Abstract
BIBN 4096 BS ([R-(R∗,S∗)]-N-[2-[[5-amino-1-[[4-(4-pyridinyl)-1-piperazinyl]carbonyl] pentyl]amino]-1-[(3,5-dibromo-4-hydroxyphenyl)methyl]-2-oxoethyl]-4-(1,4-dihydro-2-oxo-3(2H)-quinazolinyl)-,1-piperidinecarboxamide) is the first selective, highly potent, small molecule, nonpeptide calcitonin gene-related peptide (CGRP) receptor antagonist, which has been developed for the treatment of acute migraine. The objective of this study was to obtain information on the safety, tolerability and pharmacokinetics of BIBN 4096 BS following single intravenous administration of rising doses (0.1, 0.25, 0.5, 1, 2.5, 5 and 10 mg) in 55 healthy male and female volunteers. The study was of single-centre, double-blind (within dose levels), placebo-controlled, randomized, single rising dose design. Blood pressure, pulse rate, respiratory rate, ECG, laboratory tests and forearm blood flow did not reveal any clinically relevant, drug-induced changes. Sixteen adverse events (AEs) were reported by eight of 41 volunteers after BIBN 4096 BS compared to five AEs reported by four of 14 volunteers after placebo. Approximately two-thirds of all AEs related to active treatment occurred at the highest dose of 10 mg. At this dose level, all AEs were confined to the three BIBN 4096 BS-treated females, and consisted mainly of transient and mild paresthesias. Paresthesias were the single most frequent AE, whereas fatigue was the AE which occurred in the highest number of subjects. Only two AEs were of moderate intensity, all remaining AEs were of mild intensity. No serious AEs were reported. The local tolerability after intravenous administration was good. In summary, intravenously administered BIBN 4096 BS revealed a very favourable safety profile over the dose range tested in both genders. Generally well tolerated at all dose levels, it was of satisfactory tolerability in female subjects at the highest dose of 10 mg. The plasma concentration-time courses of BIBN 4096 BS showed multicompartmental disposition characteristics. Mean maximum concentration (Cmax) values appeared to be dose-proportional. Based on the results from the two high dose levels (5 and 10 mg) with sufficient individual subject data, BIBN 4096 BS exhibited a total plasma clearance (CL) of approximately 12 l/h and an apparent volume of distribution at steady state (Vss) of approximately 20 l, resulting in a terminal half-life (t1/2) of approximately 2.5 h. Inter-individual variability was moderate with a coefficient of variation of approximately 45% based on the area under the plasma concentration-time curve (AUC) values. The mean renal clearance (CLR) was approximately 2 l/h, suggesting that renal excretion plays only a minor role in the elimination of unchanged BIBN 4096 BS.
Introduction
Acute migraine is thought to be associated with dilatation of cranial vessels and activation of the trigemino-vascular system (1–3). In humans, stimulation of the trigeminal ganglion results in unilateral cranial blood flow increases (4) and evokes the release of CGRP in cranial venous blood (5). In addition, CGRP levels have been shown to be increased in the jugular blood during migraine (6) and these levels are normalized concomitant with headache relief (7). CGRP is one of the most potent endogenous vasodilatators known and its vasodilatatory effects have been shown in a variety of vessels, e.g. CGRP released from sensory fibres originating in the trigeminal ganglia dilates cerebral vessels (7); in fact, particularly the cerebral circulation is densely supplied by CGRP-containing nerve fibres which originate in the trigeminal ganglion (4). CGRP is considered to be a key mediator in the mechanism of migraine, and is at present the only neuropeptide which has been reliably measured as being released in primary headaches like migraine (4). The strong evidence for CGRP being implicated in the pathogenesis of migraine headache points to the potential usefulness of a specific CGRP receptor antagonist in the treatment of migraine.
BIBN 4096 BS is a highly potent CGRP receptor antagonist, which has recently been shown to possess selective affinity in the picomolar range for human CGRP receptors. Additionally, it potently and dose-dependently inhibited the facial skin vasodilatation induced by trigeminal stimulation in marmoset monkeys (8). The ability of BIBN 4096 BS to potently block CGRP-evoked vasodilatation has been confirmed in human extracranial (temporal) arteries (9) as well as in human cerebral arteries (10, 11). Moreover, BIBN 4096 BS has been found to block and even reverse the development of tolerance to morphine in the Central Nervous System (12), suggesting a role of CGRP receptors in nociception. Taken together, BIBN 4096 BS is a very promising new compound, which might be of major benefit in the treatment of primary headaches like migraine. The objective of this study was to investigate the safety and tolerability as well as the pharmacokinetics of BIBN 4096 BS following single intravenous administration of rising doses in healthy male and female volunteers. The dose range to be tested of 0.1–10 mg was based on preclinical pharmacology data (start dose determined as one fifth to one tenth of the expected therapeutic dose of about 0.01 mg/kg) (8) and toxicology studies (top dose determined as a fraction of the no-toxic-effect level observed in animal studies).
Methods
Ethics
The study protocol was reviewed and approved by the Independent Ethics Committee affiliated with the State Medical Council Rhineland-Palatinate (‘Ethik-Kommission der Landesärztekammer Rheinland-Pfalz’), Mainz, Germany, before study initiation. Prior to their enrolment, all participants gave informed consent in writing after a full verbal and written explanation of the study. The study was conducted in accordance with local law, the ethical principles of the Declaration of Helsinki, version as of October 1996, as well as the principles of Good Clinical Practice.
