Abstract
Pathological changes on diffusion-weighted MR scans had been described in hemiplegic migraine and perfusion changes had been demonstrated in typical migraine aura with radio-isotope studies and, more recently, MR perfusion imaging. However, there is relatively little knowledge of the pathophysiology of long-lasting migraine aura and its possibly variant phenotype, visual snow. Our aim was to investigate with advanced MR techniques whether patients with long-lasting visual disturbance showed regional alterations in cerebral water diffusion and perfusion. We have studied four patients using MR perfusion and MR diffusion imaging. Two patients had typical visual aura and two had a primary persistent visual disturbance (visual snow phenomenon). All patients had normal conventional structural MR imaging. MR diffusion-weighted images were acquired with a b-value of up to 1000 s/mm2. From the diffusion weighted images we generated maps of apparent diffusion coefficient (ADC), which were inspected visually and used for ADC measurements of predefined regions of interest, which included the visual, frontal, insular and temporal cortices. MR perfusion imaging was performed using a bolus tracking technique with dynamic susceptibility-weighted images. Colour coded maps of relative cerebral blood volume, mean transit time and bolus arrival time were generated, as well as time-signal intensity curves over the anterior, middle and posterior cerebral artery territories. The maps of the ADC and above perfusion parameters appeared symmetrical in all patients with no evidence of decreased water diffusion or cerebral perfusion in the occipital regions, or elsewhere. There was no statistically significant difference between the ADC measurements of the primary visual cortices and other cortical regions. Our findings suggest that regional changes in cerebral water diffusion and perfusion do not play an important part in the pathophysiology of persistent migraine aura or primary persistent visual disturbance.
Keywords
Introduction
Migraine aura consists of a usually transient clinical disturbance that a can be attributed to brain dysfunction (1). The first edition of the International Headache Society Classification defined typical migraine aura as lasting up to 60 min, and prolonged aura up to 7 days (2). However, it has been recognized for some time that aura symptoms in migraine sufferers may last longer than 7 days (3), and the second edition of the classification defines persistent migraine aura as at least one symptom lasting for more than 7 days (4). It has been suggested that the cortical spreading depression (CSD) of Leao (5, 6) is likely to be an experimental model for migraine aura (7). Patients with persistent aura symptoms present an opportunity to examine the migrainous brain using physiological neuroimaging methods to look for changes that might provide clues as to the nature of the biology of migraine aura.
Various forms of migraine aura that last longer than one hour have been described. The second edition of the International Classification of Headache Disorders abandoned the term prolonged aura, suggesting patients be classified as having migraine with aura or Probable Migraine with Aura (4). This leaves aura experienced between one hour and 7 days in a curious position of being described as Probable, although if the aura includes weakness and lasts 24 h or less it may qualify for inclusion in sporadic hemiplegic migraine (8), assuming there is no family history of similar attacks. Patients with migraine with persistent aura have been described where the disturbance, such as a scintillating scotoma persists for months (9). Such a case would seem to easily fulfil the new IHS criteria. Prolonged migraine aura status, in which patients had repeated but discrete attacks of aura over hours or weeks (3), has been described. This might be more difficult to accommodate in the new classification. Another group of visual symptoms labelled persistent positive visual phenomena (10) have been reported. These are not at all typical of migraine aura, and may represent a very distinct entity.
Brain imaging in spontaneous and triggered aura has demonstrated a wave of oligemia that moves from the posterior to anterior brain areas (11). Oligemic changes have been seen with positron emission tomography (PET) (12), albeit in one case, and perfusion-weighted MRI (13), and are certainly not associated with hypoxia in the occipital cortex (14). A preceding hyperaemic phase, so characteristic of spreading depression in animals (7), has now been observed during exercise-induced aura (15). A severe form of prolonged aura with motor symptoms is familial hemiplegic migraine (FHM) (16). One case has been reported of FHM with a mutation in the Cav2.1 subunit of the P/Q type (17) voltage-gated calcium channel in which the prolonged aura was associated with decreased hemispheric water movement detected by diffusion-weighted MRI (DWI) (18). Given recent elegant work in the gyrencephalic cortex of the cat showing DWI changes spreading across the brain with the initiation of CSD (19), it could be asked to what extent changes in DWI signals can be observed in various types of migraine aura. Persistent migraine aura offers a stable, albeit relatively rare patient cohort with which to study the pathophysiology of migraine aura. We have studied patients with two patients with more typical persistent visual aura (4) and two others with what we shall term Primary Persistent Visual Disturbance (PPVD), what has in the past been called visual snow.
