Abstract
We hypothesized that intravenous infusion of the parasympathetic transmitter, vasoactive intestinal peptide (VIP), might induce migraine attacks in migraineurs. Twelve patients with migraine without aura were allocated to receive 8 pmol kg-1 min-1 VIP or placebo in a randomized, double-blind crossover study. Headache was scored on a verbal rating scale (VRS), mean blood flow velocity in the middle cerebral artery (V mean mca) was measured by transcranial Doppler ultrasonography, and diameter of the superficial temporal artery (STA) by high-frequency ultrasound. None of the subjects reported a migraine attack after VIP infusion. VIP induced a mild immediate headache (maximum 2 on VRS) compared with placebo (P = 0.005). Three patients reported delayed headache (3-11 h after infusion) after VIP and two after placebo (P = 0.89). V mean mca decreased (16.3 ± 5.9%) and diameter of STA increased significantly after VIP (45.9 ± 13.9%). VIP mediates a marked dilation of cranial arteries, but does not trigger migraine attacks in migraineurs. These data provide further evidence against a purely vascular origin of migraine.
Introduction
Migraine pain is probably mediated via nociceptors around cephalic vessels (1–5). Neurotransmitters or neuromodulators such as nitric oxide (NO) and calcitonin gene-related peptide (CGRP) are involved in the nociception during migraine attacks (6–11). Despite increasing knowledge of migraine pathophysiology (12), the mechanisms responsible for the induction of migraine attacks are mainly unknown. Some premonitory symptoms, such as yawning and increased frequency of urination (13), and autonomic symptoms such as eyelid oedema, lacrimation, conjunctival injection and nasal congestion (14, 15), suggest that the parasympathetic nervous system (PSNS) may be involved in the initiation and maintenance of migraine attacks (16). Furthermore, there is evidence that, NO, known to trigger migraine attacks (7, 17), plays a crucial role in mediating the cerebral vasodilation followed by activation of the cranial PSNS (18–20). Vasoactive intestinal peptide (VIP) is a neurotransmitter in cerebral parasympathetic perivascular nerve fibres and cranial parasympathetic ganglia, both in human and animals (21–26). Elevated VIP plasma levels in the cranial circulation have been reported in a subgroup of migraineurs with pronounced autonomic symptoms (27). Recently, we reported that intravenous (i.v.) infusion of VIP induced a very mild, short-lasting headache in healthy subjects in spite of marked and prolonged dilation of superficial temporal artery (28). In patients with migraine we hypothesize that systemic administration of VIP may cause a more pronounced headache or migraine associated with marked vasodilation. To test this hypothesis, we conducted a double-blind, placebo-controlled crossover study in patients with migraine without aura and studied the effect of VIP on headache and haemodynamic variables.
Design and methods
Patients
We included 14 patients with migraine without aura [12 woman, two men, mean age 31 years (range 22–40)] according to International Headache Society (IHS) criteria (2nd edn, 2004) (29). Inclusion criteria were: frequency of migraine attacks between one and six attacks within 8 weeks, age between 18 and 40 years, weight 50–100 kg and secure prevention for fertile women (oral contraception, intrauterine device). Exclusion criteria were: any other type of headache (including >2 days of tension-type headache per month), headache on the study day or <24 h before, daily intake of medication apart from oral contraception, pregnant or nursing women, history or clinical signs of serious somatic and psychiatric diseases. The study was approved by the Ethics Committee of Copenhagen County (KA04119) and was conducted according to the Helsinki declaration 1964, as revised in Edinburgh in 2000. All patients were recruited from the Danish Headache Centre and gave informed consent. The study was registered at ClinicalTrials.gov (NCT00272896).
Experimental design
In a double-blind, placebo-controlled crossover study, the patients were randomly allocated to receive 8 pmol kg−1 min−1 VIP (Calbiochem, San Diego, CA, USA) or placebo (isotonic saline) over 25 min on 2 days separated by at least 1 week. The processing of dilution, sterile filtering and balanced randomization was done by the central pharmacy of Herlev Hospital, Denmark. We obtained the VIP and placebo as frozen dilutions for each patient in a separate box labelled with treatment of day 1 and day 2. The randomization code was delivered in sealed envelopes.
