). Each study lasted 4.5 min. A mixture of atmospheric air and xenon − 133 was re-breathed during the first 1.5 min through a closed system from a 4 litre reservoir (740 MBq/l). During the last 3 min the xenon-133 mixture was expired against atmospheric air. rCBF was recorded simultaneously in two slices positioned 50 mm and 90 mm above and parallel to the orbito-meatal plane (OM). Each slice was 16 mm thick and the distance between the centres of slices was 40 mm. The FWHM resolution of the instrument is about 16 mm in the transaxial plane. rCBF was calculated according to Celsis et al. (14).
A fixed matrix of region of interests was superimposed on the rCBF picture. The shape and size were fitted to the outlines on the brain excluding extra cranial flow and regional mean values were calculated within the predefined regions of interest. The matrix was divided into regions of interest representing the hemispheric rCBF regions and the vascular territories of supply by the anterior-, middle- and posterior cerebral arteries. The maximum whole body radiation was approximately 0.6 mSV per rCBF measurement (15). TCD recordings were only performed in the first 8 enrolled patients. For comparison of Vmean and rCBF we calculated rCBF in the territory of the MCA on the side of the head used for transcranial Doppler examination (n = 8). For the remaining patients (n = 4) the rCBF measurement from the right side of the head was used. Area weighted data from the OM + 50 mm and OM + 90 mm were used for all the results given below.
Time averaged mean of the maximal blood velocity (Vmean) in MCA was measured with transcranial Doppler ultrasonography(2 MHz, Multidop X Doppler: DWL, Sipplingen, Germany). A mean of four consecutive values of Vmean was taken (each representing the mean value of typically 4–5 heart beats automatically calculated by the computer). Recording the position in relation to the angle and distance relative to the orbito-meatal line reproduced positions of the measurements from day to day. Since rCBFmca = Vmeanmca × πr2 (16), the change in MCA diameter is given by the following equation:
Simultaneously with each rCBF and TCD measurement the end expiratory pCO2 was recorded by means of a capnograph (Datex OY, CD 101). Blood pressure and heart rate were measured with an automatic inflatable arm cuff (Omega 1400, Invivo Research Laboratory, New York, USA).
Statistics
Hemodynamic responses are given as mean and standard deviation (± SD). Otherwise nonparametric testing was used because of the small number of subject used for tcd recordings (n = 8).
To test the effect of L-NMMA on basal rCBF and Vmean in MCA the difference between CBF before and after L – NMMA is tested with Wilcoxons test. In the same way is the difference between Vmean before and after LNMMA tested.
To test the effect of L-NMMA on acetazolamide induced changes in rCBF and Vmean (MCA), the delta values for CBF (time −5 min – time 25 min) between day 1 and day 2 are compared with Wilcoxons test. So is the delta value for CBF (time −5 min – 55 min) between day 1 and day 2 compared. In the same way are the delta values for Vmean between day 1 and 2 compared.
Changes in rCBF, Vmean, blood pressure, heart rate and pCO2 over time within group each study day are tested with Friedmans test (Statgraphics® 7.0). In all tests P < 0.05 are considered statistically significant.
Results
Before L-NMMA and acetazolamide infusions it was tested whether the CBF recordings had any influence on simultaneously recorded Vmean in MCA (a possible effect of resistance in the mouth-peace during CBF measurements). No significant difference was found between Vmean during CBF and outside CBF (from 100% to 101% (± 7.6) (P = 0.81, Wilcoxon)). These data were pCO2 corrected according to (18):
rCBF(korr) = rCBF(meas)∗ eksp(0.04(pC02(Basal) − pCO2(n))),
where n is the number of measurement.