Methods
Approval from the local Research Ethics committee had been obtained and all patients had signed informed consent. For the clinical definitions we employed the second edition of the International Headache Society (4). Persistent aura without infarction (1.3.5) was diagnosed where the visual disturbance was typical of aura, flickering lights, spots or lines, or a scotoma, typical involving part but not the entire field. Primary Persistent Visual Disturbance was assigned where the primary element of the being a widespread visual disturbance that was not formed and typically grainy or snow-like in pattern.
Data acquisition
MR scanning was performed on a 1.5 T system and included axial T2-weighted fast spin-echo, coronal fluid-attenuated inversion recovery (FLAIR) and diffusion and perfusion-weighted images. Diffusion-weighted (DW) images were acquired using a single shot spin-echo EPI sequence with a TR of 1200 ms, TE of 94 ms, 24 cm field of view, 96 × 128 matrix, and a slice thickness of 5 mm with a 1.5 mm gap between sections. DWI was performed with three different b-values of 500 s/mm2, 750 s/mm2 and 1000 s/mm2.
Perfusion-weighted images were acquired during the i.v. injection of a 15 ml gadolinium bolus (meglumine gadoterate, Dotarem ®) at a rate of 5 ml/s using a single shot gradient echo EPI sequence with a TR of 1200 ms, TE of 40 ms, flip angle of 20°, 26 cm field of view, 96 × 128 matrix, and a slice thickness of 5 mm with a 3 mm gap between sections.
Data analysis
Data analysis was performed on an Advantage Workstation (GE Medical Systems, Milwaukee, USA). The diffusion-weighted and perfusion-weighted images were postprocessed using a proprietary image analysis software package (FuncTool 1.9 m, GE Medical Systems, Milwaukee, USA).
Diffusion-weighted imaging
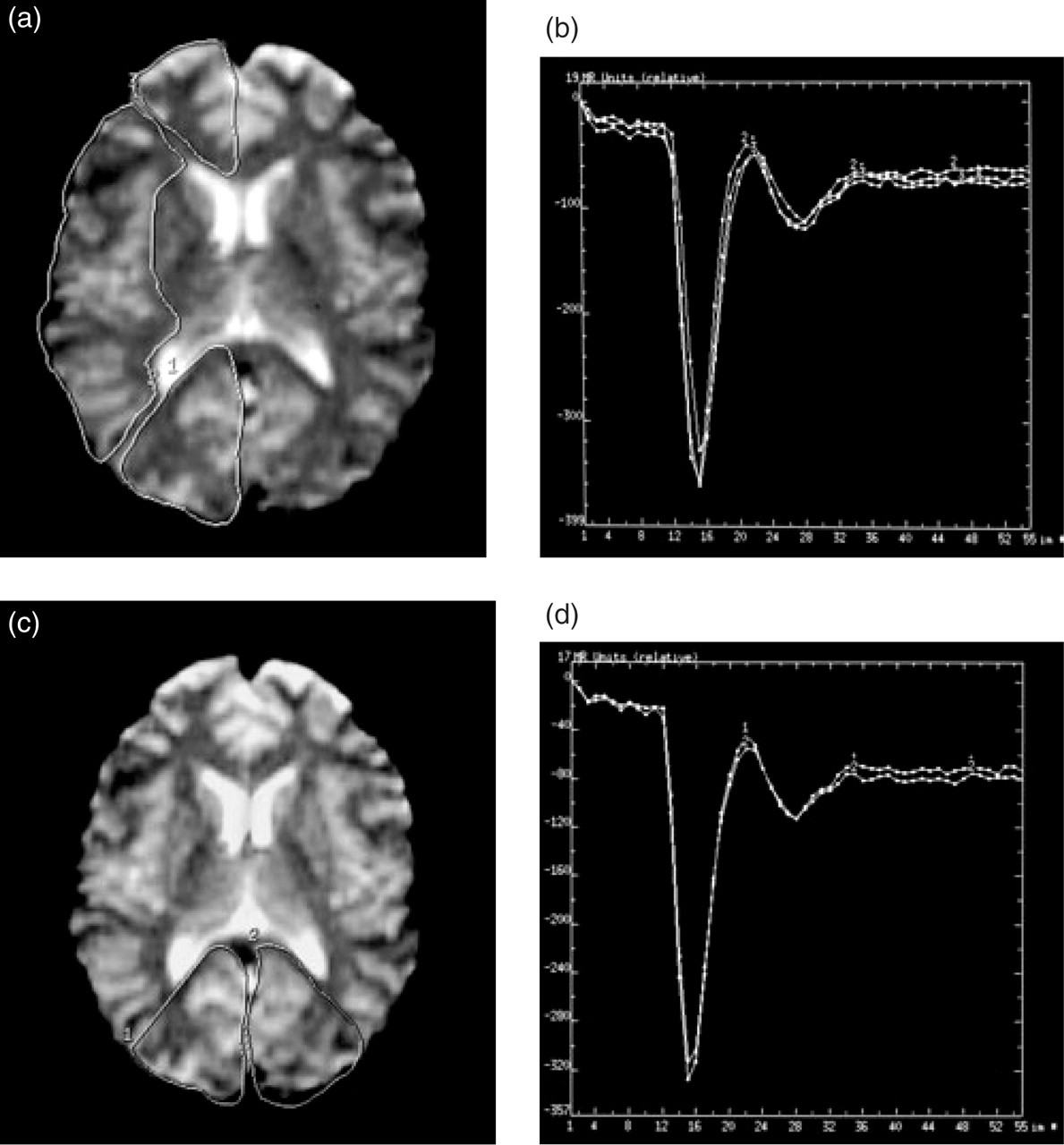
Maps of apparent diffusion coefficient (ADC) were produced from the isotropic DW images. Both the DW trace images and ADC maps were visually inspected for regional differences and abnormal areas. In addition ADC measurements were obtained from selected regions of interest which included the visual cortex, frontal cortex, temporal cortex and insular cortex of each cerebral hemisphere. Standard MRI templates (20) were used to identify the visual cortex on the axial DW images. The regions of interest were manually outlined by a single experienced operator (HRJ), carefully avoiding any inclusion of CSF in the adjacent sulci (Fig. 1). In addition to absolute ADC measurements relative ADC values of these regions were recorded, expressed as a percentage of the mean ADC value of all regions. Statistical analysis of differences between the ADC of the left and right visual cortex and the other cortical regions was performed using a two way analysis of variance (

