Abstract
We have previously proposed that histamine causes migraine via increased NO production. To test this hypothesis, we here examined if the NOS inhibitor, L-NG methylarginine hydrochloride (L-NMMA:546C88), could block or attenuate histamine induced migraine attacks and responses of the middle cerebral, temporal and radial arteries. In a double blind crossover design 12 patients were randomized to receive pretreatment with L-NMMA (6 mg/kg) or placebo i.v. over 15 min followed on both study days by histamine (0.5 μg/kg/min) i.v. for 20 min. Headache scores, mean maximal blood velocity (Vmean) in the middle cerebral artery (MCA) (transcranial doppler) and diameters of temporal and radial arteries (high resolution ultrasound) were repeatedly measured. Pre-treatment with L-NMMA, had no effect on histamine induced headache or migraine, but also had no effect on the magnitude of histamine induced – decrease in MCA blood velocity, or dilatation of neither the temporal nor the radial artery. L-NMMA constricted the temporal artery by 8% before histamine infusion, whereas the radial artery was unaffected. The temporal artery dilated 4-5 times more than the radial artery during histamine infusion. In conclusion the use of a NOS inhibitor in the highest possible dose did not block the histamine-induced headache response or arterial dilatation. Either the concentration of L-NMMA reaching the smooth muscle cell was insufficient or, histamine dilates arteries and causes headache via NO independent mechanisms. Our results showed for the first time a craniospecificity for the vasodilating effect of histamine and for the arterial effects of NOS inhibition.
Introduction
Nitric oxide (NO) appears to play an important role in the initiation of migraine attacks (1). This is based on experimental studies in migraine sufferers using intravenous infusion of glyceryl trinitrate (GTN) (2–7). We have also shown that NOS inhibition is an effective therapeutic principle for spontaneous migraine attacks suggesting a continuous involvement of NO throughout the duration of migraine attacks (8). Histamine has an effect that is strikingly similar to the effect of GTN.
Both substances elicit an immediate headache during the infusion and a migraine attack that usually occurs one to several hours after the infusion. The time profiles of headache and arterial dilatation due to these two substances are so similar that a common mediator is likely (2–11). In vitro experiments have indicated that histamine may dilate primate cerebral arteries via an endothelial H1 receptor due to activation of endothelial nitric oxide synthase (eNOS) with subsequent formation of NO (12, 13). Histamine induced immediate headache, delayed migraine and arterial dilatation are elicited via the H1 receptor since these can be almost completely prevented by the H1-receptor–antagonist, mepyramine (10, 11). Thus, it seems likely that histamine may induce migraine indirectly via increased production of NO.
In order to clarify whether histamine induces headache via production of NO, we evaluated in the present study whether histamine induced headache and arterial dilatation can be blocked or significantly attenuated by pretreatment with the nitric oxide synthase (NOS) inhibitor, L-NG-methylarginine hydrochloride (L-NMMA: 546C88, GSK).
Materials and methods
Twelve patients suffering from migraine without aura according to the classification of the International Headache Society (IHS) (14) were included (11 females, 1 male; mean age 39.9 years, range 24–49 years; mean weight 65.3 kg, range 54–86 kg). The patients had between 6 migraine attacks per month and 6 migraine attacks per year (extremes included). Patients with more than 5 days of tension type headache per month, pregnant or breastfeeding women, patients with known drug abuse or with daily use of medication (including prophylactic migraine therapy but not contraceptives); patients with chronic diseases; hypo- or hypertension or psychiatric illness; patients with a history suggestive of ischaemic heart disease or with a supine diastolic blood pressure> 90 mmHg or a heart rate < 50 beats per minute at entry of study were not included. No medication, coffee, tea, alcohol or tobacco was allowed 12 h prior to the study (antihistamine was not allowed for 1 week, sumatriptan for 24 h and ergotamine for 48 h prior to the study).
The study was approved by the Ethics Committee of the County of Copenhagen (KA95131g) and was conducted according to the Helsinki II declaration of 1964, with amendments in 1996. All patients gave their informed consent and were informed that they were free to withdraw at any time during the study.
