Abstract
To investigate the hypothesis that early treatment of a migraine attack with sumatriptan, while pain is still mild, results in higher pain free rates in comparison to delayed treatment, when pain is at least moderate, we performed a prospective, controlled and open label study. Migraineurs with or without aura who fulfilled the diagnostic criteria recommended by the International Headache Society were enrolled in the study and randomly assigned to either ‘early’ or ‘late’ treatment with sumatriptan 100 mg tablets. In the early treatment group significantly more patients were pain free at all times measured during two hours after dosing than in the late treatment group. Furthermore, patients in the early treatment group became pain free significantly sooner after dosing than patients who delayed treatment. It is concluded that migraineurs, who are able to differentiate between a migraine attack and other forms of headache, benefit from early intervention with sumatriptan 100 mg tablets.
Introduction
Migraine is a disorder with high prevalence in the western countries and constitutes an enormous public-health problem. The majority of affected patients report pain of at least moderate intensity and at least some degree of disability (1–3). Patients’ priorities when evaluating an acute headache medication include the rapidity of pain relief, the analgesic effect, a low rate of headache recurrence, low tendency to cause nausea, efficacy in treating nausea and vomiting, efficacy in reducing photophobia, oral route of administration, and a low tendency to cause sedation. Achieving complete relief of pain, rather than just a reduction of pain to mild, together with rapidity of pain relief are especially important for patients and will increase patients’ satisfaction with their migraine treatment (4, 5).
For most studies in the past with triptans patients were instructed to delay treatment until pain was at least moderate or severe. The reason for this design was the fact that ‘efficacy’ was described on a 4-point scale as a drop from 3 (severe pain) or 2 (moderate pain) to 1 (mild pain) or 0 (no pain). These protocols, together with a nonvalidated attempt to reduce costs and the belief of some patients that the intake of supposedly ‘strong’ medication like a triptan should be avoided as long as possible, led to the habit of delaying triptan treatment until pain intensity was at least moderate (6). However, this mode of treatment is not supported by recent work showing that acute sensitization may contribute to the pathophysiological progression of a migraine attack (7, 8). Newer clinical data support this theory of acute sensitization suggesting that delaying oral triptan therapy may reduce efficacy and tolerability of triptans. In these analyses early intervention with oral triptans (when pain was still mild) resulted in higher pain free rates, lower recurrence, faster pain relief and better consistency of pain relief (1, 9–15). Consequently, there should be a higher chance to achieve what patients want from an acute migraine treatment when oral triptans are taken as early as possible and when headache is still mild. Furthermore, it has been postulated that a reduced consumption of tablets together with earlier and sustained restoration of patients productivity may translate into health economic advantages (16).
Information regarding the impact of timing of treatment with sumatriptan on clinical outcome is limited, but data suggests that early intervention with sumatriptan results in a greater pain free percentage than when treatment is delayed (1, 9, 10, 17). In a posthoc analysis of a randomized, double-blind, placebo-controlled, and crossover study (9) a subgroup of patients who treated a migraine attack early was found to have higher pain free rates than expected and a trend towards lower headache recurrence rates compared to when moderate or severe headaches were treated (10). A retrospective analysis of migraine attacks treated during the mild pain phase using data from three clinical trials yielded similar results with a higher percentage of patients experiencing freedom of pain and sustained pain free response and a reduced need for redosing (1). Two recently performed double-blind studies evaluated the benefits of sumatriptan 50 mg and 100 mg tablets vs. placebo at the first sign of pain. In these prospective trials early intervention with sumatriptan resulted in higher efficacy rates compared to earlier trials, when sumatriptan treatment was delayed until attacks were fully developed (17).
A controlled study to compare prospectively the effects of sumatriptan taken early during an attack with the effects when sumatriptan is taken when pain is at least moderate or severe has not been previously undertaken. This is the first study to directly compare in a prospective, controlled clinical trial the more traditional mode of treatment with sumatriptan, when a migraine attack is fully developed, with an early intervention concept with sumatriptan, when pain is still mild. From earlier retrospective data it was hypothesized that early intervention with sumatriptan would lead to higher pain free rates two hours after dosing.