ADC map at the level of the lateral ventricles showing symmetrical appearances with no areas of decreased or increased water diffusion. Regions of interest (ROI) for ADC measurements have been drawn around the visual, insular and frontal cortices of both hemispheres. The temporal cortices have been outlined on a different slice.
Perfusion-weighted imaging
Colour coded maps of relative cerebral blood volume (rCBV), bolus arrival time (BAT), and mean transit time (MTT) were generated from the perfusion-weighted images, using a region of interest over one of the middle cerebral arteries as arterial input function. On the rCBV maps specific window and threshold settings were used to exclude large vessels over the cortical surface and to produce separate images of grey matter and white matter perfusion. The latter was achieved by producing sets of images with narrow window settings within two standard deviations (SD) of the mean blood volume of white matter and grey matter, respectively. In addition time-signal intensity curves were produced from regions of interest corresponding to the territories of the anterior, middle and posterior cerebral arteries.
Results
Cases
Table 1 summarizes the history and clinical features of the cohort. Two patients, cases 1 and 2 we have classified as Primary Persistent Visual Disturbance, and the two patients, cases 3 and 4, as Persistent Migraine Aura.
Summary of history and clinical features

Case 1
A 39-year-old female developed intermittent migraine headaches at the age of 7 years. At the age of 11 years she developed persistent flashing lights that have never left her. These she describes as thousands of small yellow, white or silvery dots over the whole of both visual fields. They can be seen even with the eyes closed. The vision can be blurred in the left hemifield on waking. Episodically, bigger flashing lights appear and gradually disappear. In addition to these persistent symptoms, she has weekly throbbing headaches, worse on the left side associated with vomiting, photophobia and phonophobia and vertigo lasting for 24–72 h. These headaches are sometimes preceded by zigzag visual disturbance for about an hour that displace the flashing lights as they move across the field of vision. She has a past history of depression, currently controlled with Paroxetine, and of esotropia corrected at the age of 4 years. The visual symptoms have proved refractory to acetazolamide, amitriptyline, pizotifen and propranolol and acute treatment with intranasal ketamine. Examination including fundoscopy, Humphrey visual fields and neuro-otological assessment was normal except for acuity reduced to 6/12 in the right eye, probably because of the childhood strabismus. She has a normal magnetic resonance imaging (MRI) brain scan, electroencephalogram (EEG), electroretinogram (ERG) and visual evoked potentials (VEPs).
