Abstract
Cluster headache is characterized by typical autonomic dysfunctions including facial and intracranial vascular disturbances. Both the trigeminal and the cranial parasympathetic systems may be involved in mediating these dysfunctions. An experimental model was developed in the rat to measure changes in lacrimation and intracranial blood flow following noxious chemical stimulation of facial mucosa. Blood flow was monitored in arteries of the exposed cranial dura mater and the parietal cortex using laser Doppler flowmetry. Capsaicin (0.01-1 mM) applied to oral or nasal mucosa induced increases in dural and cortical blood flow and provoked lacrimation. These responses were blocked by systemic pre-administration of hexamethonium chloride (20 mg/kg). The evoked increases in dural blood flow were also abolished by topical pre-administration of atropine (1 mM) and [Lys1, Pro2,5, Arg3,4, Tyr6]-VIP (0.1 mM), a vasoactive intestinal polypeptide (VIP) antagonist, onto the exposed dura mater. We conclude that noxious stimulation of facial mucosa increases intracranial blood flow and lacrimation via a trigemino-parasympathetic reflex. The blood flow responses seem to be mediated by the release of acetylcholine and VIP within the meninges. Similar mechanisms may be involved in the pathogenesis of cluster headache.
Introduction
The irritant substance capsaicin applied to oral or nasal mucosa stimulates trigeminal neurones in rat (1, 2) and is known to cause a burning sensation that can be accompanied by rhinorrhoea, conjunctival congestion and lacrimation (3, 4). Similar symptoms are found in some forms of primary headaches, e.g. cluster headache and chronic paroxysmal hemicrania (5, 6). The autonomic disturbances such as nasal congestion, rhinorrhoea and lacrimation that typically accompany these headaches indicate that, in addition to the trigeminal system, the cranial parasympathetic system is activated. This assumption has been confirmed by measurements of neuropeptide levels in the jugular blood plasma of patients suffering from cluster headache and chronic paroxysmal hemicrania (7). During spontaneous attacks of these headaches as well as cluster attacks, provoked by infusion of nitroglycerin, elevated plasma concentrations of calcitonin gene-related peptide (CGRP) and vasoactive intestinal polypeptide (VIP) have been found (8–10). While CGRP is released from activated trigeminal afferents, VIP is probably derived from parasympathetic efferents (11). Both these neuropeptides are known to be potent dilatators of intracranial blood vessels (12, 13). The pathophysiological mechanisms that link the activity of the trigeminal and the autonomic systems during the development of cluster attacks and paroxysmal hemicrania are not known. It is very likely, however, that the neuropeptides mentioned above contribute substantially to the vascular changes observed in these diseases (7). Intracranial vasodilatation causing increases in cerebral and meningeal blood flow may thus be mediated by CGRP and VIP and possibly other vasodilatatory substances such as acetylcholine released from intracranial perivascular nerve fibres.
The present study was designed to elucidate further the mechanisms underlying the involvement of trigeminal and autonomic nerve fibres in the regulation of intracranial blood vessels. We developed an animal model in which changes in dural blood flow induced by noxious orofacial stimulation could be examined. Chemical irritation of rat mucosa using capsaicin was chosen as an appropriate noxious stimulus to activate trigeminal neurones (1), and laser Doppler flowmetry was used to record blood flow in arterial vessels of the cranial dura and pia mater overlying the parietal cortex (14). Parts of this study have been presented in preliminary form (15).
Materials and methods
Anaesthesia and general animal treatment
Experiments were performed on 23 male Wistar rats (300–400 g). The experimental protocol was approved by the Ethics Committee of the local government and was in accordance with the ethical guidelines for animal research of the International Association for the Study of Pain. Details of the surgical procedures have been described previously (16, 17). Briefly, rats were anaesthetized with an initial intraperitoneal dose of 120 mg/kg thiopentone (Trapanal, Byk Gulden, Germany). The femoral artery was cannulated for monitoring mean arterial blood pressure, and the femoral vein was cannulated for the administration of drugs. Following tracheotomy, most of the animals were allowed spontaneously to breathe room air enriched with oxygen. This was sufficient to maintain ventilation and to keep systemic blood pressure constant during the experiment. In six experiments, the animals were artificially ventilated with oxygen-enriched room air and paralysed with i.v. administration of gallamine triethiodide (40 mg/kg). Body temperature was maintained at physiological levels with a temperature-controlled heating plate. The animals were kept under deep anaesthesia throughout the experiment with supplementary doses of thiopentone (15 mg) administered as required to suppress nociceptive reflexes or blood pressure changes evoked by noxious pinch stimuli. The mean dose of thiopentone given during the whole experiment was 150 mg/kg. To terminate an experiment, a lethal dose of thiopentone was injected intravenously.