Study population and design of study
Volunteers were healthy, aged 21–47 years and were of normal build. All female volunteers had negative urine pregnancy tests, and used a safe contraception during their participation. No concomitant therapies were allowed, with exception of oral contraceptives. All volunteers were recruited from the volunteers’ pool of the Human Pharmacology Centre Ingelheim (Boehringer Ingelheim Pharma GmbH & Co. KG).
This first-in-human study was conducted in a single-centre, double-blind (within dose levels), placebo-controlled, randomized, single rising dose design. Subjects received either placebo or one of seven BIBN 4096 BS single rising doses. Each of the treatment groups consisted of eight volunteers (six on active drug, two on placebo). Treatments were administered 1 h after a standardized breakfast as intravenous infusion over 10 min (volume 5 ml). Dose levels of 0.1, 0.25, 0.5, 1, 2.5, 5 and 10 mg were investigated sequentially.
Safety and tolerability
Vital signs (i.e. blood pressure and pulse rate after 10 min supine and 2 min standing, respectively, as well as respiratory rate after 10 min supine), ECG (12-lead ECG and, additionally, continuous 2-lead ECG monitoring with heart rate control from 0 to 3 h), routine clinical laboratory tests (haematology, clinical chemistry and urinalysis) and AEs were evaluated before and at regular intervals during the study. Local tolerability at the infusion site was assessed during the infusion and up to 24 h after start of infusion. As additional part of the safety and tolerability assessment, venous occlusion plethysmography of the forearm was performed before and at regular intervals after drug administration. The aim of this noninvasive method was to explore changes in vessel tone and consequently blood flow, as a possible result of CGRP-antagonism, and to reveal drug effects of a safety-relevant magnitude (primary purpose of this method in this study). Forearm blood flow was measured consistently in one forearm using a strain gauge venous occlusion plethysmograph (Gutmann, Eurasburg, Germany). The plethysmograph was operated according to the manufacturer's instructions. Subjects were resting supine and with the forearm positioned above heart level. Strain gauges were applied to the widest part of the forearm (or close to it), and the position was kept constant throughout the measurements. An inflation cuff placed on the upper arm was rapidly inflated to a pressure of 60 mmHg for 6 s, and blood flow was measured over a period of 4 s. Forearm blood flow obtained under these conditions, i.e. with venous drainage briefly interrupted and arterial inflow unaltered, reflected the average arterial inflow at rest. Values were expressed in millilitres of blood per 100 millilitres forearm tissue volume per minute. At each scheduled time point measurements were carried out in triplicate and the mean was recorded. Although forearm blood flow has been described to become nonlinear if the hand is not excluded from the circulation during measurements (13), because of the short inflation and measurement times employed in this study (see above), no relevant nonlinearity in arterial blood flow had been expected. This was eventually confirmed by the obtained plethysmographical wave forms.
Pharmacokinetics
Collection of specimens
Blood samples were collected into potassium-EDTA (ethylenediaminetetraacetic acid) tubes before drug administration, and at 5, 10 (end of infusion), 15, 20, 30, 45 min and at 1, 2 and 4 h after start of infusion at dose levels 0.1–0.5 mg. At dose levels 1–10 mg, additional samples were taken at 6, 8, 12 and 24 h. Plasma samples were harvested immediately by centrifugation. Urine samples were collected before drug administration, and at the following intervals postdose: 0–4, 4–8 and 8–24 h. Plasma and urine samples were stored at −20°C pending analysis.
Drug assay
Plasma and urine samples were analysed by high performance liquid chromatography (HPLC) coupled to tandem mass spectrometry (MS/MS). This assay had a lower limit of quantification of 5 ng/ml for both plasma and urine.
Data analysis
Plasma BIBN 4096 BS concentration-time data were analysed by standard noncompartmental methods using WinNonlin®. The peak concentration (Cmax) for each subject was obtained directly from the observed data. In addition, the following parameters were obtained at the 5 mg and 10 mg dose levels with sufficient data in the 8–24 h collection interval: terminal half-life (t1/2), area under the concentration-time curve from time zero to infinity (AUC0–∞), mean residence time (MRT), total plasma clearance (CL), apparent volume of distribution during the terminal phase (Vz), apparent volume of distribution at steady state (Vss), and renal clearance (CLR).
Statistics
The sample size of eight subjects per group (six on active drug and two on placebo) is commonly used in single rising dose studies, and is in general considered as sufficient for the assessment of safety, tolerability and pharmacokinetics following the first administration of a new agent to man (14). Safety and tolerability of seven different single dose levels of BIBN 4096 BS were determined on the basis of the investigated safety variables in comparison to placebo; they were evaluated by descriptive statistical methods. Pharmacokinetic parameters were derived as described above.