Case 2
A 46-year-old female with a 20-year history of migraine. These attacks typically started with 10–15 min of flashing lights in both hemifields, before the onset of migraine headache. Symptoms lingered for two months with a nondisabling feeling of a muzzy head, flashing lights and a zigzag pattern in her vision. Episodes occurred 4–5 times per year. At the age of 42 she experienced an episode with a severe headache, incoordination, difficulty with speech and writing. An outside MRI scan, which was not available for review was reported to show small areas of signal change in the left Sylvian fissure and left occipital lobe. She was found to have bacterial endocarditis that was treated successfully. However, since that time she has had persistent headache and visual symptoms, the latter described as blotches in her central vision, coloured at times, more visible with her eyes closed. The visual symptoms fluctuate, being worse for one week out of every two. These symptoms have been refractory to treatment with pizotifen, sodium valproate, flunarizine, topiramate, and dothiepin. Examination is entirely normal, including Humphrey visual field tests, fundoscopy and visual acuity. A repeat MRI 3 years after the acute event was normal.
Case 3
A 27-year-old female has experienced migraine since the age of 13 year, occurring once every 3 months, usually preceded by 30 min of flashing lights in both hemifields. The frequency of these increased at the age of 22–5 times per month. Since a particularly bad migraine attack at the age of 23 she has had persistent shimmering over the entire visual field bilaterally. This is described as similar to looking at a road on a hot hazy day. The effect is strongest in the lower half of the field of vision and around the peripheries. In addition to this she has periodically tiny pinpoints of bright light that last for 30 s. She still experiences a background, featureless headache for one week per month and migraine once every few months sometimes with a 20-minute exacerbation of the visual aura during which she describes herself as almost blind, or like looking away from a bright light. She is on no regular medication. Examination including fundoscopy, Humphrey visual field testing, colour vision, pupillary reflexes and visual acuity was normal. MRI brain scan and VEPs are within normal limits.
Case 4
A 33-year-old female first experienced visual symptoms at the age of 30 years. This was described as zigzag lines and blurred vision over the left hemifield. There was no associated headache or other neurological symptoms. It settled after about 15 min. Six months later she had a similar phenomenon on the right side lasting about an hour. She described it ‘like her vision being shunted inwards’. She felt slightly light headed and that the visual world had ‘altered in perspective’. Her vision has never been quite the same since with a constant feeling of looking at a bright light over the right hemifield with superimposed flashes of light every few minutes. She has exacerbations of the visual disturbance for 20–60 min every 3–6 months. Her interictal visual disturbance has become progressively worse after each of these attacks. Although the visual episodes have never been associated with headache, she has left sided throbbing headaches of moderate intensity twice a week associated with photophobia, phonophobia and exacerbated by movement. She is otherwise fit and well and on no regular medication. Examination including fundoscopy, Humphrey and visual fields was normal. MRI brain scan and VEPs are normal.
Magnetic resonance imaging
Standard axial T2-weighted and coronal FLAIR sequences where normal in all four patients and showed no areas of infarction.
Diffusion-weighted imaging
Visual inspection of the DW trace images and ADC maps showed no areas of abnormally increased or decreased water diffusion (Fig. 1). ADC measurement over the selected cortical areas for all four patients are listed in Table 2. The measured ADC values of the occipital cortices ranged from 0.811 × 10−3 mm2/s to 0. 937 10−3 mm2/s and the relative ADC measurement from 95% and 105% of the mean ADC value of all regions in the individual patient. Two-way
ADCav of the analysed cortical regions in all four patients

The ADCav values are expressed as × 10-3 mm2/s. The number in brackets is the percentage of the mean ADC of all regions in the specific patient.
Perfusion-weighted imaging
The maps of mean transit time (MTT), bolus arrival time (BAT) and relative cerebral blood volume (rCBV) appears symmetrical in all patients and showed no regional areas of hyper- or hypo-perfusion. Narrowly windowed rCBV images, designed to show grey matter and white matter perfusion separately and to reveal subtle differences in blood volume not apparent on overall rCBV maps were also symmetrical and showed no regional defects in the occipital or elsewhere (Fig. 2). Similarly there was no significant difference in the width and peak height of the time-signal intensity curves over posterior, middle cerebral and anterior cerebral artery territories (Fig. 3).
