Abstract
Triptans are the treatment of choice for acute migraine. While seemingly a homogenous group of drugs, results from a meta-analysis reveal significant differences in efficacy and tolerability among oral triptans. The incidence of drug-related central nervous system (CNS) side-effects with some triptans is as high as 15% and may be associated with functional impairment and reduced productivity. The occurrence of adverse events associated with triptans in general, and CNS side-effects in particular, may lead to a delay in initiating or even avoidance of an otherwise effective treatment. Potential explanations for differences among triptans in the incidence of CNS side-effects may relate to pharmacological and pharmacokinetic differences, including receptor binding, lipophilicity, and the presence of active metabolites. Of the triptans reviewed, at clinically relevant doses, almotriptan 12.5 mg, naratriptan 2.5 mg and sumatriptan 50 mg had the lowest incidence of CNS side-effects, while eletriptan 40 and 80 mg, rizatriptan 10 mg and zolmitriptan 2.5 and 5 mg had the highest incidence. The most likely explanations for the differences in CNS side-effects among triptans are the presence of active metabolites and high lipophilicity of the parent compound and active metabolites. Eletriptan, rizatriptan and zolmitriptan have active metabolites, while lipophilicity is lowest for almotriptan and sumatriptan. If CNS sideeffects are a clinically relevant concern in the individual patient, use of a triptan with a low incidence of CNS side-effects may offer the potential for earlier initiation of treatment and more effective outcomes.
Keywords
Introduction
Migraine is a chronic debilitating disorder that affects as many as 28 million people in the United States. Almost 1 in 4 US households has a migraineur, which represents approximately 6.5% of the male population and 18% of the female population (1). Migraine is most frequent in men and women between the ages of 35–45 years thus affecting individuals at a time in their lives when they are most productive (1).
Despite its prevalence, economic burden on society, and debilitating characteristics, migraine continues to be under diagnosed and under treated (2). The economic impact of migraine due to lost productivity is reported in a number of epidemiological, economic, observational, and interventional studies. The cost to the US economy of lost productivity due to migraine is estimated to range from $13 to $17 billion per year (3, 4). No less important is the impact of migraine on individuals. Migraine patients report missing days from work, loss of effectiveness in the workplace, and reduced levels of productivity after the onset of an acute migraine attack (5, 6). More than half of the migraineurs surveyed reported that their work or school productivity was reduced by at least 50% as a result of a migraine episode (1).
The emergence of the triptans as an effective class of specific acute antimigraine therapy has represented an enormous advance in the management of this disorder. In general, triptans produce a headache response at 2 h in 60% of migraine patients and 30% are pain free at 2 h (7). However, despite the efficacy of triptans in acute migraine, side-effects may reduce patient compliance, limit treatment initiation and even impair workplace productivity and effectiveness. In a recent survey by the National Headache Foundation, 67% of patients reported that they would delay or avoid their prescription medication for migraine pain due to concern about side-effects (8). Thus, side-effects remain a barrier to effective management of migraine disorder.
Results from a meta-analysis of oral triptans showed significant differences among triptans in the overall incidence of adverse events when compared to sumatriptan 100 mg, and specifically the incidence of central nervous system (CNS) events (7). Since the main goals of the acute treatment of migraine are to reduce disability and restore functionality (9), it may be preferable to select those triptans that are not only effective in aborting an attack, but also are least likely to burden the patient with functionally limiting side-effects. An understanding of the physiological effects of triptans and possible physiological explanations underlying the differences in CNS side-effects across triptans may result in more effective migraine treatment. This review will characterize the differences among triptans in the incidence of CNS side-effects, explore the potential mechanisms underlying these differences, and discuss the clinical implications for the migraine patient.
CNS side-effect profile of triptans
The overall placebo-subtracted incidence of side-effects with triptans ranges from less than 2% to over 20% and includes tingling, paresthesia, warm sensations, and flushing (10). While the incidence of CNS side-effects is considerably lower, CNS effects, especially somnolence, may have a greater impact on the patient and their use of migraine medication (8). CNS-related side-effects may play an important role in a patient's decision to delay or avoid treatment with a triptan. Patients who experience triptan-induced dizziness or somnolence may avoid or delay treatment while at work or when performing tasks that require alertness, such as driving or taking care of a child.