All patients reported to the laboratory at 08.00 h, headache free. The consumption of coffee, tea, cocoa and other methylxanthine-containing beverages and foods was not allowed from at least 8 h before the start of the study. An intake of medication (besides oral contraception) was accepted only after a period of four times the plasma half-life of the drug. The patients were placed in a supine position and supplied with venous catheters in antecubital veins (Venflon, 17GA; Becton Dickinson, Helsingborg, Sweden), ECG, pulsoxymeter and a cuff for blood pressure measurements. During the study, patients were kept in quiet surroundings. The baseline measurements were conducted after 30 min of rest and the whole period of measurements lasted 120 min (from baseline t0 to t120). The following variables were recorded: headache score, mean velocity of blood flow in the middle cerebral artery (V mean MCA), diameter of the superficial temporal artery diameter (STA), end-tidal partial pressure of CO2 (Pet CO2), vital signs and adverse effects every 10 min after start of infusion (except one additional measurement at t25). One of the antecubital catheters was used for the VIP or placebo infusion t0–t25 (time- and volume-controlled infusion pump; Braun Perfusor, Melsungen, Germany). Blood samples for the determination of the plasma VIP levels were collected at baseline and after 10, 20, 25, 30, 60 and 120 min from the catheter on the opposite side. The patients were discharged from the laboratory after end of experiment and asked to complete a headache diary every hour until 12 h after discharge. The headache diary included headache characteristics and accompanying symptoms according to the IHS (29), any rescue medication taken and adverse effects (30). The patients were allowed to take rescue medication of their own choice at any time.
Headache recordings
Headache intensity was scored on a verbal rating scale (VRS) from 0 to 10 [0 = no headache, 1 = a very mild headache (including a feeling of pressing or throbbing), 5 = moderate headache, 10 = worst imaginable headache]. Headache characteristics, location and accompanying symptoms were recorded throughout the examination.
Middle cerebral artery blood flow velocity
Mean velocity of blood flow in the middle cerebral artery (V
mean MCA) was recorded bilaterally by transcranial Doppler (TCD) with hand-held 2-MHz probes (Multidop X4; DWL, Sipplingen, Germany). Fixed probes were not used since they may cause discomfort and even headache (31)
Diameter of the superficial temporal artery
Diameter of the frontal branch of the STA was measured by a high-resolution ultrasonography unit (20 MHz, bandwidth 15 MHz; Dermascan C; Cortex Technology, Hadsund, Denmark) (33, 34). During measuring the head was turned to one side. Marks were drawn on the skin to ensure that the repeated measurements were performed in the same place and to ensure reproducibility in measurements from day to day. For each time point the average of four serial measurements of STA diameter was calculated.
Vital signs
Heart rate (HR) and blood pressure (MAP) were measured every 10 min using an auto-inflatable cuff (ProPac Encore®; Welch Allyn Protocol). ECG was monitored on an LCD screen and recorded on paper every 10 min. In addition, adverse events were recorded, including flushing, palpitation and heat sensation.
Plasma concentration of VIP
Blood samples for VIP measurement were collected at baseline and 10, 20, 25, 30, 60 and 120 min after start of infusion in ice-chilled tubes containing 50 IU of heparin and 500 kIU aprotinin (Trasylol, Bayer, Germany) per ml blood. Plasma was separated by centrifugation (1000 g) at 4°C within 30 min and immediately frozen at −20°C. The VIP radioimmunoassay was performed as previously described using antiserum 5603–6 at a final titre of 1.2 × 106 in a total volume of 0.8 ml/tube (35, 36). This antiserum recognizes the mid- and C-terminal regions of the VIP molecule (sequence 11–24) and displays no cross-reactivity with other known gastrointestinal peptides or neuropeptides. The label has a specific radioactivity of 0.92 nCi/fmol (∼34 Bq/fmol). The IC50 value (the concentration of VIP giving 50% displacement of label) was 24 pmol/l and the assay could detect changes of 3 pmol/l with 95% confidence.