Methods
This multicentre, controlled, and open label trial in parallel groups complied with the Declaration of Helsinki and the Good Clinical Practice Guidelines. The study was performed at 15 sites in Germany over a six month period. Approval for the study was obtained from local ethics committees and informed consent was obtained from patients. Participants received no monetary payments for taking part in the study.
Patients
Male and female (of non childbearing potential or with an acceptable form of contraception) patients between 18 and 65 years of age who fulfilled the criteria recommended in the guidelines for controlled trials of drugs in migraine by the International Headache Society Clinical Trials Subcommittee (18) were considered for the study. Inclusion and exclusion criteria are listed in Table 1. The following concomitant medication was feasible during the study: One single migraine prophylactic drug was allowed, provided that the patient had been on a stable dose for at least three months. Ergotamine-containing drugs or other triptans were not allowed 48 h before until 24 h after intake of study medication. Analgesics, antiemetics and other medication effective in migraine were not allowed 24 h before until 2 h after intake of sumatriptan.
Inclusion and exclusion criteria

Study design and treatments
Patients had to come to the clinic twice. At the first visit (screening visit) the diagnosis of migraine with or without aura according to the IHS criteria was made and eligibility for study participation was determined. In female patients of childbearing potential a pregnancy test was performed to rule out pregnancy. Patients were allocated to one of two possible treatment groups, either ‘early intervention’ or ‘delayed intervention’. A random sequence of treatment allocation numbers was generated by computer at the biostatistics department of GlaxoSmithKline Germany in advance and used to randomize patients centrally by a fax/telephone system at entry to the study, maintaining a global allocation ratio of 1 : 1. By centralized randomization, investigators had no influence on the allocation of patients to the two treatment groups in this open study.
In both treatment groups patients received a simple attack report form and study medication (one tablet of sumatriptan 100 mg) and were instructed in detail how to document an attack and when and how to use the study medication. Additionally, every patient received a leaflet with the same instructions to take home. Only attacks starting with an identifiable mild headache phase were to be treated with study medication. Migraine attacks starting with moderate or severe headache without a preceding identifiable mild headache phase (e.g. patient awakens with moderate or severe migraine headache) were not to be treated with study medication. Instead, patients were instructed to treat these attacks with their usual medication.
In the ‘early intervention’ group patients were instructed to take sumatriptan at the first sign of pain and not later than one hour after onset of pain (grade 1). In the ‘delayed intervention’ group patients were instructed to wait until pain was at least moderate or severe (grade 2 or 3) before taking the study medication (Fig. 1). In case of no or an insufficient response two hours after treatment with study medication patients in both groups were allowed to take their usual migraine medication for rescue. In case of relapse or recurrence of migraine pain patients were also allowed to take rescue medication. To ensure documentation of the attack as exactly as possible patients were instructed to fill in their attack report form continuously during the attack. For example, intensity of headache was documented by the patients right before study medication was taken and again at fixed timepoints after intake of sumatriptan.

Schematic drawing of the study design.
Within one week after treating a migraine attack with study medication patients came back to the clinic for a final visit. Attack report forms were returned and in female patients of childbearing potential another pregnancy test was carried out.
Objective, endpoints and definitions
The objective of this study was to evaluate if early intervention would increase pain free efficacy of sumatriptan. Definitions of endpoints complied with the definitions in the guidelines for controlled trials of drugs in migraine by the International Headache Society Clinical Trials Subcommittee (18). The primary endpoint and primary measure of efficacy was the percentage of patients ‘pain-free’ at 2 h after dosing in both treatment groups. Secondary endpoints included the percentage of patients pain free 30, 60 and 90 min after intake of study medication, percentage of patients with ‘migraine related symptoms’ (nausea, vomiting, photophobia and phonophobia 30, 60, 90 and 120 min after medication) and the percentage of patients with ‘recurrence’ (return of headache within 24 h after pain free response at two hours) or ‘relapse’ (return of headache within 48 h after pain free response at two hours). Furthermore, the incidence of adverse events was evaluated.