The effect of L-NMMA on basal Vmean in MCA and rCBF
Although pCO2 did not change significantly during L-NMMA infusion (P = 0.74, Friedman) all rCBF and TCD data were corrected for changes in pCO2 according to (18):
rCBF(korr) = rCBF(meas)∗ eksp(0.04(pC02(Basal)− pCO2(n))).
rCBF
The mean decrease in rCBFMCA was 6.8% (± 7.4) after L-NMMA infusion (P < 0.019, n = 12,wilcoxon). In absolute data the decrease in rCBF was from 61.8 ml/100 g min (± 8.8) to 57.5 ml/100 g min (± 8.3), Fig. 2. During L-NMMA infusion rCBF decreased significantly in the right MCA territory (P = 0.0067); the left MCA territory (P = 0.010); the right ACA territory (P = 0.011) and in the left ACA territory (P = 0.017). In the right and left PCA territory there was only a tendency to a flow reduction (P = 0.11 and P = 0.05).

The effect of L-NMMA on Vmean (MCA) and rCBF. Simultaneous measurements of regional cerebral blood flow (rCBF) in the territory of the middle cerebral artery (MCA) and of mean maximal blood velocity (Vmean) in MCA before and after infusion of L-NMMA. rCBF (•) decreased 6.8% ± 7.4 compared to baseline (∗P < 0.05, n = 12). No change of Vmean (▪) was observed after L-NMMA infusion (P = 0.83, n = 8).
TCD
There was a minor nonsignificant decrease in Vmean after L-NMMA infusion − 2.4% (± 13.5) (P = 0.83, Wilcoxon), Table 1, Fig. 2.
L-NMMA induced changes in rCBF, Vmean, blood pressure and heart rate

Values are given as mean ± SD.
indicates P < 0.05.
There was no significant L-NMMA induced changes in MCA diameter:
Δ diameter: −1.298% (∼−1.3%) (P = 0.44, Wilcoxon, n = 8) (16, 17).
Other haemodynamics
Systolic blood pressure increased 3.9% (± 4.0) (P < 0.05, Wilcoxon) and diastolic blood pressure 17% (± 6.6) (P < 0.005, Wilcoxon) due to L-NMMA infusion. Heart rate decreased 22.1% (± 9.9) (P < 0.005, Wilcoxon), Table 1.
The effect of L-NMMA on acetazolamide induced changes in Vmean (MCA) and rCBF
In these calculations neither rCBF nor Vmean data were corrected for changes in pCO2 due to the similar mechanisms of acetazolamide on pCO2.
rCBF
Acetazolamide induced a significant increase in rCBF both with and without L-NMMA pretreatment (P < 0.00005 and P < 0.0002, Friedmans test). When acetazolamide was preceded by L-NMMA (day 1) the mean rCBF increase from baseline was 30.9% (± 13.1) at 25 min and 24.2% (± 15.2) at 55 min after start of acetazolamide. Without L-NMMA pretreatment (day 8) the mean rCBF increase was 26.2% (± 13.0) at 25 min and 28.0% (± 16.2) at 55 min after start of acetazolamide. The response was not significantly different between L-NMMA and no pretreatment, neither at time 25 min nor 55 min after start of acetazolamide (P = 0.29, and P = 0.23, Wilcox), Fig. 3, Table 2.