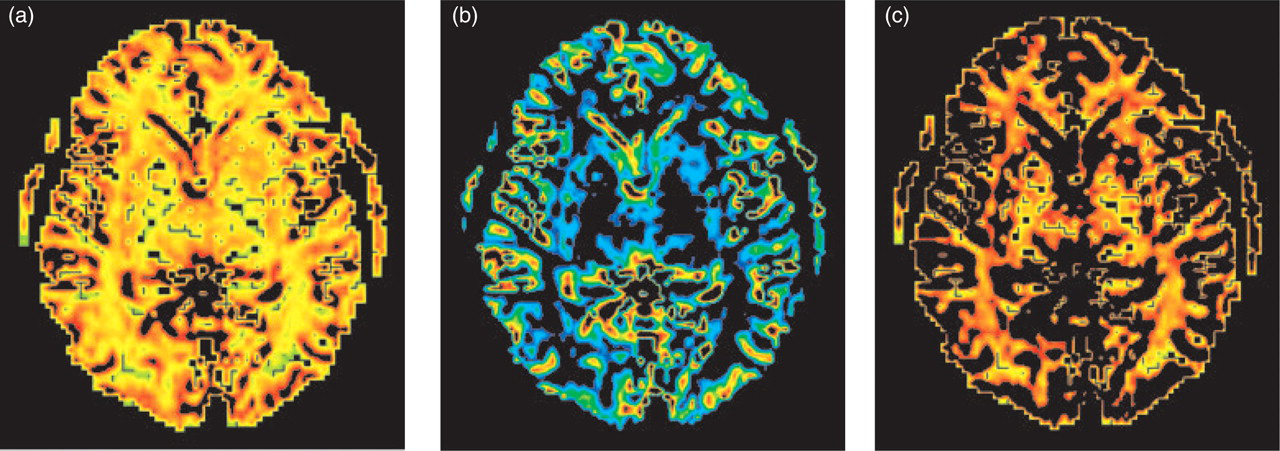
Maps of regional cerebral blood volume: (a) represents a general rCBV map, (b) and (c) represent narrowly windowed ADC maps to demonstrate grey and white matter perfusion, respectively. Neither of the rCBV maps shows asymmetries nor areas of regional hypo or hyperperfusion.
Time-signal intensity curves over the right anterior, middle and posterior cerebral artery territories (a and b) and over the right and left posterior cerebral artery territories (c and d) show no significant differences.
Discussion
Four patients with long-standing disturbing subjective visual abnormalities have been studied in detail by MRI techniques designed to detect subtle physiological disturbances at the microstructural level. In two patients with migraine with persistent visual aura and two patients with what we have termed Primary Persistent Visual Disturbance (PPVD), MR studies were entirely normal. The data suggest that these visual disturbances involve subtle functional alterations in brain processing of visual inputs. Moreover, we propose, on clinical grounds, that Primary Persistent Visual Disturbance, often called by patients visual snow, be classified and studied as an independent entity without any assumption of a relationship to migraine with persistent (typical) aura.
Patient cohort
We have studied four patients whose chief complaint is that their vision is abnormal more or less continuously. Such patients fall into two groups. Our cases 1 and 2 represent a visual disturbance that is persistent and generally involves large areas, if not the entire visual field. Such patients report bright spots, sometimes colourful change or haziness, and often compare their vision to that of a poorly tuned television. The term visual snow is often employed. Liu et al. (10) seemed to be describing such patients in their initial report. Case 2 of Rothrock (21) seems also to fit this description. We have applied the term Primary Persistent Visual Disturbance to this clinical picture to distinguish it from more typical cases with a visual disturbance that is much more migrainous in its description. This contrasts to patients with more typical aura that persists, such as our cases 3 and 4, Haas's original description (3), and case 1 of Rothrock (21). The latter cases seem best classified as migraine with persistent aura, and will be subsumed by the appropriate section of the IHS classification (4). We cannot from our negative findings provide biological evidence that these conditions are different but given the very distinct phenotype we feel they would be best studied separately in the future until the position is clearer. Having seen a child aged 10 describe exactly the same disturbance of visual snow with blotches as adults (PJG, unpublished observation), it is hard to believe that there is not a consistent underlying biology that distinguishes this condition.