Statistical analysis
The sample size of 112 patients per treatment group was calculated under the assumption of a 20% increase in efficacy of sumatriptan early intervention (i.e. improved response rate from 35% with delayed to 55% with early intervention based on a previous analysis) (10). A power of 80% and a two sided type-I error of 0.05 were used for this calculation and a correction for an anticipated 10% drop-out rate was made.
The primary population for the efficacy analysis was the intent to treat population (ITT). In case of no or inconsistent postrandomization information, no indication of any drug intake or no adequate migraine attack (no identifiable mild pain phase/headache did not start with grade 1) patients were excluded from the ITT population. Other cases with missing response information (but known study drug intake) were rated as nonresponders (conservative worst-case scenario). A per protocol population (patients who strictly adhered to the protocol) was also defined and efficacy results were calculated for this secondary population.
Response rates were compared with a two-sided Fisher's exact test between the two treatment groups. Time to becoming pain free was modelled with a 2 times 4 (for the times 30, 60, 90, and 120 min after tablet intake) contingency table and tested with the respective χ2-test. Specific care was taken to descriptively analyse the comparability of both treatment groups in terms of demographic baseline infor-mation and migraine history of patients as well as documented relative compliance of the time of intake of the study medication and possible interaction of it to the severity of the attack. All other secondary efficacy and safety analyses were also descriptive.
Results
Intent to treat population
Of 227 patients enrolled in the study, 153 patients were evaluable for the intent to treat analysis, 83 patients in the early and 70 patients in the late intervention group. Reasons for excluding patients from the intent to treat analysis were no attack meeting the specified criteria in the time frame of the study (no attack with an identifiable mild pain phase during three months after the screening visit), not taking study medication, no diary available, inconsistent data or lost to follow up. Demographic data and migraine characteristics in both groups were comparable (Table 2). The frequency of migraine attacks with aura was nearly identical in both groups (24.1% in the early, 24.3% in the late intervention group). The mean time interval to treatment after start of headache was 35 min in the early intervention group (median 20 min; 25% and 75% quantiles 10 and 40 min, respectively; range 0–345 min) and 209 min in the late treatment group (median 90 min; 25% and 75% quantiles 45 and 270 min, respectively; range 0–2025 min).
Demographic data of patients and migraine characteristics

ITT: intent to treat population. SD: standard deviation. M: male, F: female.
Figure 2 shows the cumulative pain free rates at defined times after dosing for both groups during the first two hours after treatment. The percentage of patients with complete pain relief at 2 h (primary endpoint) was significantly higher (P = 0.043) in the early intervention group (n = 59/83, 71.1%, 95% confidence interval (95% CI) 60.1–80.5%) when compared to the late intervention group (n = 38/70, 54.3%, 95% CI 41.9–66.3%). Furthermore, at all other times evaluated after dosing significantly more patients were pain free in the early intervention group (30 min: 10/83, 12%, 95% CI 6–21% vs. 1/70, 1.4%, 95% CI 0–7.7%, P = 0.01; 60 min: 32/83, 38.6%, 95% CI 28.1–49.9% vs. 6/70, 8,6%, 95% CI 3.2–17.7%, P < 0.0001; 90 min: 48/83, 57.8%, 95% CI 46.5–68.6% vs. 21/70, 30%, 95% CI 19.6–42.1%, P = 0.0006). Figure 3 shows the percentage of patients pain free for the first time at the end of a defined time interval during the first two hours after dosing, early (30 min: 10/83, 12%, 95% CI 6–21% vs. 1/70, 1.4%, 95% CI 0–7.7%; 60 min: 23/83, 27.7%, 95% CI 18.5–38.6% vs. 5/70, 7.1%, 95% CI 2.4–15.9%; 90 min: 16/83, 19.3%, 95% CI 11.4–29.4% vs. 15/70, 21.4%, 95% CI 12.5–32.9%; 120 min: 12/83, 14.5%, 95% CI 8–23.9% vs. 17/70, 24.3%, 95% CI 14.8–36%; early vs. late intervention, respectively). Patients in the early intervention group became pain free significantly sooner after dosing than patients in the late intervention group (overall P = 0.0004). Further secondary analysis of the patients who treated early revealed that significantly more patients who took sumatriptan within 20 min after the onset of headache were pain free at two hours (n = 36/42) in comparison to patients who took sumatriptan in the interval 20–60 min (n = 23/41, P = 0.004). Table 3 shows the results for accompanying migraine symptoms in both groups. Due to the delay of treatment in the late intervention group these patients were in a more severe condition as reflected by higher pre medication rates of nausea, vomiting, photophobia and phonophobia. Consequently, at all times measured during the 2 h after dosing less patients in the early intervention group were affected by these symptoms. Rates for recurrence (n = 6/59, 10.2% and n = 4/38, 10.5% in early and late intervention group, respectively) and relapse (n = 10/59, 16.9% and n = 5/38, 13.2% in early and late intervention group, respectively) were low and comparable.