Data analysis and statistics
All values are presented as mean ± SD, except headache scores, which are presented as median and quartiles in parentheses.
Calculation of sample size was based on the detection of a difference between VIP and placebo in the V mean MCA during infusion (0–30 min) and postinfusion (30–120 min) periods at 5% significance with 90% power. We assumed that the analyses of variables would show 10% interindividual variation as reported previously (28). A 10% difference between two experimental conditions was taken to be clinically significant. We estimated that 11 subjects should be included for within-person study (37), but we increased the sample size to 13 subjects, because this is statistically more desirable.
We calculated area under the curve (AUC) according to the trapezium rule (38) to obtain a summary measure and to analyse the differences in response (V mean MCA, headache score, diameter of STA, mean blood pressure, HR, PEtCO2, VIPplasma) between VIP and placebo. If there were no significant side differences of the Doppler values, the average of left and right side was used for the calculation.
The primary end-points were differences in AUC for headache score (AUCheadache 0−30min and AUCdelayed headache), V mean MCA (AUCVmeanMCA), STA diameter (AUCSTA), end-tidal CO2 (AUCPetCO2) and AUCplasmaVIP between two trial days in the period 0–30 min. Additional explorative end-points were differences in AUCheadache score, AUCVmean MCA, AUCSTA, and AUCPetCO2 between the two experimental conditions in the period 30–120 min. The secondary end-points were differences in AUCheart rate and AUCMAP between groups in the period 0–30 min. Additional explorative end-points were differences in AUCheart rate and AUCMAP between VIP and placebo days in the period 30–120 min and differences in peak response (HR and MAP) at time of maximum plasma concentration of VIP.
Analysis was performed with a paired, two-way t-test, except headache scores, which were tested using Wilcoxon's signed rank test. Baseline was defined as t0 before the start of infusion of each dose. Continuous variables were analysed for changes over time by univariate analysis of variance (with the fixed factors volunteer and time), if there was statistical difference in AUC. To reduce mass significance the following time points were selected for analysis: t0, t20, t60 and t120. If overall differences were found, Dunnett's test was applied to characterize which time points were different from baseline. The incidence of headache (including migraine) and other adverse events between the groups was compared with McNemar's test.
All analyses were performed with SPSS for Windows 12.0 (SPSS Inc.,Chicago, IL, USA). Five percent (P < 0.05) was chosen as the level of significance.
Results
There were no differences in baseline recordings of any variables between the two study days (Table 1). Thirteen patients completed the study on both trial days. One patient participated on VIP day and did not report on a placebo day for personal reasons. In one patient, data were lost on the second trial day (VIP day) and the patient accepted re-entering the study and thereupon was allocated to infusion of VIP and placebo. The data of one patient were excluded from analysis because of marked clinical hyperventilation during infusion of VIP.