Head surgery and blood flow recording
The animals were placed in a stereotaxic headholder. The eyes were covered with a piece of cotton soaked with saline. The scalp was cut along the midline and the skin was separated from the left parietal bone. Using a drill and fine forceps, a cranial window of 4 × 6 mm was cut into the parietal bone to expose the underlying dura mater. To avoid thermal lesions of the dura during this phase of preparation, the bone was continuously cooled with cold saline. Saline was also used to cover the exposed dura during the remainder of the experiment. The pattern of blood vessels visible through a stereomicroscope in the cranial window was sketched (Fig. 1a). Arterial and venous vessels in the dura mater could clearly be differentiated from cortical arteries. Needle type probes of a laser Doppler flowmeter (Moore Instruments, Axminster, UK) were orientated toward branches of the middle meningeal artery or the middle cerebral artery at sites where these blood vessels were isolated from other vessels to minimize flow signals from the surrounding tissue (Fig. 1a). In this condition, blood flow signals were derived selectively from either the middle meningeal artery (dural blood flow) or the middle cerebral artery (cortical flow). Blood flow was recorded on-line at a sampling frequency of 10 Hz. The data were stored and processed with the DRTsoft program (Moore Instruments).
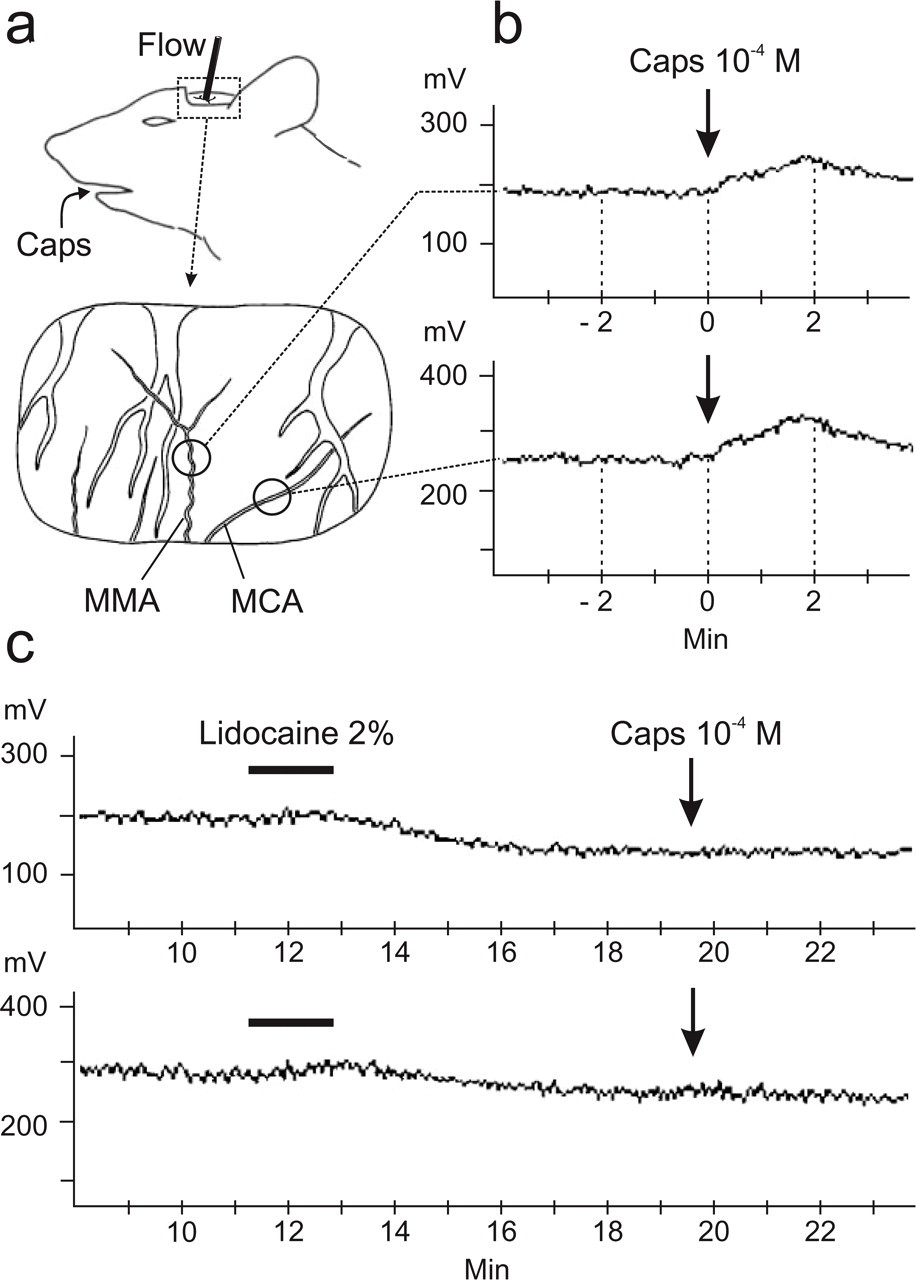
(a,b) Recording of blood flow (Flow) in branches of the middle meningeal artery (MMA) and the middle cerebral artery (MCA) visible in the cranial window (a) in a typical experiment; circles in (a) show recording sites. (b) Application of capsaicin (Caps) to oral mucosa causes increases in dural and cortical blood flow (measured in mV); mean flow in the 2-min interval after Caps is compared with mean flow in the 2-min interval before Caps. (c) After pre-administration of lidocaine onto the dura in the cranial window, Caps failed to increase dural and cortical flow [same experiment as in (b)].
Administration of substances