Cumalative pain-free rates at defined times after dosing in the ‘intent to treat’ population. Only patients who reported pain-free at 2h were rated as a success and are included. ▪ late intervention; □ early inervention. ∗P < 0.05.
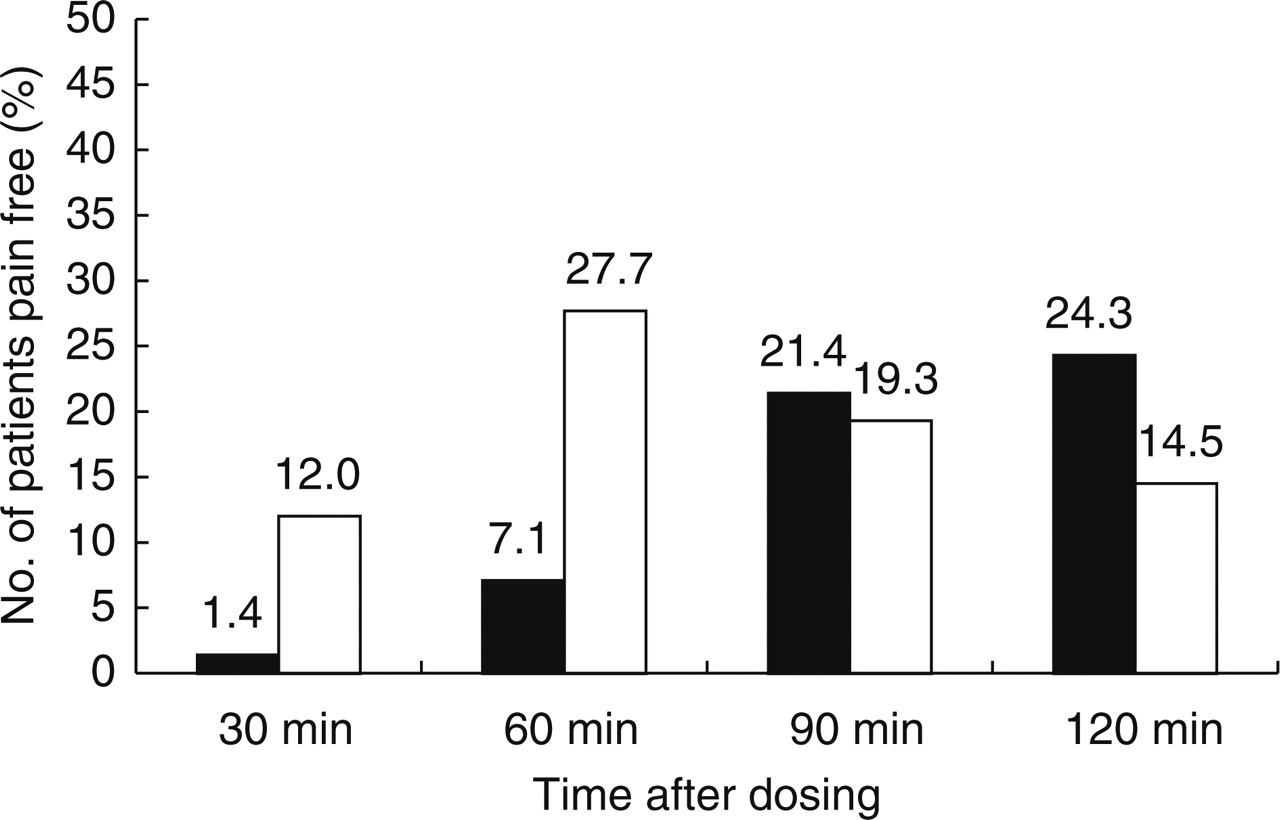
Patients that are pain-free for the first time at the end of defined times in the ‘intent to treat’ population. All patients who became pain-free at any time during 120 min after dosing are included. ▪ late intervention; □ early inervention.