Diffusion-weighted imaging
None of our patients showed any abnormalities on the diffusion trace images or ADC maps, including one patient who had reputedly shown signal abnormalities on a T2-weighted MRI scan in the Sylvian fissure and left occipital lobe on an outside MR scan, performed 4 years prior to the current study. This scan was unfortunately not available to us for review to verify the authenticity of these findings, and all subsequent MRI imaging in this patient had been normal.
Changes on diffusion-weighted MRI scans have previously been described in hemiplegic migraine, which can be considered as an extreme form of prolonged aura with motor symptoms, but not in patients with visual aura.
Chabriat et al. (18) were the first to report reversible diffusion-weighted changes in patient with hemiplegic migraine and mutation of CACNA1A gene. Their patient, a 33-year-old woman who had repeated attacks of hemiplegic migraine in the past, underwent MRI scanning at 21 days, 5 weeks and 3 months after her most recent attack. Standard T2- and T1- weighted images were normal at all stages but diffusion-weighted images were abnormal at 21 days and 5 weeks, showing an approximately 25% decrease in diffusion in the affected hemispheres on the earlier scan, when the patient had still a dense hemiplegia. 3 months after the attack, there had been complete clinical recovery with normalization of the diffusion-weighted images. Reversible signal abnormalities have even been described on T2-weighted imaging, during two separate episodes of hemiplegic migraine in a child (22). T2-signal increase was associated with cortical swelling and both had normalized after each episode. Not all studies of patients with migraine aura have detected changes using DWI.
Lindhal et al. (23) did not find any abnormalities on DWI in a 21-year-old patient with hemiplegic migraine studied at 6 h, 4 days and 3 months whilst perfusion imaging was abnormal on the first two scans. DWI was also normal in a study using DWI and PWI in four migraineurs with spontaneous visual auras who underwent MRI scans 20–45 min after the onset of the aura (13). None of these patients showed any areas of restricted diffusion on the trace images and the ADC maps were also symmetrical in all patients. Comparison of the ADC measurements from the affected and contralateral visual cortices showed only minor differences ranging from a 5% increase to a 6% decrease of ADC measurements on the affected side, well within the accepted 10% range of measurement variation of this technique. This degree of variation of the ADC values is similar to the findings in our study where the ADC values in the visual cortices varied between 95% and 105% of the average ADC of all regions, which was statistically not significant. The discrepancy of DWI findings between patients with hemiplegic migraine and visual aura reflects either a fundamental difference in the pathophysiological process or a difference in the magnitude of changes. Given the profound changes of cerebral oedema with coma and death that can be seen with relatively mild head trauma (24) in patients with the S218L CACNA1A mutation (25), it seems likely that the biology underlying some forms of aura does involve alterations of water diffusion. Alteration of water diffusivity may not play a significant role in visual aura or be so subtle that current measurements techniques with an inherent variability of approximately 10% are too crude to detect them.
One way of improving the detection of subtle differences in MR parameters is to perform a histogram analysis of MR measurements between groups of patients and normal controls. This technique has been used to analyse mean diffusivity and fractional anisotropy of normal appearing brain tissue in patients with multiple sclerosis (26), leukoaraiosis (27) and cerebral autosomal dominant arteriopathy with subcortical infarcts and leuko-encephalopathy (CADASIL) (28). In these diseases the histogram analysis revealed differences in mean-diffusivity between patients and age-matched healthy subjects, implying the presence of ultra structural damage. This method has been used to analyse differences in diffusion tensor imaging between 34 migraineurs and 17 age-matched controls (29). The authors found a reduction in peak height of the mean diffusivity histogram in normal appearing brain tissue in patients with migraine and hypothesize that this may reflect mild ischaemic damage causing some loss of structural barriers. Interestingly the changes in mean diffusivity and inferred structural damage were much less marked than in patients with CADISIL, MS or leukoariosis and there was also no difference in mean diffusivity between patients with and without migraine aura (29). Although it would be interesting to apply this form of analysis to patients with visual disturbances such as we describe here, the relative rarity of this condition makes adequate data for a group comparison difficult to obtain.
Perfusion-weighted imaging
Alterations in cerebral perfusion have been well documented in patients with migraine.