Capsaicin (Sigma-Aldrich, Taufkirchen, Germany) was used to stimulate oral or nasal mucosa. To stimulate the oral mucosa, a thin flexible plastic tube, filled with 0.2 ml saline, was positioned so that its opening was between the upper surface of the tongue and the hard palate. Over a 30-s period, 0.25 ml of either capsaicin, vehicle or saline solution was applied, followed by 0.25 ml saline to flush the tube. To stimulate the nasal mucosa, 50 µl of capsaicin solution were applied over a 15-s period to a site 2 mm into the left nostril using an Eppendorf pipette. In most experiments two or three progressively increasing doses of capsaicin were applied, at intervals of 10 min, to the oral or nasal cavity, then the cavity was thoroughly washed with saline. After a waiting period of at least 30 min, provided that the basal blood flow had returned to the prestimulus level, one of several substances (see below) was applied onto the exposed dura or injected intravenously before the same doses of capsaicin were re-tested. In other experiments one of the pharmacological substances was initially applied to the dura, followed by one or two subsequent exposures to capsaicin. At the end of each experiment, a final capsaicin stimulus confirmed that the blood flow response was in the same range as at the beginning of the experiment. Capsaicin was stored in a stock solution of 10−2 M in ethanol and dissolved in saline to concentrations of 10−3−10−5 M. Vehicle solutions with 1% and 10% ethanol in saline applied to the oral or nasal cavity were found to have no effect on meningeal blood flow. Therefore, in most cases saline was used for control experiments. For topical application of substances to the dura mater, the cranial window was filled with 50 µl of solution. A dam of Vaseline along the lateral, rostral and caudal rim of the cranial window prevented the solutions from draining away. Lidocaine (2% solution; Braun, Melsungen, Germany), the VIP-receptor antagonist [Lys1, Pro2,5, Arg3,4, Tyr6]-VIP (10−4 M; Sigma) or atropine sulphate (10−3 M; Sigma), dissolved in saline, were topically applied to the dura. Hexamethonium chloride (20 mg/kg) was slowly injected through the femoral vein cannula.