Accompanying migraine symptoms at defined times after dosing

Late, late intervention group (n = 70); early, early intervention group (n = 83).
Per protocol population
For the per protocol analysis another 14 patients were excluded from the intent to treat set, leaving 139 evaluable patients (73 in the early and 66 in the late intervention group). Most patients excluded belonged to the early intervention group and were taken out from analysis due to the fact that they did not treat their migraine when pain was still mild but later, when pain was at least moderate (n = 8). Other reasons for excluding patients were a migraine attack in the 48 h before taking study medication (n = 1) or comedication with other drugs effective in migraine before taking sumatriptan (n = 5).
Figure 4 shows the cumulative pain free rates at defined times after dosing for both groups during the first two hours after treatment. The percentage of patients pain free after 120 min was 74% (95% CI 62.4–83.6%, n = 54/73) in the early treatment group compared to 54.5% (95% CI 41.8–66.9%, n = 36/66) in the late intervention group (P = 0.021). Furthermore, at all other times evaluated after dosing significantly more patients were pain free in the early intervention group (30 min: 10/73, 13.7%, 95% CI 6.8–23.8% vs. 1/66, 1.5%, 95% CI 0–8.2%, P = 0.01; 60 min: 29/73, 39.7%, 95% CI 28.4–51.9% vs. 6/66, 9.1%, 95% CI 3.4–18.7%, P < 0.0001; 90 min: 44/73, 60.3%, 95% CI 48.1–71.6% vs. 19/66, 28.8%, 95% CI 18.6–41.8%, P = 0.0003).

Cumalative pain-free rates at defined times after dosing in the per protocol population. Only patients who reported pain-free at 2h were rated as a success and are included. ▪ late intervention; □ early inervention. ∗P < 0.05.
Adverse events
For analysis of adverse events all patients who took study medication and were available for follow up were evaluated (n = 182 out of n = 227 who entered the study; early intervention n = 96, late intervention n = 86). No serious adverse events were reported during the study. In the early intervention group 21.9% (n = 21) of patients reported any adverse event while in the late intervention group 11.6% (n = 10) experienced an adverse event (Table 4). Adverse events considered by investigators as drug related were observed in 13.5% (n = 13) of patients in the early intervention group and in 5.8% (n = 5) of patients in the late intervention group and are reported in Table 5.
Adverse events

Late, late intervention group (n = 70); early, early intervention group (n = 83). Possibly multiple entries per subject. CNS, central nervous system; PNS, peripheral nervous system; n, number of patients.
Adverse events considered by investigators as drug related

Possibly multiple entries per subject. CNS, central nervous system; PNS, peripheral nervous system; n, number of patients.
Discussion
The aim of this randomized, controlled clinical trial was to compare two modes of treatment with sumatriptan, delayed and early intervention, regarding efficacy and tolerability. Early treatment with sumatriptan (within 60 min after beginning of the pain phase) significantly improved treatment success as measured by pain free rates, speed of becoming pain free and accompanying migraine symptoms. Further improvement may be possible when sumatriptan is taken even earlier (within 20 min after onset of headache). No serious adverse events were reported during the trial. Both, early and late treatment were well tolerated although adverse events and drug related adverse events appeared to be more frequent in the early intervention arm.