Based on theoretical considerations, a CGRP antagonist might primarily affect blood pressure, heart rate and peripheral blood flow. For this reason the safety parameters blood pressure, pulse rate (both parameters in supine and standing position) as well as forearm blood flow were additionally evaluated by an analysis of covariance (
Because of the exploratory nature of this trial no adjustment of the α-level for multiple comparisons was performed (level of significance fixed at α = 5%, i.e. P-value of 0.05). A large number of statistical comparisons was made for the vital parameters. Therefore, P-values have to be interpreted only as a measure of evidence for an effect in the present data and similarly, confidence intervals have to be interpreted only as interval estimates for effects in the present data, but no generalizing conclusions are possible. The main focus is on the magnitude of the effects and their clinical meaning.
The statistical analysis was carried out with the software product SAS (SAS Institute, Cary, NC USA), version 8.02.
Results
Safety and tolerability
Forty-one volunteers received BIBN 4096 BS and 14 volunteers received placebo. Figures 1–4 summarize the time course of vital signs’ measurements. Statistically significant effects were not detected with respect to supine blood pressure. A statistically significant increase in standing systolic blood pressure compared to placebo was detected at 10 mg BIBN 4096 BS (95% CI 0.4, 12.2; P = 0.0357). The maximum mean increase compared to baseline (predose) was 3.3 mmHg (from 119.2 to 122.5 mmHg) at 1 h, while on placebo a decrease of 5 mmHg (from 119.3 to 114.3 mmHg) was observed (Fig. 2). The magnitude of this systolic blood pressure increase was not of clinical relevance. A statistically significant standing diastolic blood pressure decrease compared to placebo was detected at 2.5 mg (95% CI − 15.5, − 1.9; P = 0.0135). The maximum mean decrease compared to predose was 16 mmHg (from 87 to 71 mmHg) at 1 h, while on placebo a maximum decrease of 2.9 mmHg (from 81.8 to 78.9 mmHg) was observed (Fig. 2). No objective or subjective findings were related to this diastolic blood pressure decrease. Of note is also the high baseline (predose) diastolic blood pressure of treatment group 2.5 mg compared to placebo (difference of approximately 5 mmHg). Moreover, such effects were not detected at the two highest dose levels, indicating that there was no dose dependency. With respect to supine pulse rate a statistically significant increase compared to placebo was observed at 1 mg (95% CI 1.6, 9.2; P = 0.0066). The maximum mean increase compared to predose was 7.6 beats per minute (from 58.7 to 66.3 beats per minute) at 8 h, while on placebo a maximum increase of 4.6 beats per minute (from 60.1 to 64.7 beats per minute) was observed. This finding occurred at a time (8 h) when drug plasma concentrations were no longer detectable (Fig. 6) and was of no clinical relevance. There were no effects on pulse rate in supine position at higher doses (Fig. 3). Finally, differences were not detected for pulse rate in standing position (Fig. 4). There were no clinically significant effects on respiratory rate, routine clinical laboratory tests (haematology, clinical