Design and methods
In a double blind crossover study, subjects attended the clinic twice at least one week apart. Patients were randomised to receive L-NMMA (546C88, GSK, UK) (6 mg/kg) or placebo (5% dextrose) infused intravenously over 15 min Thereafter all received 20 min i.v. infusion of histamine (0.5 µg/kg/min). Patients were randomly assigned by computer (Medical Stat®, version 2.12). Medical staff not involved in the study performed computer-randomization and preparation of study drugs. Four patients started with placebo and eight with L-NMMA. This quota was chosen by the computer and not known before the study. Headache intensity was scored on an 11-point numerical verbal rating scale from 0 to 10. 0: represented no headache, 1: pre-pain (a feeling of pressing or throbbing), 5: a headache of medium severity and 10 representing the worst possible headache. Other headache characteristics necessary for precise classification of the induced headaches according to the criteria of the International Headache Society (IHS criteria) were recorded, and at every recording it was evaluated whether the headache fulfilled the IHS criteria for migraine without aura. Time averaged mean of the maximal blood velocity (Vmean) in the middle cerebral artery was simultaneously measured in both left and right middle cerebral artery (MCA) with transcranial Doppler ultrasonography (2 MHz, Multidop X Doppler: DWL, Sipplingen, Germany) (15). A mean of four consecutive values of Vmean were taken (each representing the mean value of typically 4–5 heart beats automatically calculated by the computer). The luminal diameter of the superficial temporal artery at the usual headache side (if usual headache was bilateral the luminal diameter of the temporal artery was measured at the right side) and of the radial artery on the arm at the opposite side were measured with a high resolution ultrasound unit with A- and B- mode facilities (Dermascan CR, Cortex technology, Hadsund, Denmark). High frequency ultrasound (20 Mhz, band width 15 Mhz) was directed perpendicular to the skin over the artery, which was located by visible pulsations. The luminal diameter was derived from the high amplitudes reflecting the interfaces between blood and vessel wall (16). In order to reproduce the positions from day to day the angle and distance relative to the orbito-meatal line and to the distal volar crest of the wrist were recorded. Blood pressure and heart rate were measured with an automatic inflatable arm cuff (Omega 1400, Invivo Research Laboratory, New York, USA) and end-tidal pCO2 was measured throughout the period using a capnograph (Poet, Criticare systems) connected to an anaesthetic mask.
Procedure
The subjects arrived at the laboratory at 8.30 a.m. A cannula (Viggo Venflon® 1.4 mm) was placed in the right cubital vein for L-NMMA/placebo and histamine infusions. Baseline values of headache intensity and characteristics, mean blood velocity (Vmean) in both MCAs, the luminal diameter of the superficial temporal and of the radial artery, blood pressure, heart rate and pCO2 were recorded after 30 min of rest in supine position. Then L-NMMA or placebo was infused intravenously for 15 min by a volumetric pump (Braun® Perfuser). Two minutes after termination of the L-NMMA/placebo infusion, histamine was infused for 20 min using the same pump. Headache intensity and characteristics, Vmean, diameters of the superficial temporal and radial arteries, blood pressure, heart rate and end tidal pCO2 were recorded before and every five minutes during and after histamine infusion until 80 min after start of the histamine infusion. At every recording the presence of facial flushing was also noted. After discharge the patients recorded headache intensity and characteristics and medication use once every hour for the following 11 h. Treatment was offered if needed although sumatriptan and ergotamine were withheld for at least 6 h after L-NMMA infusion.
Methods for in vitro studies of human tissue
For studies of vasomotor responses, human arteries from the cerebral cortical surface (pial artery) were obtained at autopsy within 6–24 h postmortem. Specimens were collected in accordance with Danish legislation and approved by The Danish Ethical Committee, Copenhagen (KA95213m).