The incidence of CNS-related side-effects for 6 triptans, compared to sumatriptan 100 mg as the reference drug and dose, has been reported in a meta-analysis of 53 clinical trials of oral triptans (Fig. 1) (7). Differences may have existed in the data collection methods for side-effects in individual trials, but the use of placebo-subtracted data for the incidence of CNS side-effects gives us an estimate of the magnitude of increased or decreased risk with a given triptan. The most common CNS treatment-related side-effects with triptans were dizziness and somnolence (Table 1). At clinically recommended doses, the incidence of CNS side-effects appeared to be lower with almotriptan 12.5 mg (−1.5%) and naratriptan 2.5 mg (1.9%). In contrast, zolmitriptan 5 mg (11.5%), rizatriptan 10 mg (9.4%) and eletriptan 40 mg (7.5%) and 80 mg (14.6%) exhibited the highest incidence CNS side-effects (Fig. 1). However, with the exception of almotriptan 12.5 mg, the 95% confidence intervals associated with all other triptans and dosages overlapped with sumatriptan 100 mg. A dose-related increase in CNS side-effects was apparent with eletriptan, rizatriptan, sumatriptan and to a lesser extent, zolmitriptan.
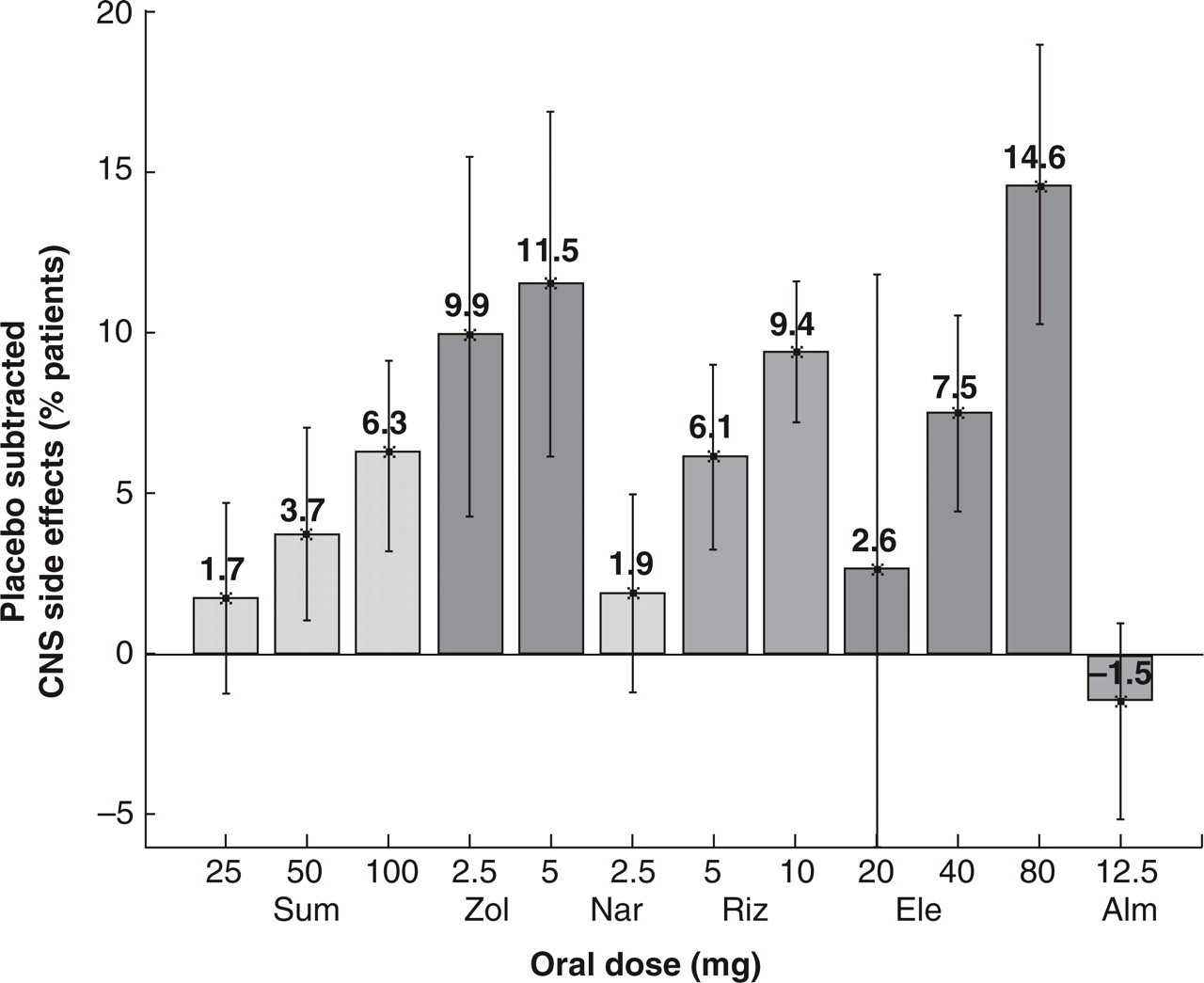
Placebo-subtracted incidence of CNS side-effects for each triptan and dosage (10). Sum, Sumatriptan; Zol, Zolmitriptan; Nar, Naratriptan; Riz, Rizatriptan; Ele, Eletriptan; Alm, Almotriptan.
Central nervous system (CNS) side-effects with oral triptans

Given that direct comparator studies used the same method to collect side-effect data, placebo-subtracted data represent the best way to compare CNS side-effects between the triptans. These findings are supported by head-to-head comparisons of triptans also reported in the meta-analysis (Table 2) and are consistent with the incidence of side-effects reported in product data sheets (10). In this analysis, differences were significant for eletriptan (40 mg and 80 mg) compared to sumatriptan 50 mg, eletriptan 80 mg vs. sumatriptan 100 mg and rizatriptan 10 mg vs. sumatriptan 25 mg.
Incidence of CNS side-effects in direct comparative trials of triptans (Adapted with permission (10))