chemistry and urinalysis), 2-lead ECG monitoring with heart rate control from 0 to 3 h and 12-lead ECG. With respect to QTc (heart rate-corrected QT interval using Bazett's formula) a time-matched analysis of central tendency and a categorical analysis were performed as recently suggested (16), for which 12-lead ECGs recorded 10, 30 min and 1, 2, 3, 4, 6, 8 and 24 h after start of infusion were considered (Tables 1 and 2). Baseline values were obtained predose on the day of drug administration. As shown in Table 1 placebo-corrected mean changes were between 0.1 and 12.8 ms. Effects were without dose dependency. In fact, at the three highest dose levels of 2.5–10 mg maximal mean QTc changes were consistently smaller than at lower doses of 0.25–1 mg; at 2.5 mg even a decrease of approximately 4 ms was seen. Moreover, maximal mean QTc changes after 0.1, 0.25, 1 and 2.5 mg were observed at times when BIBN 4096 BS plasma concentrations were no more detectable (Table 1 and Fig. 6). At 0.5 mg, where the second highest increase of approximately 10 ms was detected, only low BIBN 4096 BS plasma concentrations were present at the time of the maximal mean QTc increase, and at all other ECG times only QTc decreases were seen. Besides the 2.7 ms increase after the highest dose of 10 mg of BIBN 4096 BS at 1 h, all other ECG times showed a QTc decrease at this dose. Taken together, the maximal mean QTc increase was below the treshold of 5 ms (16) at 2.5 mg and at the highest dose of 10 mg. The maximal mean QTc increase did not relevantly exceed 5 ms at 1 and 5 mg. The categorical analysis (Table 2) revealed a total of four subjects (at 0.25 mg, 0.5 mg and placebo) with individual QTc increases ≥ 30 ms, without any dose dependency. Once again, changes were even lower after high doses than after low doses; in fact, there were no QTc increases ≥ 30 ms in the four highest treatment groups. The highest increase of 54 ms was seen after 0.25 mg at 8 h when BIBN 4096 BS plasma concentrations were no more detectable (Table 2 and Fig. 6). There were no females with individual QTc increases ≥ 30 ms. No QTc increases ≥ 60 ms or absolute QTc ≥ 450 ms after drug administration were detected. The results of the analysis of forearm blood flow measurements by strain gauge venous occlusion plethysmography are summarized in Table 3 and Fig. 5. There were no statistically significant differences between placebo and any of the seven dose levels tested. The largest changes were obtained at the two highest dose levels; however, while an increase in forearm blood flow was found at 5 mg, a reduction was found at the 10 mg dose level indicating that all changes were in the range of natural fluctuations. Sixteen AEs were reported by eight of the 41 active-treated volunteers after single intravenous BIBN 4096 BS doses compared to five AEs reported by four of the 14 volunteers after placebo (Table 4). The overall AE incidence rate for all active treatment groups was lower than for placebo (19.5 vs. 28.6%), and the average number of AEs per subject showed a similar frequency for overall active (16 AEs in 41 subjects) and placebo (five in 14) treatment of about 0.4 AEs per subject. Higher AE incidence rates compared to placebo were observed