Before the clinical implementation of MR perfusion imaging, blood flow phenomena in migraine had been studied with radioisotopes, including intracarotid injections of 133Xenon, single photon emission tomography (SPECT) (30) and positron emission tomography (PET) (31). Olesen et al. (30) reviewed the timing and topography and blood flow changes in migraine with aura in 63 patients. On repeated radioisotope studies, they observed a decrease in blood flow posteriorly in one hemisphere before and during the aura and a gradual change from abnormally low to abnormally high blood flow in these areas during the headache phase. The hyperaemic phase outlasted the headache phase in some patients. Andersson et al. (31) studied 11 patients with red wine induced migraine using PET. During migraine aura they found only a small area of decreased oxygen metabolism in the thalamus but no significant blood flow alterations in the visual cortex. They found, however, a decrease in occipital blood flow and oxygen metabolism (rCMRO2) in the headache phase. Flow and metabolism were therefore reduced in parallel so that the regional oxygen extraction (rOER) remained unchanged. The authors argue that this makes a primary vascular cause of CBF reduction unlikely and supports the neural spreading depression hypothesis. This is consistent with current views of the basically neurovascular physiology of such changes in migraine (32).
The first study of migraine aura to use MR perfusion imaging was performed by Cutrer and colleagues (13). This was part of the abovementioned study of four patients with spontaneous visual aura, who had negative findings on diffusion-weighted imaging. On MR perfusion, all four patients showed hypo-perfusion of the occipital cortex contralateral to the affected hemifield. The relative cerebral blood flow (rCBF) was decreased by 16–53% and relative cerebral blood volume (rCBV) by 6–33%, whereas the mean transit time was increased by 10–54%. The same group subsequently performed a MR perfusion study in a larger cohort of 19 migraineurs during 28 migraine episodes, including 13 patients without migraine aura. This study confirmed a decrease of rCBF and rCBV, on average by 27% and 15%, respectively, and an increase of MTT (on average by 32%) during visual aura, which persisted up to 2.5 h into the headache phase. However, no significant perfusion changes were found in migraine without aura.
In hemiplegic migraine hyperperfusion of the contralateral hemisphere has been described as the dominant feature in three case reports. Two of these used MR perfusion imaging (23, 33) and one used SPECT (34) at 6 h, 47 h, 14 days after the onset of symptoms, the hemiplegia having resolved in one of the three cases at the time of the MRI scan. The literature suggests that perfusion changes in migraine are complex and multiphasic. Hypoperfusion appears to be the predominant feature in the aura phase and can certainly extend into the headache phase (30).
In our patients with persistent visual phenomena we did not detect any regional changes in relative cerebral blood volume (rCBV) or mean transit time (MTT). With the available software we were not able to perform direct measurement of the rCBF. According to the central volume principle rCBF is directly related to the rCBV and MTT (rCBF = rCBV/MTT). Any reduction in rCBF would have therefore have entailed either a decrease in relative cerebral blood flow or increase in MTT, which we did not observe. Whereas diffusion-weighted imaging allows absolute quantification of the apparent diffusion coefficient (ADC), absolute quantification of cerebral blood flow with susceptibility-weighted MR bolus tracking is currently still unreliable. With this method, mean transit time measurements are less subject to model assumptions than CBF measurements and therefore the more robust of the two parameters (35). Cerebral perfusion changes in the order of magnitude previously described in migraine with visual aura would have certainly be detectable with our method and shown up as regional differences on the colour maps of MTT and rCBV, particularly as we used special windowing techniques to demonstrate white and grey matter perfusion separately. Significant changes in occipital lobe perfusion appear therefore not to be implicated in the generation of the visual phenomena our patients report. The presence of very subtle differences in occipital perfusion can, however, not be excluded. Such small changes may become more readily detectable with improvement in MR technology, including high field imaging at 3 Tesla and above, which improves both spatial resolution and signal to noise ratio and with more quantitative MR perfusion methods such as arterial spin labelling (36).
In summary, we were not able to detect any significant changes in cerebral water diffusion and perfusion in patients with either migraine with persistent aura or Primary Persistent Visual Disturbance. It appears unlikely that these physiological parameters form a major part of the underlying pathophysiological process. Very subtle changes of these MR parameters in patients with persistent migraine aura are not excluded and further MR studies using high field imaging and histogram analysis of groups may be able to detect or refute these findings. These visual disturbances should probably be kept separate and their neurophysiology more extensively investigated with modern methods of analysis.
Footnotes
Acknowledgements
The work reported has been supported by the Wellcome Trust. NJG was supported by GlaxoSmithKline. PJG is a Wellcome Trust Senior Research Fellow.