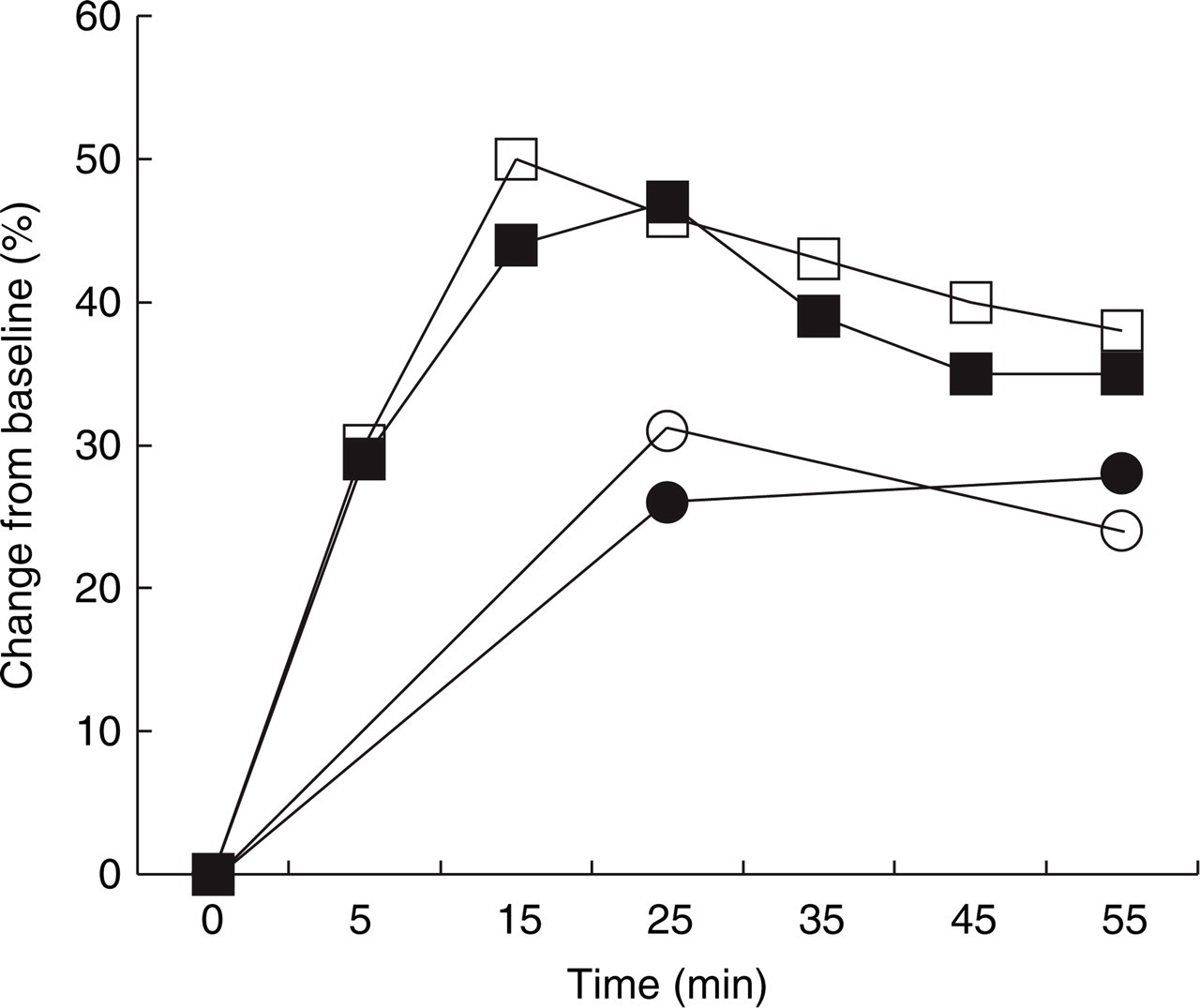
Acetazolamide induced increase in Vmean and rCBF with and without L-NMMA pretreatment. Acetazolamide induced a significant increase in rCBF both with (○) (P < 0.00005) and without L-NMMA pretreatment (•) (P < 0.0002), but there was no significant difference between L-NMMA and no pretreatment, neither at time 25 min (P = 0.29) nor 55 min after start of acetazolamide (P = 0.23, n = 12). Acetazolamide also induced a significant increase in Vmean With L-NMMA pretreatment (□) (P < 0.0006) and without L-NMMA (▪) (P < 0.0001), but there was no significant difference between study days, neither 25 min (P = 0.67) nor 55 min after start of acetazolamide (P = 0.62, n = 8).
The effect of acetazolamide with and without L-NMMA pretreatment

Changes in rCBF, Vmean, blood pressure, heart rate and end tidal pCO2 due to acetazolamide infusion ± SD.