at doses of 5 and 10 mg (50%), while the rate at 0.25 mg was found to be at placebo level (33.3%). No AEs were reported at 0.1, 0.5, 1 and 2.5 mg. Approximately two-thirds (11 of 16) of all AEs related to active treatment occurred at the highest dose of 10 mg. These AEs were confined to the three BIBN 4096 BS-treated females of this dose and consisted mainly of transient and mild paresthesias with onset during the infusion (usually 5–7 min after start of infusion), and which usually resolved spontaneously within 15–20 min. In particular, the following was reported by the three female volunteers: flushing and feeling of warmth in the face, feeling of crawling in the head, numb feeling and feeling of cold of hand and forearm at the infusion site; feeling of warmth in the face, stabbing in throat and head, and congestion in the head; feeling of warmth in the upper part of the body and feeling of crawling in the whole body. None of the male volunteers at the highest dose level of 10 mg reported any adverse event. Paresthesias were the single most frequent AE (seven events in three female subjects on active drug), whereas fatigue was the AE which occurred in the highest number of subjects (six subjects, three of them on placebo). Generally, AEs were of mild intensity except for two AEs which were of moderate intensity. These two AEs occurred in two different female volunteers at the 10 mg dose level and included a very transient (13 min) flushing during and after infusion as well as lower abdominal pain. The latter appeared only after more than 35 h after drug administration, at a time where because of BIBN 4096 BS's terminal half-life of about 2.5 h no drug plasma levels could have been expected; it was therefore assessed as not drug-related by the investigator, and resolved spontaneously within 7 h. No severe or serious AEs were reported. The local tolerability after intravenous administration was good; no swelling, induration, warmth or pain was reported. One female subject on 5 mg BIBN 4096 BS showed a mild and very transient (3 min) erythema with itching at the infusion site.

Systolic and diastolic blood pressure after 10 min supine. Graphs display mean values + standard deviation. For better readability only the mean values of the three highest dose levels (▪ 2.5 mg, n = 5; ○ 5 mg, n = 6; • 10 mg, n = 6) and placebo (□n = 14) are shown.

Systolic and diastolic blood pressure after 2 min standing. Graphs display mean values + standard deviation. For better readability only the mean values of the three highest dose levels (▪ 2.5 mg, n = 5; ○ 5 mg, n = 6; • 10 mg, n = 6) and placebo (□n = 14) are shown.
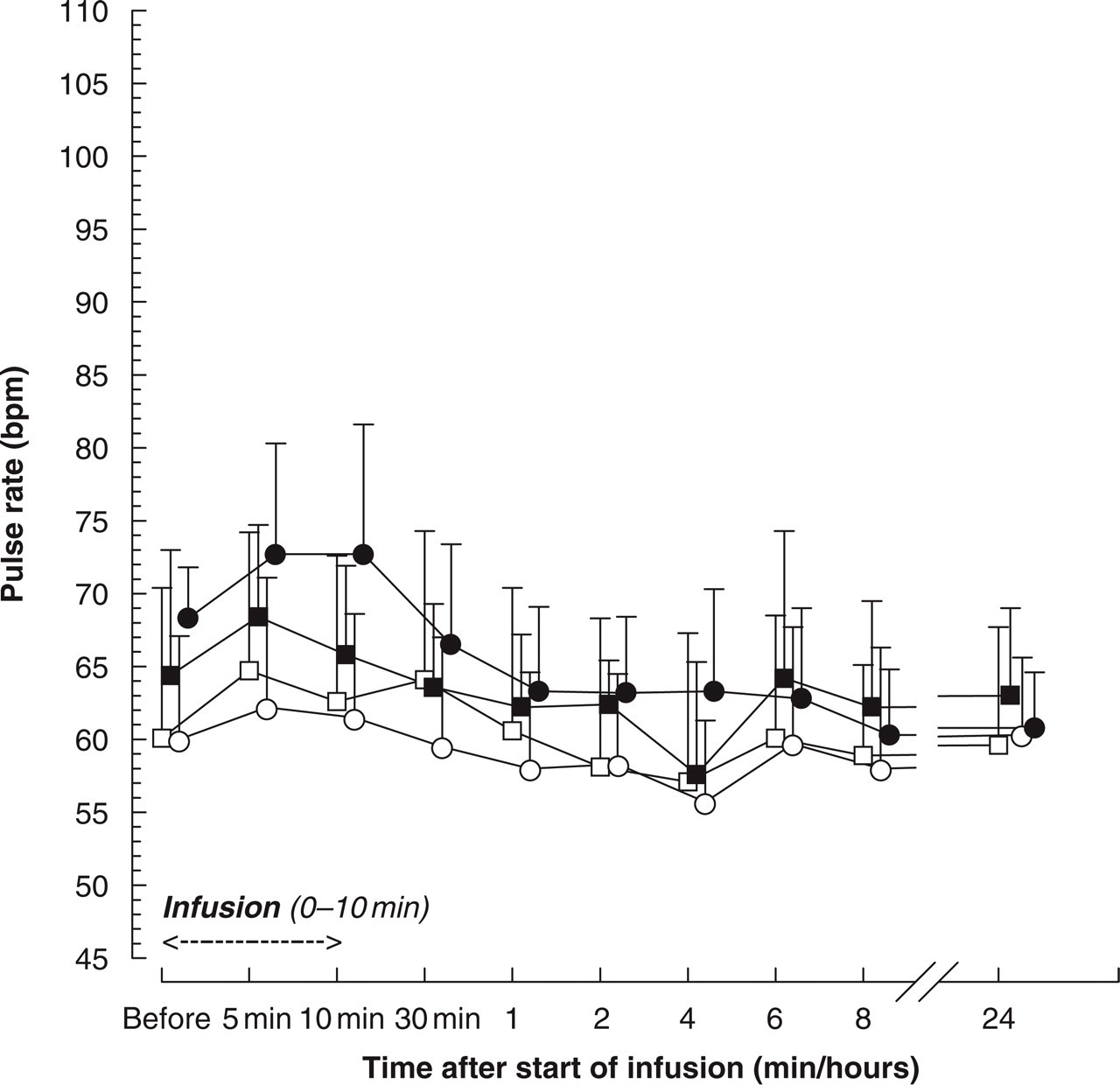
Pulse rate after 10 min supine. Graphs display mean values + standard deviation. For better readability only the mean values of the three highest dose levels (▪ 2.5 mg, n = 5; ○ 5 mg, n = 6; • 10 mg, n = 6) and placebo (□n = 14) are shown. bpm, beats per minute.
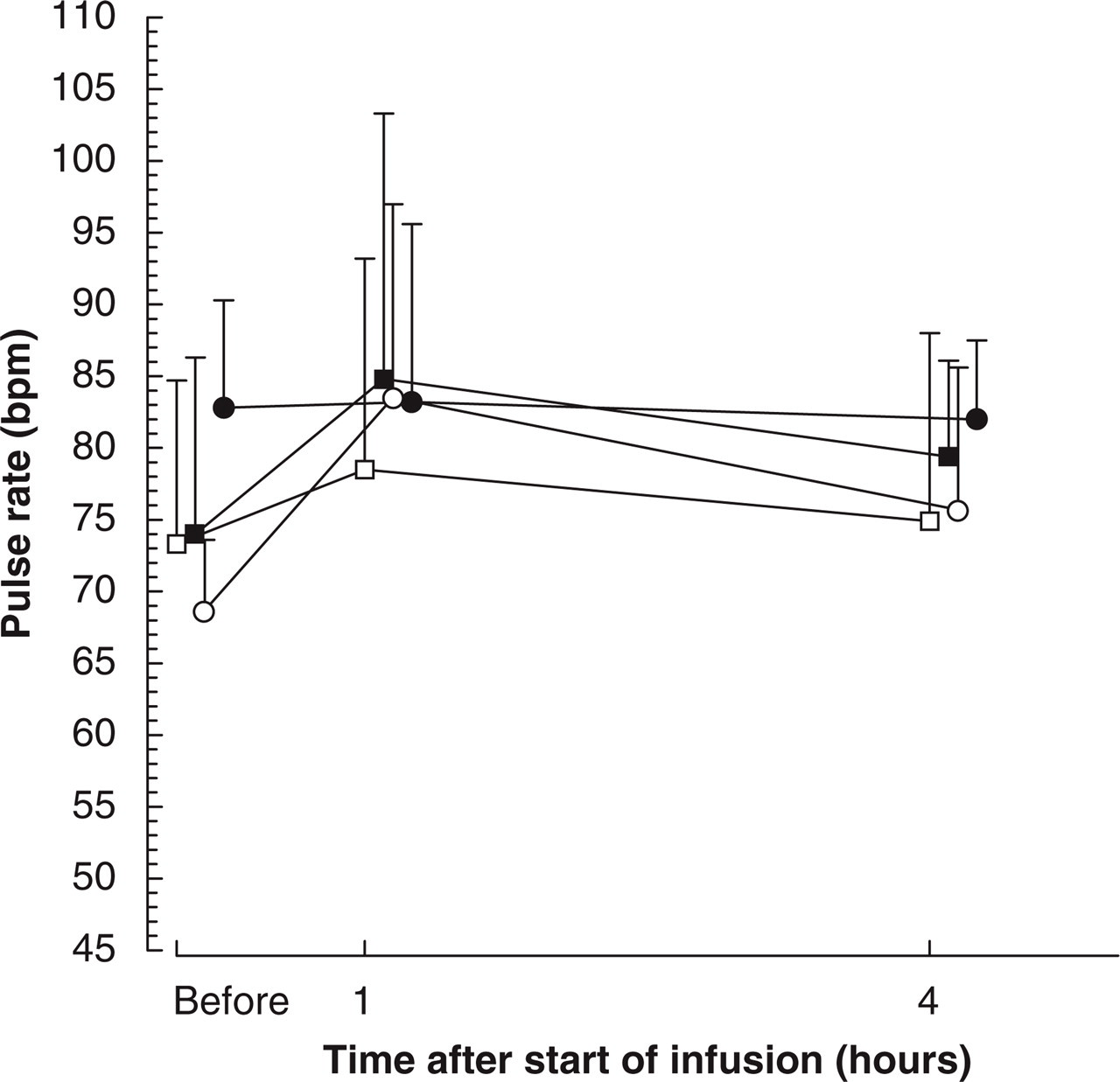
Pulse rate after 2 min standing. Graphs display mean values + standard deviation. For better readability only the mean values of the three highest dose levels (▪ 2.5 mg, n = 5; ○ 5 mg, n = 6; • 10 mg, n = 6) and placebo (□n = 14) are shown. bpm, beats per minute.