A negative value indicates that the comparator drug had a higher incidence of side-effects.
P < 0.05,
P 0.05 to < 0.005.
Triptan mechanisms of action
Migraine attacks are believed to be the result of a complex combination of physiological factors that include neurogenic vasodilation of intracranial/extracerebral blood vessels as well as sensitization of peripheral and central trigeminal nociceptive pathways (11, 12). Triptans relieve migraine symptoms via 3 possible mechanisms of action: (1) selective intracranial/extracerebral vasoconstriction; (2) inhibition of trigeminal sensory nerve activation and the release of vasoactive neuropeptides; (3) inhibition of neurones in the trigeminal-cervical complex in the brain stem and upper cervical column (13). It is believed that triptans stimulate 5-HT1B receptors, located predominantly on cranial blood vessels, and cause the vasoconstriction that counteracts the neurogenic vasodilation that may be responsible for the throbbing component of the headache. Triptans are also believed to stimulate 5-HT1D receptors located on peripheral and central trigeminal nociceptive nerve terminals and inhibit nociceptive transmission from pain-sensitive intracranial structures. Peripheral stimulation of 5-HT1D receptors blocks the release of vasoactive and pro-inflammatory neuropeptides which promote meningeal vasodilation and sterile neurogenic perivascular inflammation. In addition, triptans may also interact with 5-HT1B/1D/1F receptors on central neurones, although this remains to be defined (11, 13).
Possible mechanisms of CNS side-effects
Although the aetiology of CNS side-effects occurring with oral triptans is still unclear, the occurrence of these side-effects could be related to a number of factors that result in differences in the incidence between individual drugs. For instance, some triptans may exhibit a different binding affinity for 5-HT receptors or for other receptors not relevant to their antimigraine efficacy, but that result in adverse events. Alternatively, differences in the pharmacokinetic profiles of each drug, such as bioavailability, lipophilicity, or metabolism may be important. These possibilities will be reviewed in the following sections.