Certain features of migraine headache are indicative of increased intracranial mechanosensitivity (worsening of headache by sudden head movement, climbing stairs or coughing) suggesting sensitization of meningeal afferents to mechanical stimuli as a possible pathophysiological correlate (7, 8). Evidence for peripheral and central sensitization of pain neurons in migraine has been found in animal models (7, 19) as well as in humans (8, 20, 21). Acute sensitization may therefore contribute to the pathophysiological progression of a migraine attack and hence the therapeutic rationale should be to suppress development of peripheral and central sensitization. Early treatment, before pain progresses to moderate or severe, could theoretically reduce or even avoid sensitization and hereby contribute to an improvement of efficacy of an acute migraine medication.
Although triptans were investigated in the past regarding treatment effects when administered during the aura phase without demonstrating any benefits in terms of prevention or delay of headache (22, 23), at present there is little information regarding the timing of acute migraine medication use in relation to headache onset. In most reports conclusions were drawn from retrospective analyses from studies initially not designed to evaluate a potential benefit of early intervention with triptans. Such post hoc analyses, based on pre-existing data that were collected for some other purpose, have to be considered as studies that ‘happen’ rather than actually being ‘designed’ and have several drawbacks including variation in the treatments and methods of evaluation used, missing observations and a general vagueness about what was done and why (1, 13, 15, 24–27). Therefore, no definite conclusions can be drawn from these studies only a hypothesis can be generated.
This present study is the first to prospectively and directly compare the effects of sumatriptan 100 mg early intervention with delayed intervention. Patients included in this study were all sumatriptan naive and had a history of comparable migraine attacks with attacks starting with a mild pain phase and progressing to at least moderate pain intensity. Furthermore, patients had to have a defined frequency of migraine attacks of 1–6 per month. Patients were centrally randomized to one of the two treatment modes and extensively informed on how and when to take the study medication. Finally, only attacks starting with an identifiable mild pain phase were treated with study medication and evaluated for the study, hereby assuring that the characteristics of migraine attacks were comparable in both groups.
Results from our study confirm the hypothesis generated from data in the above cited retrospective and prospective sumatriptan analyses, indicating that migraine pain may be more difficult to treat when it has progressed to moderate or severe stages. Furthermore, this data suggests that central sensitization may start to develop within 60 min after onset of pain and hence patients would benefit from treating their migraine attack as soon as possible. Patients who waited before treating their attack suffered more and for a longer time from accompanying migraine symptoms. This is not surprising and reflects the fact that patients who treat a migraine attack before it is fully developed do not reach the same level of disability at baseline like patients who delay treatment. This may be one explanation for the increased efficacy of sumatriptan early intervention in comparison to late intervention. Surprising is the fact that more patients in the early intervention group seemed to experience drug related adverse events. However, even though the absolute number of adverse events was low in this study, it is evident that the most common reported adverse events were dizziness and nausea, symptoms that are not only possible side-effects of sumatriptan but may also be caused by the migraine attack itself. It is very possible that the mode of reporting adverse events and the perception of patients plays an important role here. It can not be ruled out that in the late treatment group symptoms were interpreted as migraine related while in the early treatment group symptoms were rather interpreted as drug related.
Another point that is noteworthy is that in comparison to former trials with sumatriptan, we observed a high absolute rate of pain free response in the early and in the late intervention group. This can be explained by the fact that patients in an open label study know that they receive an active migraine treatment in any case and hence may experience a placebo effect higher than in studies with a placebo arm. It has been shown that response rates in migraine trials are significantly higher when no placebo control is used (28). This circumstance, together with the influence of the study situation itself and the fact that patients may feel better cared for in the context of a study may result in higher absolute efficacy rates than expected.
In conclusion, this prospective and controlled clinical trial shows further evidence of acute sensitization in the pathophysiology of a migraine attack and evidence of increased efficacy of sumatriptan 100 mg tablets when a migraine attack is treated early after the onset of pain.