Forearm blood flow. Graphs display mean values + standard deviation. For better readability only the mean values of the three highest dose levels (▪ 2.5 mg, n = 5; ○ 5 mg, n = 6; • 10 mg, n = 6) and placebo (□n = 14) are shown.

Mean BIBN 4096 BS plasma concentration-time profiles. (• 0 mg, □ 0.25 mg, Δ 0.5 mg, ▾ 1 mg, ▴ 2.5 mg, ▪ 5 mg, ○ 10 mg)
QTc time-matched analysis of central tendency
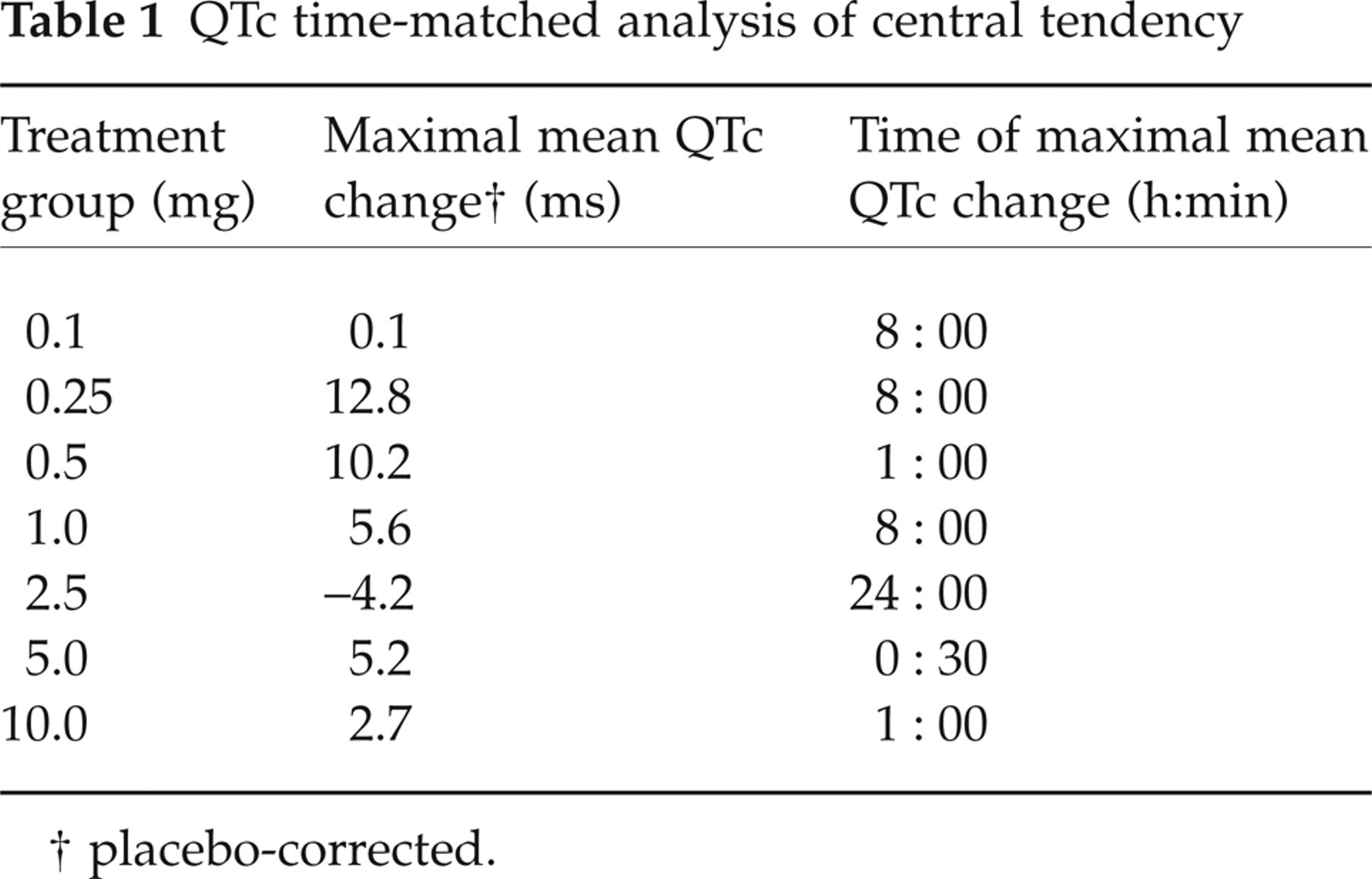
placebo-corrected.