Receptor binding
All triptans are 5-HT1B/1D receptor agonists in the low nanomolar range, and show a similar lack of affinity for other serotonin receptors. Thus, the differences in side-effect profiles observed among triptans are unlikely to be caused by differences in binding characteristics at 5-HT receptors (11, 14). Another possibility is binding of triptans to other neurotransmitter receptors. Results from a number of in vitro receptor binding assays show no evidence of binding cross reactivity between triptans and other neurotransmitter receptors (15–18).
Pharmacokinetic differences
The pharmacokinetic characteristics of oral triptans are summarized in Tables 3 and 4. Sumatriptan has relatively low oral bioavailability and a short half-life. In contrast, newer triptans exhibit enhanced pharmacokinetic characteristics compared with sumatriptan, including rapid attainment of therapeutic plasma levels, greater oral bioavailability, and greater ability to cross the blood:brain barrier.
Pharmacokinetics of oral triptans

Tmax, time to maximum concentration; t1/2, biological half-life; Cmax, peak plasma concentration; MAO, monoamine oxidase; CYP450, cytochrome P450; CYP3A4, 3A4 isoform of cytochrome P450.
Female/male values;
(11)
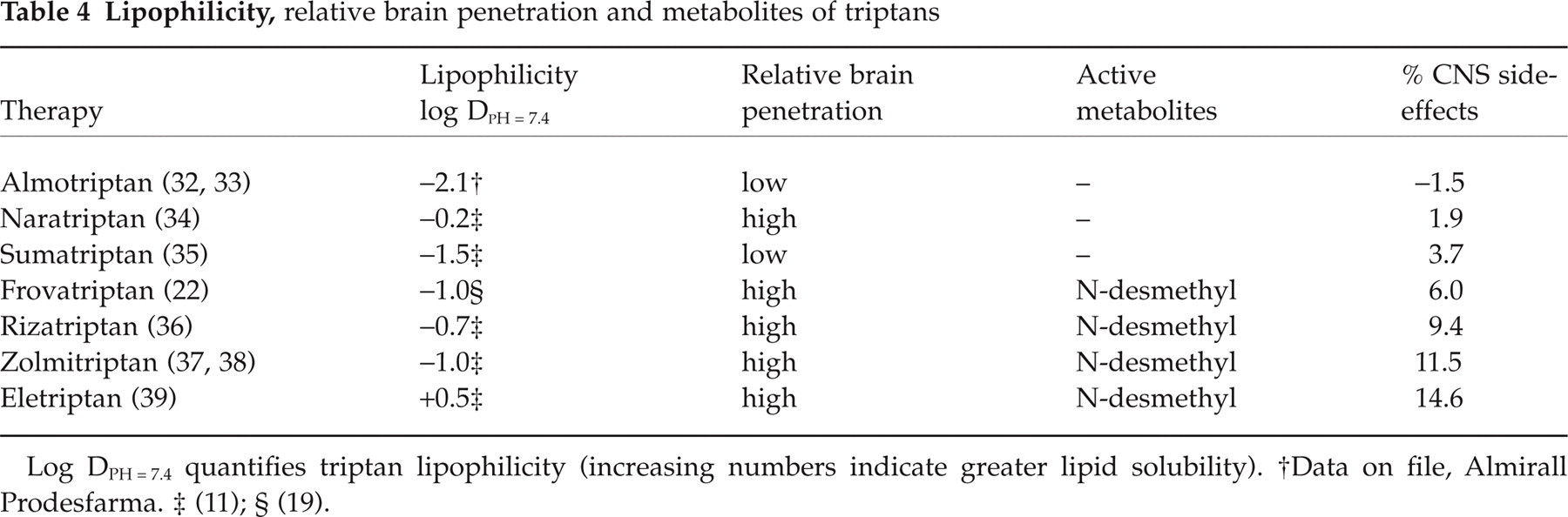
Log DPH = 7.4 quantifies triptan lipophilicity (increasing numbers indicate greater lipid solubility).
Data on file, Almirall Prodesfarma.
(11);
(19).