Data collection and processing
Data were collected only if the basal flow was stable over a period of at least 15 min. Changes in dural or cortical blood flow induced by application of saline or capsaicin solutions to oral or nasal mucosa were assessed as the difference in mean flow during the 2-min period immediately before and immediately after chemical application (Fig. 1b). The difference values were normalized to the mean flow in the 2-min period before substance application (100%). A one-way analysis of variance (ANOVA) extended by the post hoc least significance difference (LSD) test was used to compare the relative changes in flow caused by capsaicin at different concentrations with the effect of saline. Similarly, changes in blood flow, caused by topical administration of substances to the dura or i.v. injection, were assessed by comparing the normalized changes in flow. Significance was accepted at the 5% level for all statistical tests. All flow values are listed as mean ± SEM.
Lacrimation measurements
The cotton pad covering the left eye was removed. Immediately before application of saline or capsaicin to the oral or nasal mucosa, a small piece of filter paper was attached to the medial angle of the eye. Two minutes later the filter paper was removed and weighed. The increase in weight was used as a measure of lacrimation. Differences in lacrimation were assessed by ANOVA with repeated measurements extended by the LSD test.
Results
Increases in intracranial blood flow induced by capsaicin application to facial mucosa
Before stimulation, intracranial blood flow recorded from the middle meningeal artery (dural blood flow) and from branches of the middle cerebral artery (cortical blood flow) was stable. Capsaicin solutions applied to either oral or nasal mucosa caused a concentration-dependent increase in dural and cortical blood flow (Table 1, Fig. 1b). These blood flow responses started immediately, usually before the capsaicin application was complete, and reached a maximum within the 2-min period following the onset of stimulation.
Summary of blood flow changes (%) to capsaicin stimulation of facial mucosa
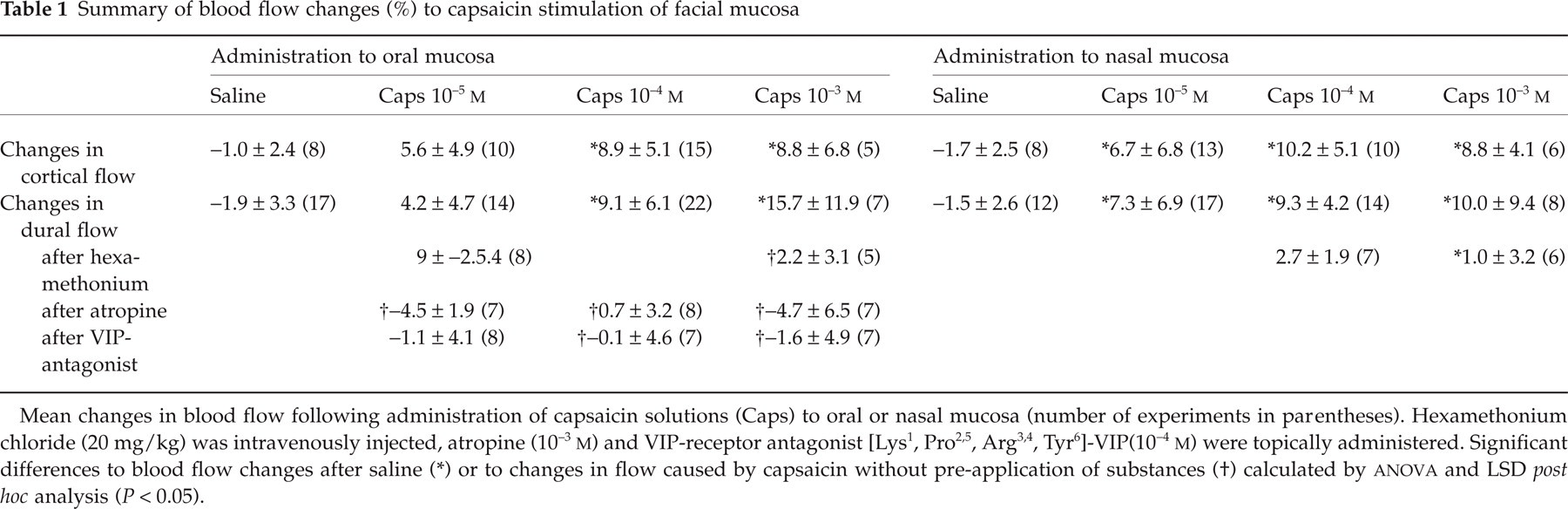
Mean changes in blood flow following administration of capsaicin solutions (Caps) to oral or nasal mucosa (number of experiments in parentheses). Hexamethonium chloride (20 mg/kg) was intravenously injected, atropine (10– 3 M) and VIP-receptor antagonist [Lys1, Pro2,5, Arg3,4 , Tyr 6]-VIP(10 −4 M) were topically administered. Significant differences to blood flow changes after saline
(∗) or to changes in flow caused by capsaicin without pre-application of substances
(†) calculated by ANOVA and LSD post hoc analysis (P < 0.05).