Immediately after removal, the vessel segments were placed in a buffer solution containing : 119mM NaCl, 15mM NaHCO3, 4.6mM KCl, 1.5mM CaCl2, 1.2mM NaH2PO4, 1.2mM MgCl2, 11mM glucose. Circular vessel segments with an inner diameter of 0.3–0.6 mm (cerebral arteries) and a length of 2–4 mm were mounted in two Multi Myographs, each for parallel experiments of up to 4 vessels in separate tissue baths (Model 610 M, JP Trading, Denmark). The vessel segments were suspended between two L-shaped metal holders (0.15 mm in diameter). The distance between the holders could be varied by a micropositioner coupled to one of the holders, thereby allowing adjustment of the resting tension of the segments. The other holder was connected to a transducer for registration of alterations in vascular tone. The Multi Myograph was connected to a Pentium computer with Myodaq for Windows software (Myonic Software, Denmark). The arterial segments were given a tension of 2–4 mN depending on the vessel size (usually 3 mN) and were allowed to accommodate for 1–1.5 h until the tension had stabilized at the desired level. In the tissue baths the arteries were immersed in a temperature-controlled (37°C) buffer solution that continuously was bubbled with a mixture of 95% O2 and 5% CO2 giving a pH of about 7.4. The presence of intact endothelium was assessed by a good relaxant response to acetylcholine (ACh). Vessel reactivity was tested by exposure to a buffer solution containing 125 mM KCl, obtained by an equimolar substitution of NaCl for KCl. Only vessels with a reproducible potassium-induced contraction after washout with the sodium buffer solution were used for further investigation. The potassium-induced contraction amounted to 23.8 ± 5.9 mN (n = 5, from 2 patients). For the study of relaxant responses, the vessel segments were precontracted by the addition of prostaglandin F 2a (PGF2a) at a concentration of 3 × 10−6 M. At the time of precontraction where the relaxant agent first was added to the tissue baths (the stable level of tension) the precontraction amounted to 19.6 ± 3.9 mN (n = 10). Concentration-response data were obtained by cumulative addition of histamine to the tissue bath. L-NMMA was added to the tissue baths 20 min before the responses to histamine agonists were tested. The values for relaxation are expressed as percentage of the level of precontraction due to PGF2a. The responses were characterized in terms of Imax (maximum relaxant effect obtained with an agonist), IC50 (the concentration eliciting half maximum relaxant effect) and pIC50 (negative logarithm of IC50) values. Values are given as mean ± SEM with median values within brackets. Number of experiments = n.
Drugs used in in vitro studies: Histamine (Sigma®, USA), Prostaglandin F 2α (Dinoprost®, Upjohn, USA), L-NMMA (NG-Monomethyl-L-Arginine Acetate (RBI/Sigma®, Natch, MA, USA). A stock solution was prepared by dissolving the drugs in distilled water. All drugs were just before the experiment further diluted in buffer solution. The concentrations are expressed as the final molar concentration in the tissue bath.
Statistics
Headache scores are given as median and range. The difference between the number of patients who experienced headache in the first 60 min (immediate headache) and in the period from 60 min to 12 h after start of histamine infusion (delayed headache) and throughout the whole period, when histamine was preceded by administration of L-NMMA or placebo was tested with Fishers exact test (Med. Stat®). Differences in maximal headache score and total headache experienced (the summed headache score) in the first 60 min and from 60 min to 12 h after start of histamine infusion were analysed with Wilcoxon's rank test for paired data (Statgraphics® 7.0). Friedman's test was used to test for time differences in headache intensity within group (Statgraphics® 7.0) and changes were located with Dunns test.
Hemodynamic responses were calculated as percentage of baseline, and are given as mean and standard error of the mean (± SEM).
For each subject data were reduced to values at baseline, last measurement during the L-NMMA/placebo infusion, 30 min(last measurement during histamine infusion), 55 and 95 min from start of the L-NMMA/placebo infusion(40 and 80 min after start of histamine infusion). The overall differences in Vmean, luminal diameters, blood pressure and heart rate between L-NMMA and placebo pretreatment were compared with MANOVA and differences were then located with a multiple range test (Confidence intervals, Statgraphics® 7.0). Differences over time within the group were analysed with analysis of variance (MANOVA, Statgraphics® 7.0) before data were reduced to the five time points, and changes were then located with a multiple range test (Confidence intervals, Statgraphics® 7.0).
Differences between the effect of L-NMMA and placebo on basal mean blood velocity, arterial diameters, blood pressure, heart rate, and pCO2 (in percentage of baseline) before histamine infusion were tested by a paired t-test (baseline values – last measurement during L-NMMA/placebo infusion (15 min)). This test was also used to determine if there were any differences between the effects of L-NMMA as compared with placebo on histamine induced maximal response (15 min – last measurement during histamine infusion).
Statistical evaluation of in vitro studies of vasomotor responses was performed by Student's unpaired t-test. In all tests P < 0.05 was considered statistically significant.