Figure 2(a–d) show triptans sorted in ascending order according to the parameter studied (i.e. Tmax, t1/2, Cmax, or bioavailability) and the corresponding incidence of placebo-subtracted CNS side-effects obtained from the meta-analysis. Little or no correlation exists between the incidence of CNS side-effects for a given triptan and the time it takes to reach maximum plasma concentration (correlation coefficient =−0.53, Fig. 2a), biological half-life (correlation coefficient =−0.06, Fig. 2b), maximum plasma concentration (correlation coefficient = 0.51, Fig. 2c) or bioavailability (correlation coefficient =−0.24, Fig. 2d). The low correlation coefficients suggest that pharmacokinetic parameters are weak predictors of CNS side-effects with triptans. For triptans with a similar Tmax and biological half-life (t1/2), the incidence of CNS side-effects may be quite different (Fig. 2a). For instance, almotriptan, which has the lowest incidence of CNS side-effects, has a Tmax and t1/2 similar to zolmitriptan (Tmax= 01.75 hours), a triptan known to have a higher incidence of CNS side-effects. Similarly, no association is observed between Cmax or bioavailability and the incidence of CNS side-effects (Fig. 2c–d). For instance, naratriptan has the highest bioavailability but has one of the lowest incidences of CNS side-effects, while eletriptan exhibits lower bioavailability and a higher incidence of CNS side-effects. The lack of association between Cmax and the incidence of CNS side-effects also is observed (Fig. 2c).

Correlation coefficients between placebo-subtracted CNS side-effects and pharmacokinetic parameters: (a) -0.59; (b) -0.06; (c) 0.52; (d) -0.24 (M), -0.28 (F).
A positive relationship is observed between the absolute dose of some triptans (the administered dose multiplied by its bioavailability) and the incidence of CNS side-effects within individual triptans (Fig. 3), which suggests that the incidence of CNS side-effects with some triptans is dose-dependent.

Correlation coefficients between CNS side-effects of oral triptans and absolute dose. (a) Within therapies; (b) Across therapies. Correlation coefficient 0.53. □ Sumatriptan; ○ Zolmitriptan; ▪ Rizatriptan; • Eletriptan.
Lipophilicity measures the solubility of a compound in lipid compared with water and can be used as an estimate of the ability of that compound to penetrate the blood:brain barrier. A positive correlation (correlation coefficient = 0.68) exists between the incidence of CNS side-effects and the lipophilicity of a triptan (Table 4 and Fig. 4). The correlation between CNS side-effects and lipophilicity presented here is in contrast to a recent report that found no correlation between side-effects and lipophilicity of triptans (19). However, that study was conducted before almotriptan, frovatriptan, and eletriptan were available commercially, and these 3 triptans were not included in the analysis.

Correlation coefficient between incident CNS side-effects and lipophilicity of each triptan. Correlation coefficients 0.68.
Although a positive correlation exists between lipophilicity coefficients for triptans and the incidence of CNS side-effects, this does not necessarily suggest cause and effect. For example, naratriptan 2.5 mg is associated with a low incidence of CNS side-effects even though it exhibits high lipophilicity. Interestingly, in a dose-finding study with naratriptan, the placebo-subtracted incidence of all adverse event rates was 2% (2.5 mg-dose), 9% (5 mg-dose), 14% (7.5 mg-dose) and 12% (10 mg-dose) (20). This suggests that at low dosages, lipophilicity is less important. Overall, the evidence from 7 triptans suggests that lipophilicity is correlated with the incidence of CNS side-effects. Thus, highly lipophilic triptans that penetrate the blood:brain barrier may be associated with higher levels of CNS side-effects, although a role for other pharmacokinetic or pharmacodynamic factors cannot be ruled out. For instance, some evidence suggests that the brain penetration of triptans is limited by the P-glycoprotein pump, which may offset the effects of high lipophilicity (21).