TCD
With L-NMMA pretreatment (day 1) acetazolamide increased Vmean 45.7% (± 19.7) at 25 min and 38.1% (± 31.0) at 55 min (P < 0.0006, Friedman). When acetazolamide was administered without l-NMMA pretreatment (day 8) Vmean increased 46.9% (± 18.5) 25 min and 35.1% (± 19.3) 55 min after acetazolamide (P < 0.0001, Friedman), but there was no significant difference between study days, neither at time 25 min nor 55 min after start of acetazolamide (P = 0.67, and P = 0.62, Wilcoxon), Fig. 3, Table 2.
Twenty-five minutes after start of acetazolamide (without L-NMMA pretreatment) the percentage increase from baseline of Vmean was 46.9% (± 18.5) vs. 30.7% (± 6.8, n = 8) in rCBF. After 55 min the percentage Vmean increase was 35.1% (± 19.3) vs. 24.9% (± 18.1, n = 8) in rCBF.
Other haemodynamics
The systolic blood pressure did not change over time due to acetazolamide neither after L-NMMA pretreatment (day 1) (P = 0.71, friedman) nor without pretreatment (day 2) (P = 0.50, friedman), and there was no significant difference in systolic blood pressure between study days neither at time 25 min (P = 0.76, Wilcox) nor at time 55 min after acetazolamide (P = 0.69, Wilcox).
The diastolic blood pressure decreased significantly over time after L-NMMA pretreatment, day 1 (P < 0.007, friedman), but not without pretreatment, day 8 (P = 0.21, friedman). The diastolic blood pressure was significantly different between study days both at time 25 min (P < 0.005, Wilcox) and at time 55 min after diamox (P < 0.05, Wilcox). The heart rate increased significantly over time after L-NMMA pretreatment, day 1 (P < 0.0003, friedman), but not without pretreatment, day 8 (P = 0.11, friedman). Heart rate was significant different between study days both at time 25 min (P < 0.05, Wilcox) and at time 55 min (P < 0.007, Wilcox), Table 2.
Other symptoms and signs
No pathological ECG changes were observed. No symptoms or signs were reported after L-NMMA but after acetazolamide perioral paresthesia and paresthesia in distal extremities were reported in several subjects. One subject reported a feeling of fullness in the head and another reported a mild headache. Furthermore, acetazolamide had a diuretic effect.
Discussion
We found that L-NMMA in the dose 6 mg/kg given over 15 min slightly but significantly decreased global and regional cerebral blood flow (rCBF), whereas the blood velocity in the middle cerebral artery (Vmean) was not affected. No significant change in the MCA diameter calculated from rCBF and Vmean was observed. L-NMMA did not affect acetazolamide increases in Vmean or rCBF. The percentage increase of Vmean due to acetazolamide was 1.5 times that of rCBF indicating a constrictor effect of acetazolamide on large cerebral arteries.
Nitric oxide in the regulation of resting cerebral blood flow
From animal experiments it is evident that NO participates in the maintenance of resting CBF, although with differences due to choice of NOS-inhibitor, species and route of administration (6). A direct extrapolation from animal studies to studies in man is therefore not possible and only few studies of the effect of NOS inhibition on resting CBF have been conducted in man. Recently, the effect of 3 mg L-NMMA//kg over 5 min on resting CBF was studied in the basilar and in both internal carotid arteries by the use of a gradient echo pcMRI technique. At this small dose there was no significant change in CBF (9). When a bolus of 10 mg L-NMMA/kg followed by 10 mg/kg/h was used in a PET study of resting global CBF, a 20% decrease was found (10).
The effect of L-NMMA on basal CBF has also been assessed by colour velocity ultrasonic imaging of internal and common carotid artery volume flow. An intravenous bolus of 10 mg/kg L-NMMA produced a 15% reduction in basal mean ICA flow while flow in CCA was reduced with 30% (8).