Results
Headache
No subjects reported headache at baseline or immediately after infusion of L-NMMA or placebo.
Immediate headache was defined as headache occurring during the histamine infusion or in the subsequent 40 min (i.e. a period of 60 min). In this time period 11 of 12 patients pretreated with placebo experienced headache, which in two patients fulfilled the IHS criteria for migraine without aura. In comparison nine of 12 patients pretreated with L-NMMA experienced headache during this period, and four of these fulfilled the IHS criteria for migraine without aura (P > 0.05, Fishers exact test, Table 1). The histamine-induced headache changed significantly over time both after placebo and L-NMMA pretreatment (P < 0.000005, Friedman) and (P < 0.0005, Friedman, Fig. 1). L-NMMA pretreatment did not influence maximal headache score (median 2.5, range 0–8, after L-NMMA vs. median 2, range 0–8, after placebo; P = 0.80, Wilcoxon) or the sum of headache scores in this period(229 after L-NMMA vs. 222 after placebo; P = 0.80, Wilcoxon).

Headache during histamine infusion after pretreatment with L-NMMA (a) and placebo (b). Individual responses are shown and the bold line represent mean of 12 subjects. L-NMMA pretreatment did not affect histamine induced immediate headache.
This table gives the number of patients who experienced headache and migraine (IHS) in the first 60 min (immediate headache) and in the period from 60 min to 12 h after start of histamine infusion (delayed headache) and throughout the whole period, when histamine was preceded by administration of L-NMMA or placebo

Fisher's exact test.
Delayed headache was defined as headache in the period from 1 to 12 h after start of histamine infusion. Six of 12 patients pretreated with placebo continued to have or developed headache. In five of these patients the headaches fulfilled the IHS criteria for migraine without aura. Five of 12 subjects pretreated with L-NMMA continued to have or developed headache, and in three of these the headaches fulfilled the IHS criteria for migraine without aura (P > 0.05, Fishers exact test, Table 1). In these patients maximal headache score was median 4 (range 2–7) after L-NMMA pretreatment and median 5 (range 2–7) after placebo pretreatment (P = 0.59, Wilcoxon). Five of six patients in the placebo treated group and four of five patients in the L-NMMA pretreated group received headache medication at a medium of 4 and 3 h after start of infusion, respectively. All the patients who took analgesic medication were patients who developed migraine. On this basis the following results regarding delayed headache have to be interpreted with caution.
The maximal intensity of the delayed headache was reached at a median of 2.5 h (range 1–8 h) after start of the histamine infusion in the placebo group and at a median of 5 h (range 1–10 h) in the L-NMMA treated group (P = 0.50, Mann–Whitney).
The total headache experienced (summed headache score) in this time period was 133 in L-NMMA pretreated patients and 112 in placebo pretreated patients
(P = 0.72, Wilcoxon).
Blood velocity changes in the middle cerebral artery
The mean blood velocity (Vmean) in MCA was simultaneously measured on both right and left sides. There were no differences in Vmean between the two sides, neither when patients were pretreated with L-NMMA nor with placebo (L-NMMA P= 0.43; placebo P= 0.21, MANOVA). However, measurements were only taken for 60 min after histamine infusion and do unfortunately not reflect the migraine period adequately. In the following results it was chosen to use the measurement from the right side of the head in the calculations.
L-NMMA pretreatment did not affect Vmean in MCA; 0.2 ± 3.5% from baseline after L-NMMA, and 0.2 ± 2.7% from baseline after placebo (P = 0.99, paired t-test). Nor was the magnitude of the histamine-induced decrease in Vmean affected (from 0.2 ± 3.5% to − 20.2 ± 2.7% of baseline after L-NMMA pretreatment and from 0.2 ± 2.7% to − 26.0 ± 2.3% after placebo pretreatment; P= 0.26, paired t-test (Fig. 2). A slight but significant attenuation of the histamine induced decrease in Vmean was found in the period 55–95 min in the L-NMMA pretreated group (MANOVA, P= 0.03, Fig. 2).