The normalized increases in dural blood flow were significant at capsaicin concentrations of 10−4 and 10−3 M for oral stimulation and at concentrations of ≥ 10−5 M for nasal stimulation, although there was considerable variation in responses between individual experiments (Table 1, Fig. 2). In some experiments the highest capsaicin concentration(10−3 M) caused a short-lasting increase in systemic blood pressure. These experiments were excluded from further analysis.

Changes in dural blood flow caused by intraoral application of capsaicin (Caps) at different concentrations (▪); ∗significant difference with respect to saline application. After pre-administration of a vasoactive intestinal polypeptide antagonist (VIP-A, 10−4 M) onto the dura in the cranial window, Caps failed to increase dural blood flow ( ); ∗significant difference with respect to response to Caps without VIP-A.
); ∗significant difference with respect to response to Caps without VIP-A.
Significant increases in cortical blood flow were induced by application of capsaicin solution to the facial mucosa at concentrations of 10−4 and 10−3 M for oral stimulation or ≥ 10−5 M for nasal stimulation (Table 1). At both stimulation sites capsaicin concentrations of 10−4 M produced maximal increases in flow.
In two additional experiments, lidocaine (2%) topically applied to the exposed cranial dura mater prevented increases in dural and cortical blood flow after capsaicin application (10−4 M) to the nasal mucosa. Lidocaine itself caused a small decrease in basal blood flow (Fig. 1c).
Effects of hexamethonium, atropine and VIP-antagonists on dural blood flow increases induced by capsaicin application to the facial mucosa
Intravenous injection of hexamethonium chloride (20 mg/kg) caused an immediate and significant decrease in basal dural blood flow by 22 ± 6%, which lasted up to 45 min and was accompanied by a decrease in systemic blood pressure by about 20%. Following topical hexamethonium chloride, capsaicin (10−5−10−3 M) did not cause any significant change in dural blood flow when applied to either the oral or the nasal mucosa (Table 1).
Atropine solution (10−3 M, 50 µl) topically applied to the exposed cranial dura caused a gradual but non-significant decrease in basal dural flow of 5.6 ± 7.7% in the 2-min period following application without changing the systemic pressure. After atropine, capsaicin (10−5−10−3 M) applied to the oral mucosa did not cause any increase in dural blood flow (Table 1).
The VIP-receptor antagonist [Lys1, Pro2,5, Arg3,4, Tyr6]-VIP (10−4 M) topically applied to the dura did not change the basal dural blood flow (−0.9 ± 11.1%). Following application of the VIP-antagonist, capsaicin (10−5−10−3 M) applied to oral mucosa did not cause any change in dural blood flow (Table 1, Fig. 2).
Lacrimation induced by capsaicin application to the oral mucosa and the effect of hexamethonium
Capsaicin (10−4 M) applied to either the oral or the nasal mucosa caused a significant increase in lacrimation. Saline application was ineffective. Following i.v. injection of hexamethonium chloride (20 mg/kg), capsaicin application to the nasal mucosa was no longer effective in stimulating lacrimation (Fig. 3).