Baseline values [mean (± SD)] in 12 migraineurs on two trial days before administration of vasoactive peptide (VIP)/placebo
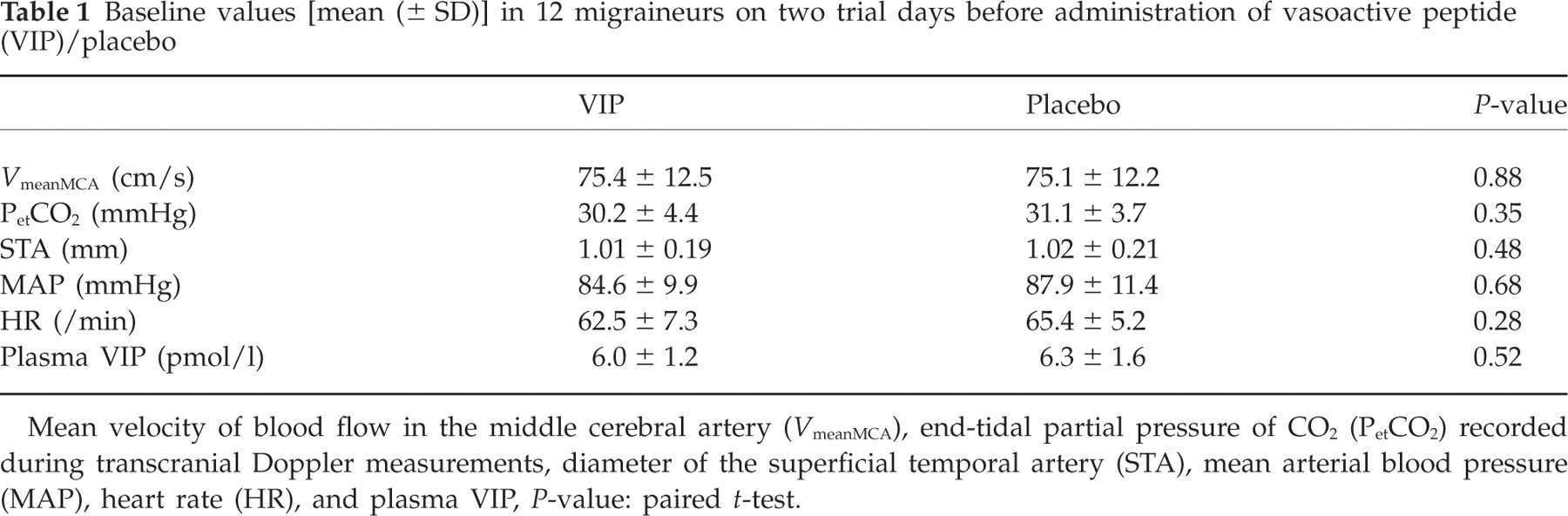
Mean velocity of blood flow in the middle cerebral artery (V meanMCA), end-tidal partial pressure of CO2 (PetCO2) recorded during transcranial Doppler measurements, diameter of the superficial temporal artery (STA), mean arterial blood pressure (MAP), heart rate (HR), and plasma VIP, P-value: paired t-test.
Headache
During the immediate phase (0–30 min), 10 patients reported a very mild headache on the VIP day, whereas none of the patients reported headache on the placebo day (Table 2). The AUCheadache 0−30min on the VIP day, 23 (6–25), was significantly larger than on the placebo day, 5 (0–40) (P = 0.005) (Fig. 1). During the postinfusion period (30–120 min), seven patients reported headache on the VIP day and four on the placebo day. There was no difference in the AUC30−120min recorded during the postinfusion period between VIP, 0 (0–0) and placebo days (0–9) (P = 0.44).
Clinical characteristics of 12 migraineurs

a, Headache VIP day, time 0–120 min; b, delayed headache on VIP day, time 3–11 h; c, headache on placebo day, time 0–120 min; d, delayed headache on placebo day, time 3–11 h; photo., photophobia; phono., phonophobia.

Individual and median headache scores on a verbal rating scale (VRS) in the immediate phase (0–30 min). There were significantly higher pain responses after VIP infusion (▪) compared to placebo (□) (P = 0.005). Thick lines show median pain scores.
Three patients reported delayed headache on the VIP day (in the period 3–11 h after start of infusion) and two patients on the placebo day (in the period 3–11 h after start of infusion) (Table 2). No difference was found in the AUCdelayed headache recorded from 120 min to 12 h after start of infusion between VIP, 0 (0–3) and placebo days, 0 (0–0) (P = 0.89).
Eleven subjects reported no migraine attack after either VIP infusion or placebo, and one subject reported migraine after placebo infusion but not after VIP. McNemar's test gives P = 1.0, but is effectively meaningless due to the small cell counts. However, calculating the exact confidence interval (CI) of the binomial distribution (0 out of 12), it is noteworthy that the upper limit of a 95% CI for the frequency of migraine after VIP infusion in our study is 26.5%, which is considerably less than the 80% seen when using glyceryl trinitrate infusion (7).