Mean (SEM) changes in Vmean after L-NMMA (•) and placebo (○) pretreatment. L-NMMA neither affected baseline mean blood velocity (Vmean) in MCA (P = 0.99), nor the magnitude of the histamine induced decrease in Vmean (P = 0.26). An overall MANOVA detected a significant attenuation of the histamine-induced response in the period 55–95 min in L-NMMA pretreated patients (P = 0.03).
pCO2 decreased significantly over time in both groups (placebo, P < 0.0001; L-NMMA, P < 0.0005, MANOVA), but there was no difference between the L-NMMA and the placebo pretreated group (P = 0.47, MANOVA). For this reason data were not pCO2 corrected.
Changes in luminal diameters of the radial and temporal arteries
Temporal artery diameter measurements were only completed for 10 patients. There was a significant reduction in the luminal diameter of the temporal artery before histamine infusion in the L-NMMA pretreated group compared to the placebo pre treated group (to − 8.5 ± 2.8% after L-NMMA and to +11.0 ± 4.2% after placebo; P < 0.005, paired t-test). L-NMMA had no significant effect on the magnitude of the histamine induced dilatation (from − 8.5 ± 2.8% to +36.6 ± 5.5% after L-NMMA pretreatment, and from +11.0 ± 4.2% to +58.7 ± 7.4% after placebo pretreatment; P= 0.79, paired t-test), Fig. 3.
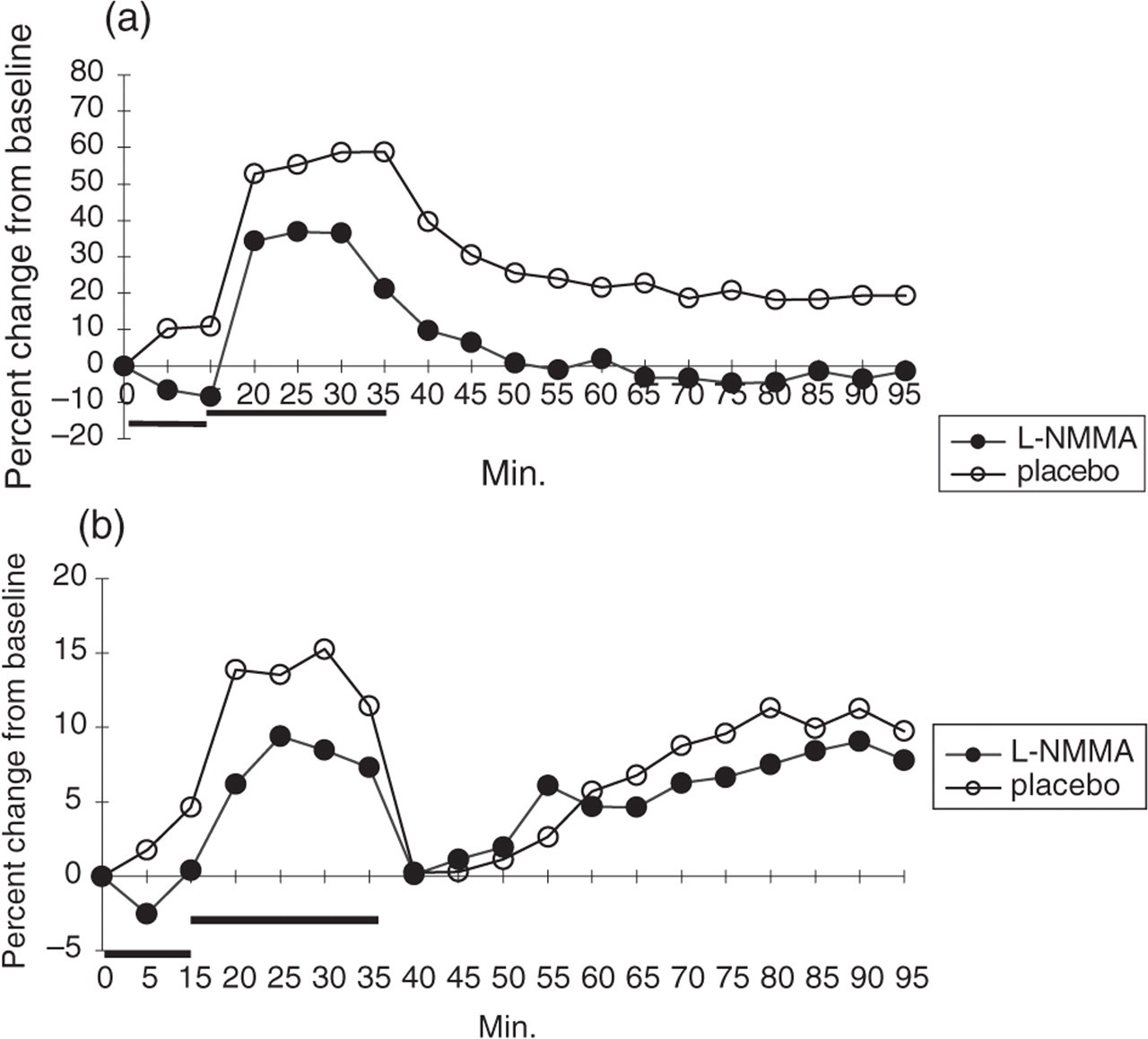
Mean (SEM) changes in (a) the diameter of the temporal artery and (b) the radial arteries after L-NMMA (•) and placebo (○) pretreatment. L-NMMA significantly constricted the diameter of the temporal artery to − 8.5 ± 2.8% of baseline (P = 0.002, paired t-test), but had only a tendency to affect the baseline diameter of the radial artery (P = 0.056), in contrary L-NMMA had no effect on the magnitude of the histamine induced dilatation of the temporal artery (P = 0.79) or of the radial artery (P = 0.58).
In the radial artery there was a tendency to a small attenuating effect of L-NMMA on baseline diameter before histamine infusion (to +0.4 ± 1.9% after L-NMMA pretreatment and to +4.6 ± 1.3% after placebo pretreatment; P= 0.056, paired t-test), but there was no effect of pretreatment with L-NMMA on the magnitude of the histamine induced dilatation of the artery (+0.4 ± 1.9% to +8.5 ± 2.7% after L-NMMA pretreatment vs. +4.6 ± 1.3% to +15.3 ± 3.5% after placebo pretreatment, P= 0.58, paired t-test, Fig. 3).