In sedated, nimodepine treated patients with contra-lateral vascular pathology, CBF has been determined by intracarotid 133Xe injection technique after intracarotid administration of L-NMMA 50 mg/min for 5 min. The intra-arterial concentration was estimated to be approximately 25-fold greater than if injected intravenously and it produced a 13–20% decrease in rCBFMCA (19).
In the present study we found that L-NMMA infusion (6 mg/kg i.v.over 15 min) significantly reduced resting rCBF (6.8%) in the MCA territory. Significant reductions of rCBF were found in both ACA and MCA territories, but in the territory of the posterior cerebral artery, there was only a tendency for a decrease.
We cannot exclude the possibility that the timing of L-NMMA inhibition has been too close to the CBF measurements and a more pronounced decrease in CBF would have been observed if we had delayed the CBF measurement. In a closed cranial window model of rats the CBF response to whisker stimulation was only minimally decreased 30 min after topical administration of the NOS-inhibitor L-NA but 60 min after application the attenuation of rCBF was significant (20). However, we regard this as unlikely because we measured at the time of peak blood pressure response. Furthermore, the size of rCBF reduction is smaller than the effect observed by authors using a higher dose (10) and larger that what has been observed with a smaller dose (9).
Our controlled, crossover study can be criticized for lack of blinding. The open design without placebo was chosen in order to reduce radiation exposure (15) yet be able to not only determine the effect of L-NMMA but also its effect on the acetazolamide response. Our study used computerized calculations and it is highly unlikely that blinding would alter such results. For the same reasons we did not apply L-Arginine after L-NMMA to test the specific effect of NOS inhibition (21).
L-NMMA resulted in an increase in MAP. This is unlikely to have reduced the magnitude of the fall in CBF seen in response to NOS inhibition, because changes were well within the limits of auto regulation (23). Furthermore, Whites group administered the systemic vasoconstrictor noradrenalin in doses that produced a blood pressure equivalent to that of L-NMMA. Noradrenalin did not reduce CBF to the same degree as L-NMMA indicating that NO inhibition has a specific effect on the cerebral circulation (8).
Nitric oxide in the regulation of the basal tone of the middle cerebral artery
Only few studies have dealt with simultaneous measurements of blood velocity in large cerebral arteries and measurements of cerebral blood flow after NOS-inhibition. This is the only reliable way to estimate diameter changes of the large arteries under the influence of drugs. We have previously studied the effect of four doses of L-NMMA ranging form 0.3–6 mg/kg on MCA blood velocity and found no effect of any of the doses (24). Including the present study, we have now recorded Vmean in MCA after infusion of 6 mg/kg L-NMMA/kg given over 15 min in 26 patients and we have never found a significant change in MCA velocity (24, 25).
We calculated the changes in the diameter of the middle cerebral artery from the measured values of rCBFMCA and Vmean (16, 17) and found no significant effect of L-NMMA. This strongly suggests that the basal tone of MCA is not under influence of NO.
To our knowledge, only one study has reported L-NMMA induced changes in MCA velocity. Schmetterers group found an 11% decrease in MCA velocity using 6 mg L-NMMA/kg for 5 min followed by 60 µg/kg min for 95 min (7), but in another study from the same group using a slightly smaller dose no such change was found (26). When using 5 mg/min L-NMMA for 5 min no changes in MCA velocity was reported (19). Finally an intravenous bolus of 10 mg/kg L-NMMA produced no changes of blood velocity in MCA measured with TCD but a simultaneous 15% reduction in mean ICA volume flow assessed by colour velocity ultrasonic imaging suggested a constriction of MCA (8). Based on those results and the fact that the NO-donor GTN dilates MCA without affecting CBF (17), it was proposed that MCA has a basal NO-tonus (8). However, since L-NMMA does not affect MCA velocity, it is more likely that the proposed MCA constriction represents a physiological adaptation to the reduction of CBF.