Lacrimation caused by application of capsaicin (Caps) to the oral or the nasal mucosa (▪); ∗significant difference with respect to saline application. Pre-administration of hexamethonium chloride (20 mg/kg i.v.) abolished the lacrimation response to intranasal capsaicin stimulation ( , ∗).
, ∗).
Discussion
In this study the noxious stimulant capsaicin was applied to the rat oral or nasal cavity. Such a stimulus is considered to be similar to the noxious conditions that cause both mucosal irritation and facial or head pain in man when the upper airways are inflamed (18, 19) or when irritant substances such as high concentrations of capsaicin are ingested or inhaled (3, 4). Capsaicin activates nociceptive afferents through vanilloid (VR-1) receptors (20). Compared with experiments in other tissues (21), relatively high concentrations of capsaicin were used in the present study. This is not unusual, however, since neurones in the rat spinal trigeminal nucleus with intraoral input have been shown to be readily activated when capsaicin is applied to the tongue at concentrations of at least 10−4 M (1). From neurophysiological and psychophysiological data it is known that repetitive application of capsaicin to oral mucosa causes sensitization as well as desensitization depending on the dose and interval of application (22–25). By using application intervals of at least 10 min, stable blood flow responses to capsaicin were achieved in the present study, suggesting that sensitizing and desensitizing processes in the trigeminal system may have been balanced.
Capsaicin applied to the rat oral or nasal cavity increased the blood flow in dural arteries (dural flow) and in pial arteries of the parietal cortex (cortical flow) as measured by laser Doppler flowmetry. We concluded that this response was mediated by nerve fibres innervating the meninges, because it could be blocked by the local anaesthetic lidocaine topically applied to the exposed dura mater. The cranial dural and pial arteries are innervated by thin sensory nerve fibres, which originate in the trigeminal ganglion, as well as postganglionic sympathetic and parasympathetic fibres (26–30). We supposed that the evoked increases in flow were mediated through vasodilatatory substances released from these nerve fibres. Hexamethonium, a blocker of synaptic transmission in autonomic ganglia, reduced the increases in flow, suggesting that this response was dependent on postganglionic autonomic nerve fibres, although it cannot be excluded that the decrease in blood pressure caused by hexamethonium contributed to this effect. Since it was expected that stimulation of sympathetic efferents would cause vasoconstriction and thereby decrease intracranial blood flow, we supposed that parasympathetic rather than sympathetic fibres were involved in the blood flow response induced by capsaicin. This hypothesis was confirmed by the finding that local administration of atropine, an antagonist of acetylcholine at muscarinic receptors, onto the exposed dura also blocked the dural blood flow response very effectively. It seems likely that basal activity in parasympathetic fibres causes a slow continuous release of acetylcholine and that this contributes to the maintenance of basal blood flow in the meninges, since systemic hexamethonium as well as local atropine application reduced the basal flow. Additional evidence for activation of the parasympathetic system following noxious stimulation of the mucosa was provided by the lacrimation response which was also blocked by hexamethonium. Finally, we showed that topical administration of [Lys1, Pro2,5, Arg3,4, Tyr6]-VIP, a competitive VIP-receptor antagonist, onto the exposed dura abolished the increase in dural flow evoked by capsaicin stimulation of the oral mucosa.
Results contradictory to the present study were reported by Peitl et al. (31). This group stimulated the rat trigeminal ganglion with electrical pulses to induce increases in meningeal blood flow, which could have resulted from either antidromic conduction in trigeminal afferents supplying the meninges or activation of a parasympathetic pathway. Following systemic administration of hexamethonium (10 mg/kg), Peitl et al. (31) found no effect on the increase in blood flow produced with electrical stimulation, although blood pressure decreased in a manner similar to that observed in the present study. Moreover, there was no effect on the stimulated increase in blood flow after local administration of the VIP antagonist (p-chloro-D-Phe6-Leu7)-VIP to the dura. Thus it seems likely that the release of CGRP from antidromically invaded dural sensory fibres overrides the comparably small effect produced by the release of mediators from dural parasympathetic fibres, both of which should be activated with electrical stimulation of the ganglion.