QTc categorical analysis

Least square means and 95% confidence intervals of the differences between treatment groups and placebo for forearm blood flow

baseline-adjusted. Abbreviations: CI, confidence intervals; LCL, lower confidence limit; LSM, least-square means; UCL, upper confidence limit.
Adverse events by preferred term – number of subjects with AEs (and number of AEs) after BIBN 4096 BS and placebo
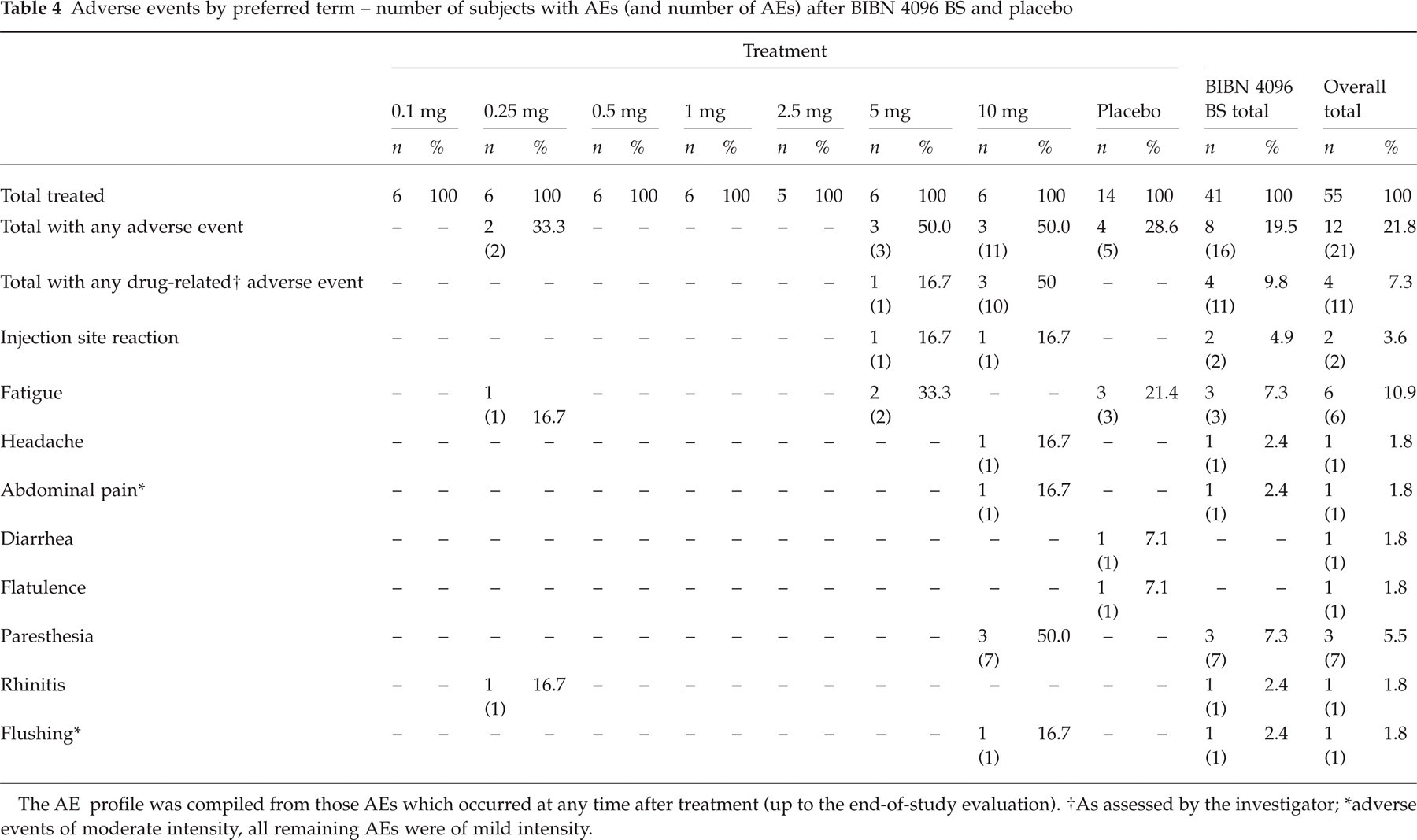
The AE profile was compiled from those AEs which occurred at any time after treatment (up to the end-of-study evaluation).
As assessed by the investigator;
adverse events of moderate intensity, all remaining AEs were of mild intensity.