In general the temporal artery was more sensitive to L-NMMA treatment before histamine infusion than the radial artery (P < 0.05, paired t-test). Furthermore, the temporal artery was 4–5 times more sensitive to histamine than the radial artery from +11.0 ± 4.2% to +58.7 ± 7.4% in the temporal artery; from +4.6 ± 1.3% to +15.3 ± 3.5% in the radial artery at the last measurement during histamine infusion (time 15–20 min after its start) (P < 0.02, paired t-test), Fig. 3.
For changes in blood pressure and heart rate during L-NMMA and histamine infusion please see Table 2. The appearance of flush was not affected by the L-NMMA pretreatment.
Systolic- and diastolic blood pressure and heart rate during L-NMMA placebo infusion (0–15 min) and histamine infusion (15–35 min)

Values are given as mean ± SEM. L-NMMA pretreatment significantly increased systolic blood pressure (P < 0.001) and the diastolic blood pressure (P < 0.001), before histamine infusion, but had no effect on the magnitude of the histamine-induced decrease in diastolic blood pressure (P = 0.36458). Surprisingly, the magnitude of systolic blood pressure decrease during histamine infusion was more pronounced in L-NMMA pretreated patients than in placebo pretreated patients (P < 0.03). L-NMMA significantly decreased heart rate before histamine infusion (P < 0.0001), but did not affect the magnitude of the histamine-induced response (P = 0.84).
In one healthy subject blood samples from the cubital vein were drawn for determination of the plasma concentration of histamine before and at the end of 20 min of histamine infusion (0.5 µg/kg/min). This was found to be 2.4 nM prior to and 50 nM at the end of the histamine infusion (Immunotech Histamine assay).
Results of in vitro studies
Cumulative application of histamine to the tissue baths induced concentration-dependent relaxations of circular segments of human pial arteries precontracted by PGF2a. Pre-treatment of the vessel segments with 1 µM L-NMMA did not cause any significant blockade of the histamine induced relaxation. The respective pIC50 values and Emax values were in controls 7.69 ± 0.26 and 94 ± 4% (n = 7, p> 0.05) and in presence of 1 µM L-NMMA 7.53 ± 0.26 and 89 ± 5% (n = 7, p > 0.05, Fig. 4a). However, in the presence of 10 µM L-NMMA we found significant blockade of the histamine induced relaxation. The pIC50-value decreased from 7.58 ± 0.33 in controls to 6.56 ± 0.26 (P < 0.05) in presence of 10 µM L-NMMA and Emax decreased nonsignificantly from 111 ± 11% (n = 5) to 64 ± 18% (n = 5, P > 0.05). Significant differences in responses to histamine alone, as compared to histamine responses in the presence of L-NMMA 10 µM was observed at concentrations of 10−7 M to 10−6 M (Fig. 4b).