Taken together, our experiments suggest that the capsaicin-evoked increases in meningeal blood flow are mediated by acetylcholine and VIP released from parasympathetic efferents that innervate dural (and probably also cortical) blood vessels. Preliminary examinations (not published) show a sparse and rather variable innervation of the rat dura mater with VIP immunoreactive nerve fibres. At high capsaicin concentrations sympathetic fibres may be additionally activated. This may explain why the blood flow response in pial vessels was not further increased in the presence of capsaicin concentrations > 10−4 M. It may be interesting to compare vascular responses of dural and pial blood vessels in future experiments.
Results similar to those described in the present study have previously been reported for stimulation of cat facial skin by Karita and Izumi (32–34). Heat, capsaicin, electrical and noxious mechanical stimuli applied to orofacial areas innervated by the second and third trigeminal branch as well as electrical stimulation of the facial and glossopharyngeal nerves (35) caused increases in blood flow in the lacrimal and submandibular glands, palate and lower lip, which could be blocked by systemic pre-administration of hexamethonium. The authors interpreted this response as a somato-autonomic reflex and assumed a vasodilator centre connecting spinal trigeminal nucleus with parasympathetic nuclei in the brain stem. The same group recently presented more evidence for this hypothesis showing that microinjection of lidocaine into the spinal trigeminal nucleus reduced the parasympathetic reflex vasodilatation in the cat lower lip evoked by lingual nerve stimulation (36). Parasympathetic vasodilatation in facial tissues is a well-known experimental and clinical phenomenon [see (37) for a review], and intracranial vasodilatation may occur in parallel under relevant experimental or pathophysiological conditions. The parasympathetic activity is mediated via presynaptic parasympathetic fibres in the facial nerve and transmitted through the otic and sphenopalatine ganglia to orofacial skin and intracranial structures (28). This was experimentally verified by Suzuki et al. (38), who stimulated postganglionic fibres from the rat sphenopalatine ganglion directly with electrical pulses and measured increases in cortical blood flow by laser Doppler flowmetry. Using the same stimulation conditions, Delepine and Aubineau (39) induced another vascular response, namely plasma extravasation in rat dura mater, which was abolished by systemic administration of atropine. The preganglionic fibres to the parasympathetic ganglia subserving cerebrovasodilator functions may arise from the greater petrosal nerve cell group, a subgroup of the salivatory nucleus, in the medullary brain stem (40). Microinjection of glutamate into this region in the cat reduced cerebrovascular resistance and this effect was abolished by bilateral denervation of the parasympathetic postganglionic fibres originating in the sphenopalatine ganglion (40).
There is little doubt that in cluster headache, and other rare forms of headache associated with autonomic dysfunctions of facial areas, both the trigeminal and the parasympathetic system are involved (41), possibly coupled by similar reflex mechanisms as shown in the present and other experimental studies in animals. In a very recent study, subcutaneous capsaicin injection into the forehead of healthy volunteers was reported to cause the same autonomic symptoms as those observed in cluster headache (42). Plasma levels of the sensory neuropeptide CGRP (released from trigeminal afferents) and VIP (probably released from parasympathetic efferents) are increased in venous blood samples from the jugular taken during attacks of cluster headache and paroxysmal hemicrania (9, 10). The presumed co-release of these potent vasodilators together with other vasodilatatory neuropeptides and acetylcholine in the meninges may cause massive vasodilatation. Vasodilatation may sensitize perivascular nociceptors to mechanical stimuli such as arterial pulsation, particularly when it is facilitated by an inflammatory component like plasma extravasation (39). It is still unclear, however, whether the trigeminal activation precedes the parasympathetic activation in the pathogenesis of cluster headache and whether their coactivation is causally linked. Further experimental and clinical studies are necessary to answer these questions.
Footnotes
Acknowledgements
The authors thank Mrs J. Schramm and Mrs B. Vogler for their excellent technical assistance and Dr R. Carr for valuable comments on the manuscript. The study was supported by grants from the Wilhelm Sander-Stiftung and the DFG (SFB 353, B3).