Pharmacokinetics
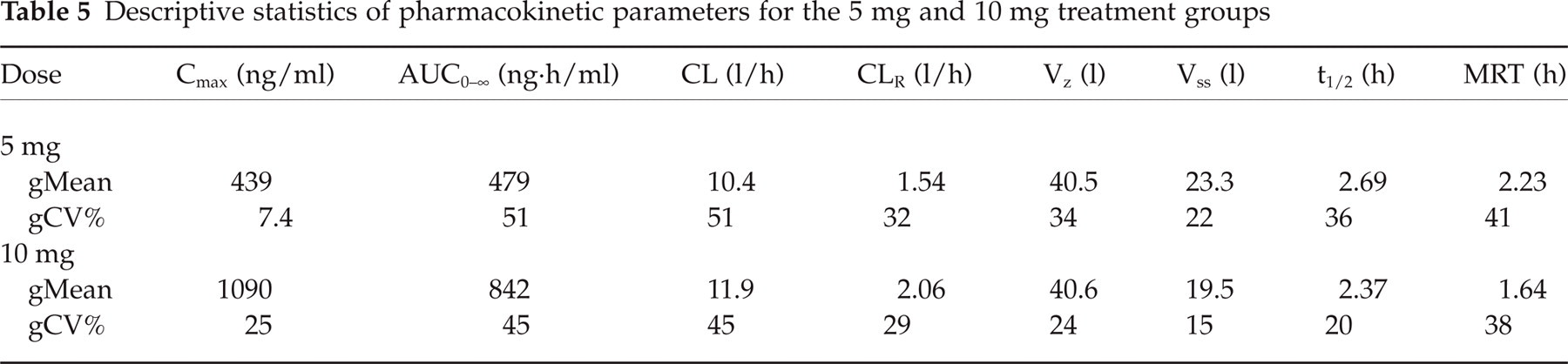
Mean plasma BIBN 4096 BS concentration-time profiles for all dose levels are depicted in Fig. 6. Samples from the lower treatment groups showed measurable concentrations only around the peak concentration. After administration of the two highest dose levels (5 and 10 mg), plasma concentration-time profiles with sufficient data points could be observed that were characterized by multicompartmental disposition characteristics. Geometric mean Cmax values were 10.7, 20.6, 60, 112, 210, 439 and 1090 ng/ml for the respective dose levels and appeared to be dose-proportional. Pharmacokinetic parameters obtained for the 5 and 10 mg dose are summarized in Table 5. BIBN 4096 BS exhibited a total plasma clearance (CL) of approximately 12 l/h and an apparent volume of distribution at steady state (Vss) of approximately 20 l, resulting in a terminal half-life (t1/2) of approximately 2.5 h. Inter-individual variability was moderate with a coefficient of variation of approximately 45% based on the AUC0–∞ values. Approximately 15% of the dose was excreted in urine as unchanged drug, the majority within the first 4 h. The mean renal clearance (CLR) was approximately 2 l/h, suggesting that renal excretion plays only a minor role in the elimination of unchanged BIBN 4096 BS.
Descriptive statistics of pharmacokinetic parameters for the 5 mg and 10 mg treatment groups
Discussion
Doses ranging from 0.1 mg to 10 mg BIBN 4096 BS did not produce any clinically relevant changes in vital signs, ECG and routine clinical laboratory tests. The few statistically significant findings described above were attributable to random findings due to the multiple comparisons performed. There was no clinically relevant effect of BIBN 4096 BS on QTc. The venous occlusion plethysmography did not reveal an effect of BIBN 4096 BS on forearm blood flow. However, the following limitations of the method used have to be considered. There was no positive control, i.e. no compound with known vasoconstrictor properties was administered as a reference compound, and results obtained by venous occlusion plethysmography are known to show a relatively high intra- and inter-individual variability (13, 15). If the results of the
BIBN 4096 BS plasma concentrations declined rapidly below the lower limit of quantification for most of the dose levels. The Vss of about 20 l suggests relatively little penetration into tissues. After intravenous administration to different species, Vss was found to be moderate in rats and rabbits, low in dogs and low to high in monkeys (low in Rhesus, moderate in Cynomolgus and high in Marmoset monkeys). Since the target CGRP receptors are located on extracerebral, intracranial arteries (19), good distribution into tissues as reflected by a high Vss is no prerequisite for CGRP receptor occupancy by BIBN 4096 BS. It has to be shown in migraine patients, whether the relatively short-terminal half-life of about 2.5 h results in sufficient duration of pain relief and low relapse rate. Because of its apparently slow receptor off-kinetics (22, 23), however, BIBN 4096 BS might preserve antimigraine efficacy even at decreased plasma levels. BIBN 4096 BS appears to be eliminated mainly via nonrenal pathways. In the 5 and 10 mg treatment groups, a trend for an effect of oral contraceptives on body weight-corrected AUC could be observed, with higher AUC and lower clearance values in women taking oral contraceptives, whereas there was no such difference between women not taking oral contraceptives and men. Because of the small sample size, however, this finding needs to be confirmed by additional studies. As the metabolism of BIBN 4096 BS has not been investigated in detail yet, possible mechanisms of interaction remain unknown. Irrespectively whether an interaction existed, the occurrence of AEs after treatment with BIBN 4096 BS would be expected to depend rather on higher Cmax than AUC values (see above).
In conclusion, intravenously administered BIBN 4096 BS revealed a very favourable safety profile over the dose range tested and in both genders. Generally well tolerated at all dose levels, it was of satisfactory tolerability in female subjects at the highest dose of 10 mg. Thus, BIBN 4096 BS is a safe and very promising new compound, which might be of major benefit in the treatment of primary headaches like migraine.
Footnotes
Acknowledgements
We would like to thank Vera Lothholz for her assistance in carrying out the study, Dave Dube for his analyses of plasma and urine samples, Elizabeth Potocka for her assistance in pharmacokinetic analysis, and Alexandros Sagriotis for his assistance in statistical analysis.