Middle cerebral artery blood flow velocity
During the immediate phase the AUC0−30min for V mean MCA decreased significantly on the VIP day compared with the placebo day (P = 0.001) (Fig. 2). During the postinfusion period (AUC30−120min), there was no difference between VIP and placebo days (P = 0.86). The univariate analysis of variance showed significant changes over time on the VIP day (P < 0.001), but not on the placebo day (P = 0.1). There were significant differences at t20 (P < 0.001) and t120 (P = 0.002) compared with baseline (t0) on the VIP day. Peak decrease in V mean MCA occurred 20 min after start of VIP infusion. The mean decrease in V mean MCA was 16.3 ± 5.9% on the VIP day and 0.01 ± 4.3% on the placebo day compared with baseline (Fig. 2). The mean difference in response between VIP and placebo at t20 was −16.3 ± 3.6% (95% CI −18.6, −14). There were no differences in the PetCO2 (recorded during TCD scan) between VIP and placebo days, during either the immediate (AUC0−30min, P = 0.08) or postinfusion phase (AUC30−120min, P = 0.35).

Individual and mean blood flow velocities in the middle cerebral artery (Vmean MCA) assessed by transcranial Doppler ultrasonography. Infusion of vasoactive intestinal peptide (VIP) (▪) resulted in significant decrease in Vmean MCA during immediate phase (0–30 min) compared to placebo (□) (P < 0.001). Thick lines show mean values.
Superficial temporal artery
During the immediate phase (AUC0−30min) and postinfusion period (AUC30−120min), the diameter of STA increased significantly on the VIP day compared with the placebo day (P < 0.001) (Fig. 3), and
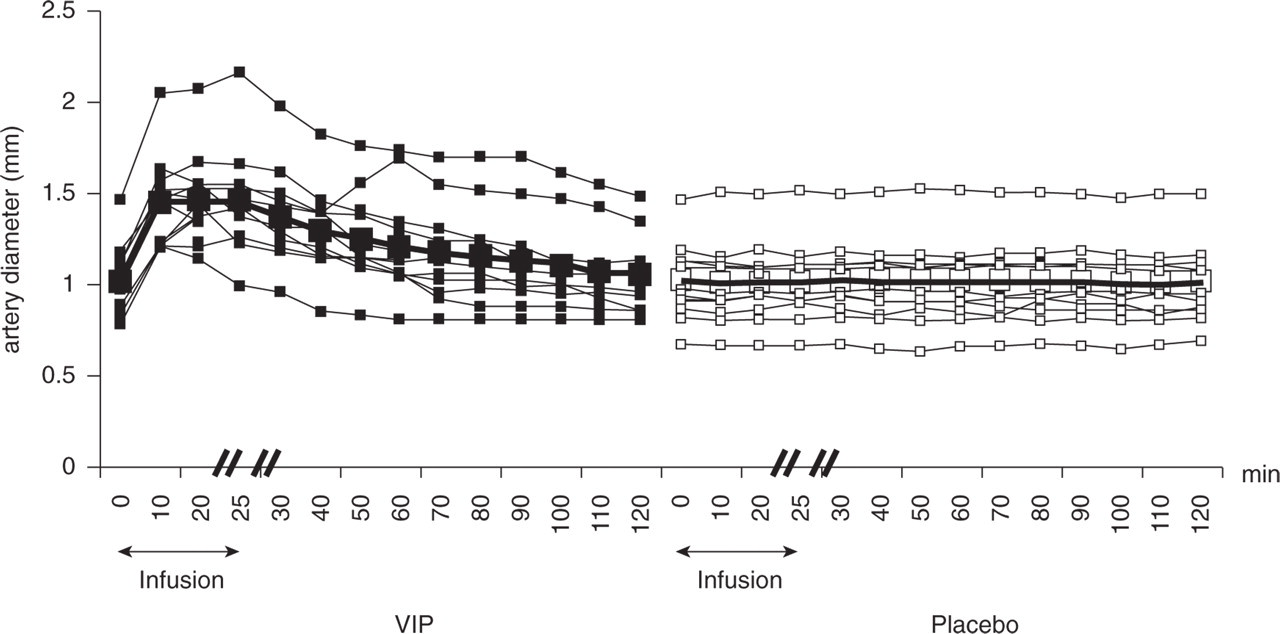
Individual and mean diameter of the superficial temporal artery (STA) assessed by a high-resolution ultrasonography. During the immediate phase (0–30 min), the diameter of the STA increased significantly after VIP (▪) compared to placebo (□) (P < 0.001). Thick lines show mean values.