The presence of active metabolites may also contribute to the intensity and incidence of triptan-related CNS side-effects (Table 4). Almotriptan, naratriptan, and sumatriptan have no active metabolites and a relatively low incidence of CNS side-effects, while frovatriptan (22), rizatriptan (23), zolmitriptan (24, 25), and eletriptan (26) all have N-desmethyl active metabolites and a higher incidence of CNS side-effects. A literature search of in vitro studies, animal studies, and human studies revealed no published papers on the contribution of active metabolites of triptans to the side-effect profile of the parent compound. The N-desmethyl metabolite of zolmitriptan exhibits 5-HT1D agonist activity, has a half-life 50% longer than its parent compound, and may contribute to the overall efficacy of zolmitriptan (25). The N-desmethyl metabolite of frovatriptan exhibits one third the binding affinity of the parent compound and a half-life about 3 times longer(71–93 h) (22). The N-desmethyl metabolite of eletriptan exhibits a half-life and Tmax similar to those of the parent compound, but peak concentrations are approximately one third of the parent molecule (26). Although the precise contribution of active metabolites to treatment efficacy and incidence of CNS side-effects is unknown, their relationship to triptans with higher rates of CNS side-effects is noteworthy. It may be hypothesized that these active N-methyl metabolites bind to 5-HT receptors or other receptor sites that are associated with triptan-related CNS side-effects. Alternatively, the N-methyl metabolites may be more lipophilic than the parent compound and achieve plasma and brain concentrations that are more likely to produce CNS side-effects.
Clinical implications
Differences between triptans in the overall incidence of side-effects have been observed in head-to-head clinical trials and are confirmed by the results of a recent meta-analysis (7, 10). In particular, 10-fold differences can be encountered in the incidence of CNS side-effects between some of the triptans, with the highest incidence for eletriptan, rizatriptan, and zolmitriptan and the lowest incidence with almotriptan, sumatriptan, and naratriptan. Based on our review, we can hypothesize that the most likely explanation for these differences is the combination of active metabolites and high lipophilicity of individual triptans.
The experience of CNS side-effects may be a factor in the decision of the patient to initiate or avoid triptans for an acute migraine attack. In a recent survey study of 2444 severe migraine sufferers, 37% of patients who had or were currently using prescription-based acute antimigraine medications, reported often or always experiencing adverse effects (8). The most commonly reported adverse events in triptan-users were sleepiness/tiredness (47%), difficulty thinking (26%), racing heartbeat (24%) and dizziness (18%). Sleepiness and tiredness was the side-effects ranked by patients (20%) as the most troublesome adverse event. Two thirds of patients reported that they had avoided or delayed taking a current prescription medication due to concerns about adverse events. The impact of delaying or avoiding the use of acute medications was more intense pain(60%), the need to be lying down (59%), extended duration of the headache (59%), the need to cancel social activities (26%) and absence from or decreased performance at work or school (46%).
As these data illustrate, a delay in taking acute antimigraine medication can have a significant impact on outcome. Identifying a well-tolerated triptan therefore may encourage patients to initiate therapy early in the attack when symptoms are mild and more responsive to treatment. The importance of early administration of triptans has recently been highlighted in a number of studies. A greater proportion of patients experience complete pain relief when sumatriptan, rizatriptan, or almotriptan are taken early in a migraine attack, before pain intensity progressed to moderate or severe levels (27–30). Furthermore, recent data confirms the lack of effectiveness of triptans when they are administered after the development of cutaneous allodynia (31). Cutaneous allodynia develops in about 75% of migraine patients within 1 h after the onset of a migraine attack and reflects the development of central trigeminal sensitization (12).
In conclusion, the incidence of CNS side-effects differs between the triptans. The most likely explanations for the differences in CNS side-effects among triptans are the presence of active metabolites and high lipophilicity of the parent compound and active metabolites. Triptans with the lowest rates of CNS side-effects should be considered for those who must remain active following their migraine treatment and for those that have experienced unwanted CNS side-effects from other triptans.
Footnotes
Acknowledgements
The primary and corresponding author has consulted for and received honoraria and research grants from each of the pharmaceutical companies who market the triptans mentioned in this manuscript including: GlaxoSmithKline, Merck, Pfizer, Pharmacia, Ortho-McNeil, Almirall and AstraZeneca.