Data on the relationship between cardiac output and Vmean in MCA is limited. According to Adams et al. (27) TCD velocities should be relatively independent of small changes in cardiac output. In the present study a highly significant decrease in heart rate after L-NMMA was observed, which may have opposed a possible L-NMMA induced increase in MCA velocity and thereby could have caused an underestimation of MCA constriction.
The effect of NOS-inhibition on acetazolamide induced increases in MCA blood velocity and cerebral blood flow
From animal experiments there is substantial evidence that NOS inhibitors attenuate the increase in CBF elicited by hypercapnia although NO may not be the final mediator to produce vasodilatation (6). Inhalation of CO2 and infusions of acetazolamide have similar actions namely increase of cerebral blood flow but the molecular mechanisms are not completely the same. Acetazolamide is first line agent in the treatment of the headache in pseudotumour cerebri, working by a decrease in CSF production (28). Furthermore, acetazolamide has been suggested to be effective in the acute treatment of migraine aura status (29), but not in migraine prophylaxis (30). Acetazolamide is an inhibitor of carbonic anhydrase and decreases extra-cellular pH due to increase of H2CO3 and its dissociation products H+ and HCO3 – (31), whereas intracellular pH remains unchanged (32, 33). In contrast hypercapnia causes acidosis in both compartments (34).
The difference between hypercapnia and acetazolamide stimuli has been demonstrated in animals. In conscious rabbits cortical-cerebral blood flow was measured by hydrogen polarography. In the presence of the NOS-inhibitor L-NAME the hypercapnic induced increase in cortical cerebral blood flow was completely inhibited whereas the acetazolamide induced increase was only moderately reduced (35). In rats acetazolamide induced the same CBF-increase after pretreatment with the NOS-inhibitor NOLAG and with saline. By contrast, hypercapnia induced CBF- increase was markedly reduced after NOLAG pretreatment (36). NOS activity in brain tissue (cerebellum) may be markedly influenced by pH so that acid pH increases NO production (37). CO2 causes intracellular acidosis and NO might therefore be increased. This may explain that hypercapnic but not acetazolamide induced CBF increase in man is inhibited by NOS-inhibition.
In the present study the effect of iv infusion of L-NMMA on acetazolamide induced changes of both blood velocity in the middle cerebral artery and cerebral blood flow was studied with a combination of transcranial Doppler ultrasonography and SPECT. The CBF measurements were performed at time points where the largest response to acetazolamide was expected (38) and where CBF was expected to be under influence of L-NMMA (20). We found that NOS-inhibition had no effect of the acetazolamide-induced changes in MCA velocity or in cerebral blood flow. Together with previous animal studies the present results demonstrate that acetazolamide induced cerebral haemodynamic changes are not mediated by NO.
Our transcranial Doppler data showed that Vmean(MCA) 25 min after acetazolamide increased approximately 1.5 times more than rCBF in the territory of MCA. Fifty-five minutes after acetazolamide the increase of Vmean was 1.4 times higher than of rCBF (n = 8). Since Vmean increased more than the simultaneously measured rCBF our data suggest that the diameter of MCA is reduced after acetazolamide.
Conclusions
L-NMMA 6 mg/kg decreased resting rCBF significantly. Although L-NMMA decreased rCBF only to a minor degree, use of this nonspecific NOS inhibitor in migraine with aura where cerebral blood flow is reduced, may be problematic. Future specific NOS inhibitors, currently known to be under development, may not pose this problem.
Acetazolamide increased Vmean more than rCBF suggesting that it reduces the diameter of MCA. Using the velocity measurements to estimate the effect of acetazolamide on cerebral blood flow therefore results in an overestimation of blood flow. Our data also demonstrate that the action of acetazolamide on cerebral conduit arteries and arterioles in man is not NO-dependent.
Footnotes
Acknowledgements
We thank Mrs Bente Dall and Mrs Eva Broedsgaard for technical assistance. The Glaxo Wellcome Foundation supported the study financially.