Mean per cent change (%) from baseline in Vmean MCA (▪), STA diameter (□), heart rate (▴), and mean arterial blood pressure (
Mean arterial blood pressure and heart rate
No difference was found in AUCMAP between VIP and placebo days during either the immediate phase (P = 0.09) or the postinfusion phase (P = 0.66). The AUCheart rate was significantly larger on the VIP day than on the placebo day (P < 0.001). There was no difference in HR in the postinfusion period between two trial days (P = 0.3).
Plasma VIP
The AUCplasmaVIP was significantly larger on the VIP day than on the placebo day in the period 0–30 min (P < 0.001), but not in the postinfusion period 30–120 min (P = 0.07) (Fig. 5).

Individual and mean plasma concentration of vasoactive intestinal peptide (VIP) after infusion of VIP (▪) and placebo (□). Infusion of VIP resulted in a significant increase in plasma VIP compared to placebo (P < 0.001). Peak plasma concentration of VIP was measured at t20 min and was 108.4 pmol/l ± 21.5. Thick lines show mean concentrations.
Adverse events
The patients reported palpitations (n = 11) and heat sensation (n = 12), and flushing (n = 12) on a VIP day was recorded (Table 3). Two patients reported heat sensation on the placebo day. One patient became anxious because of the palpitations and a strong heat sensation during the VIP infusion. The patient hyperventilated until cessation of infusion.
Adverse events recorded and reported during time periods 0–30 min and 30–120 min

P-value, McNemar's test.
Discussion
The major outcome of the present study is that vasodilation of cranial arteries induced by one of the most potent vasodilators in parasympathetic nerve fibres, VIP, does not trigger migraine attacks in patients with migraine without aura.
Possible VIP involvement in migraine?
The presence of the trigemino-parasympathetic reflex is well established in animal and human experiments (39). Thus, injections of capsaicin to oral and nasal mucosa in rats resulted in lacrimation and an increase in dural and cortical blood flow mediated by parasympathetic autonomic fibres (40). Furthermore, capsaicin injected into the forehead of healthy volunteers leads to ipsilateral autonomic symptoms such as miosis, lacrimation and conjunctival injection (41). The origin of parasympathetic fibres innervating cerebral vessels is the sphenopalatine and otic ganglion (21). Experimental studies have reported that blocking of the sphenopalatine ganglion with intranasal lidocaine might be effective in the treatment of migraine attacks (42–44).
VIP and NO are co-transmitters in the PSNS and are both found in perivascular nerve fibres of porcine MCA (19), rabbit cerebral arteries (25) and in rat and rabbit parasympathetic ganglia (21, 25). Furthermore, it has been shown that VIP-mediated dilation of porcine basilar artery partly depends on NO (45). Vasodilation induced by parasympathetic stimulation (pig submandibular gland) or i.v. VIP may be inhibited by NO synthase inhibitors (18).
Collectively, these data suggest that nociception during migraine attacks may be triggered or mediated by activation of the PSNS. Moreover, i.v. infusion of VIP may lead to vasodilation co-mediated by NO release from parasympathetic efferents and thereby to migraine attacks in patients with migraine (7).
Why did VIP fail to induce migraine?