Relaxant responses in human cerebral arteries precontracted by 3 × 10−6 M prostaglandin F 2α to the cumulative application of histamine with or without L-NMMA in a concentration of A: 1 µM and B: 10 µM. Values given represent mean ± S.E.M., n= 5–7. Statistical evaluation was performed by Student's unpaired t-test, P < 0.05.
Discussion
Histamine induced headache and its mechanisms
In a previous study in migraineurs histamine was shown to cause an immediate headache during infusion and a delayed migraine headache with peak headache intensity approximately five hours after start of the histamine infusion. The intensity of the immediate headache was significantly attenuated and the delayed migraine completely abolished when patients were pretreated with the H1-receptor antagonist, mepyramine (10). During the histamine infusion blood velocity in MCA measured with Transcranial Doppler decreased by 26% (11) indicating a dilatation of this artery since the regional cerebral blood flow (rCBF) is unaffected by histamine (17). This dilatation was largely but not completely abolished by pretreatment with mepyramine (11). Histamine does not cross the blood brain barrier (18) and therefore, when administered systemically it works exclusively on endothelial receptors. The endothelium of human cerebral arteries contains dilatory H1-receptors that when activated cause formation of nitric oxide (NO) an effect that in vitro can be blocked by H1-receptor antagonists and inhibited by NOS inhibitors (12, 13). It is thus plausible, that histamine induced headache migraine and arterial dilatation might be triggered by NO.
Several studies have also pointed towards NO as the mediator of glyceryl trinitrate (GTN)-induced headache, migraine and arterial dilatation, which have an analogous time profile to the histamine induced responses (2–4, 6, 7). Theoretically, GTN could induce headache/arterial dilatation via liberation of histamine from perivascular mast cells and basophils (19), but pretreatment with mepyramine before GTN infusion does not to affect GTN induced headache or arterial responses (5, 20). From the above-mentioned studies it has been concluded that the most likely common trigger of histamine and GTN induced immediate headache, delayed migraine and arterial dilatation is NO (1).
NO seems to be involved not only in the initiation of migraine attacks but throughout the course of spontaneous migraine attacks, because L-NG-methylarginine hydrochloride (L-NMMA:546C88), the same NOS inhibitor as used in the present study, was effective as a treatment of spontaneous acute migraine attacks in 67% of the treated patients (8).
The hypothesis of the present study was therefore, that pretreatment with the NOS inhibitor L-NMMA before histamine infusion would attenuate or abolish histamine induced headache and arterial responses. This hypothesis was effectively rejected because there was no effect and not even a trend towards an effect of L-NMMA pretreatment on either histamine induced immediate headache, delayed migraine, or the magnitude of the arterial dilatation caused by histamine. There are several possible reasons for these results:
It is most reasonable to accept the results and conclude that histamine dilates cerebral arteries and triggers migraine via molecular mechanisms other than NO; despite the fact that several in vitro studies have demonstrated a relationship between H1-receptor activation and NOS activation (12, 13, 21, 22). At least two of these studies have concerned human cerebral arteries (12, 13). The possibility of a false negative study or Type II error might also be considered.
It has been proposed that analogues of L-arginine such as L-NMMA demonstrate tissue – and agonist- dependence in terms of their ability to inhibit receptor mediated events involving the liberation of NO (23). An in vitro study using rat aorta and acetylcholine as the agonist proposed that NG-monomethyl-L-arginine selectively inhibits basal but not agonist stimulated activity of NOS (24). However, this is not in agreement with other studies (25, 26).