If VIP and NO coexist in the PSNS, one would expect the same response after i.v. VIP as after infusion of the NO donor, glyceryl trinitrate (GTN), i.e. initial vasodilation and induction of a delayed migraine attack (7). The mechanisms of NO-induced delayed migraine have not been fully clarified. Studying the transcriptional regulation of inducible NOS (iNOS) after GTN infusion and the effect of its inhibition in dura mater, a delayed induction (4–6 h) of iNOS in the dura mater of rodents after GTN infusion has been reported, and it has been suggested that up-regulation of iNOS may lead to migraine attacks in patients with migraine (46). Interestingly, nuclear factor (NF) κB activity is enhanced by GTN infusion and this is followed by iNOS expression (47). In contrast to NO, VIP and pituitary adenylate cyclase activating polypeptide prevented iNOS transcription in macrophages by inhibiting NFκB activation and interferon (IFN) regulatory factor-1 transactivation (48, 49). Furthermore, it has been shown that induction of iNOS in rat brain tissue treated with IFN-γ may be inhibited by VIP (50). These data show that NO and VIP act predominantly synergistically with respect to vessel tone, but the two transmitters may act as antagonists in inflammatory processes. This may explain why VIP failed to induce migraine attacks. Another possibility is that VIP might not have reached the relevant receptors and therefore failed to induce migraine pain. The effect of VIP is mediated mainly through VPAC1 and VPAC2 (VIP and PACAP) receptors, which have a binding affinity in the lower nanomolar range (51). There are contradictory results concerning VIP-receptor localizations. VPAC1 has been found on vascular smooth muscle cells in isolated cerebral arteries of the rat, and VIP-immunoreactive nerve fibres seem to have intimate contact to the receptor protein (52). Human basilar and MCAs showed receptor mRNA (VPAC1, VPAC2, PAC1) in samples with and without endothelium (53). Different VIP-receptors involved in vasodilation have been found in porcine basilar arteries: VPAC1 in the endothelium (NO- and endothelium-dependent), VPAC2 in the outer layers of the smooth muscles (NO- and endothelium-independent) and natriuretic peptide clearance receptor throughout the arterial wall including nerves (stimulation of nNOS in intramural nerve fibres) (45). At present, it is unclear if the different locations of the receptors are due to species differences, large vs. small arteries or basal vs. superficial arteries. Exogenous VIP, as in our study, reaches a high concentration on the endothelial VIP-receptors. Vasodilation caused by activation of these receptors is probably due to release of NO. It remains unknown to what extent VIP reached intramural VIP-receptors or possible VIP-receptors on sensory perivascular nerve terminals and whether these receptors are important for migraine induction. Furthermore, slow passage of VIP across the blood–brain barrier (54–56) may have prevented migraine if VIP-receptors on perivascular nerve terminals are relevant for migraine induction.
Experimental vasodilation and delayed migraine attack
Spontaneous migraine attacks are accompanied by dilation of the MCA (57). Experimental migraine induced by GTN, histamine, CGRP and dipyridamole are associated with initial vasodilation (7, 8, 58, 59), and it has been suggested that vasodilation may play a major role in triggering migraine attacks. It has been demonstrated, however, that sildenafil (a PDE 5 inhibitor) triggers migraine attacks without changes in the MCA diameter (34). Thus, migraine induction can occur without arterial dilation.
So far, all cerebral vasodilators that have been tested are able to induce headache in normal subjects and migraine in migraine sufferers. VIP is the first substance that markedly dilates intra- and extracranial arteries, but nevertheless does not induce migraine. These results further weaken the hypothesis of a purely vascular origin of migraine.
Footnotes
Acknowledgements
The authors thank lab technicians Kirsten Brunsgaard, Lene Elkjær and Juliano Olsen for their dedicated and excellent assistance. We also thank Associate Professor Peter Dalgaard from the Department of Biostatistics at the University of Copenhagen for statistical advice. The study was supported by grants from the University of Copenhagen, Denmark, the medical faculty (IMF) of the University of Münster, Germany, and from the Lundbeck Foundation under ‘Centre for Neurovascular Signaling’ (LUCENS).