The dose of L-NMMA(6 mg/kg over 15 min) could perhaps have been too small to compete with the subsequent dose of histamine(0.5 µg/kg/min for 20 min). The plasma concentration of histamine after 20 min of histamine infusion was approximately 50 nM. From a previous study (yet unpublished) we have estimated the plasma concentration of L-NMMA at this time point after 6 mg/kg i.v. over 15 min to be 8.22 µg/ml. This plasma concentration equals a molar concentration of L-NMMA of 33 µM. Our in vitro studies performed on isolated human cerebral arteries showed that 10 µM, but not 1 µM, of L-NMMA was enough to induce a significant shift of the histamine concentration–response curve. Thus, the given dose of L-NMMA should be high enough to show an effect also in vivo. On the other hand, L-NMMA acts inside endothelial cells of cerebral arteries (i.e. behind the blood brain barrier (BBB)) while a possible histamine action on endothelial H1 receptors would occur at the endothelial surface, i.e. outside of the BBB. The failure to block the histamine induced headache and intracranial dilatation could therefore be due to insufficient penetration into endothelial cells of L-NMMA. L-NMMA is transported via the y + aminoacid transport system and one fifth of the plasma concentration(6.6 µM) of L-arginine is estimated to cross the BBB in the capillaries (ref. GSK, UK). Its penetration into cerebral arteries is unknown (18, 27). However, the efficacy of L-NMMA in spontaneous migraine attacks (8) suggests that it does get into arteries and periarterial space.
In a study with a similar design as in the present study, healthy volunteers instead of migraine patients were given L-NMMA (5 mg/kg for 5 min followed by 50 µg/kg/min for 40 min) or placebo before and during histamine infusion (0.16 µg/kg/min for 15 min) (28). The headaches were not affected by L-NMMA pretreatment. Histamine induced flush was also not inhibited by L-NMMA pretreatment in accordance with the present results.
Other effects of L-NMMA and histamine
We found that histamine alone induced a 26% decrease in MCA blood velocity, which is in accordance with results previously found in our laboratory (11) but in contrast to the study of Schmetterer et al. (28). In that study no histamine induced decrease in Vmean was found, probably due to the use of a small dose of histamine, but the use of healthy volunteers and other differences in methodology can also be important. Surprisingly they did not detect the expected blood pressure increase (28) typically induced by L-NMMA (29, 30).
Vmean in MCA was unaffected by L-NMMA. In a recent SPECT-study of the regional cerebral blood flow (rCBF) during L-NMMA infusion (same dose as in the present study) the cerebral arterioles were significantly affected although only to a minor degree (6–7% decrease in blood flow in the MCA territory) (in preparation). Since rCBF = blood velocity × (cross-sectional area of the feeding artery) (31), the estimated constriction of MCA is 1.3% (P = 0.44). Therefore, it can be concluded that NO exerts no tonic effect on the diameter of the MCA.
The present study shows for the first time that L-NMMA constricts the superficial temporal artery and that there is only a nonsignificant tendency to constrict the radial artery. The temporal artery and perhaps the radial artery therefore have a basal production of NO. The same is true of the peripheral arterioles (the diastolic blood pressure increased 16%). The superficial temporal artery is more sensitive to NOS-inhibition, than the radial artery and it is also more sensitive to the dilating effect of histamine. Arterial craniospecificity has previously been shown using the exogenous NO-donor, GTN (32). It seems likely that the induced headache and the total absence of any other pain induction after these substances may causally relate to this cranial arterial hypersensitivity.
We conclude that, at the dose given, the NOS inhibitor, L-NMMA, did not affect histamine induced headache, migraine or arterial responses. Although we used the highest possible dose L-NMMA for the human experiments, it may have been insufficient to achieve the expected effect. Alternatively, histamine may induce migraine via other mechanisms. Furthermore, an interesting arterial craniospecificity for histamine and NOS inhibition was found in cranial arteries.
Footnotes
Acknowledgements
L-NGmethylarginine hydrochloride (L-NMMA:546C88) was kindly provided by Glaxo Wellcome. The skilful assistance of technician MrsKirsten Enghave and Mrs Bente Leisner is gratefully acknowledged. Furthermore, the assistance of Dr Per Stahl Skov, the Reference Laboratory, for determination of histamine in plasma samples and of Dr Misba Beerahee, Glaxo-Wellcome, for estimation of plasma concentration of L-NMMA 546C88 is gratefully acknowledged. The study was supported financially by the Cool Sorption Foundation and Glaxo-Wellcome.
